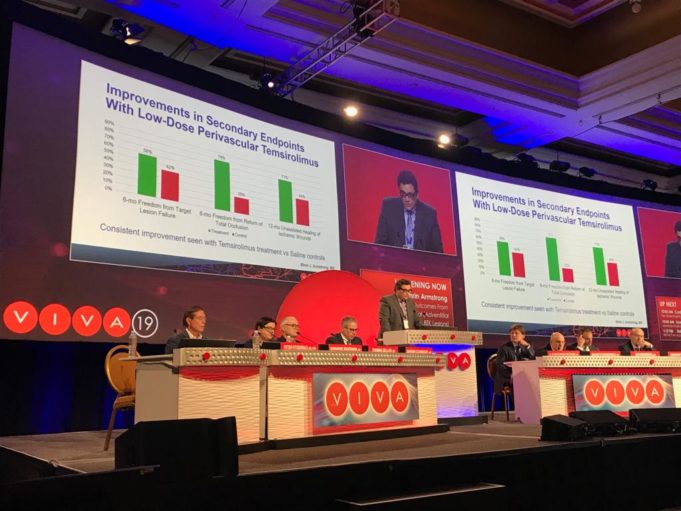
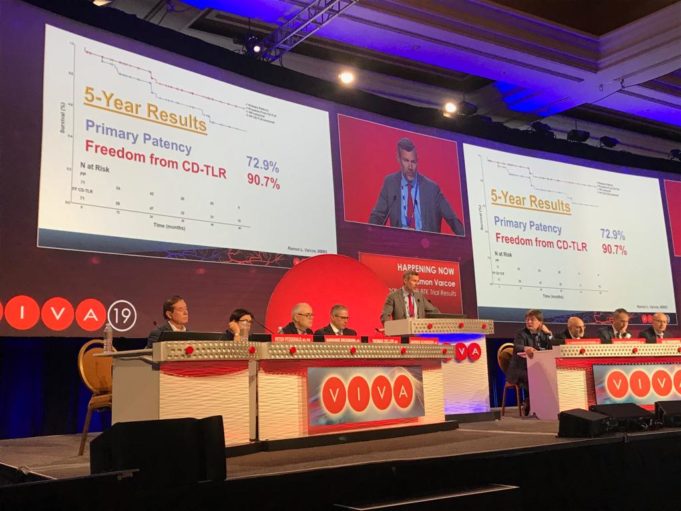
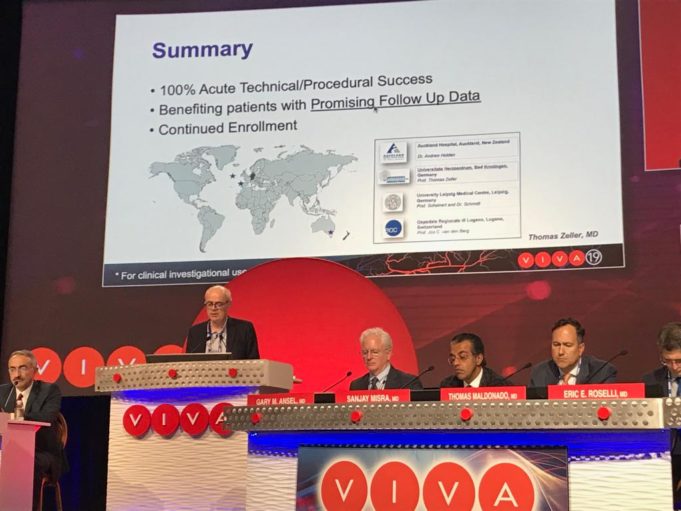
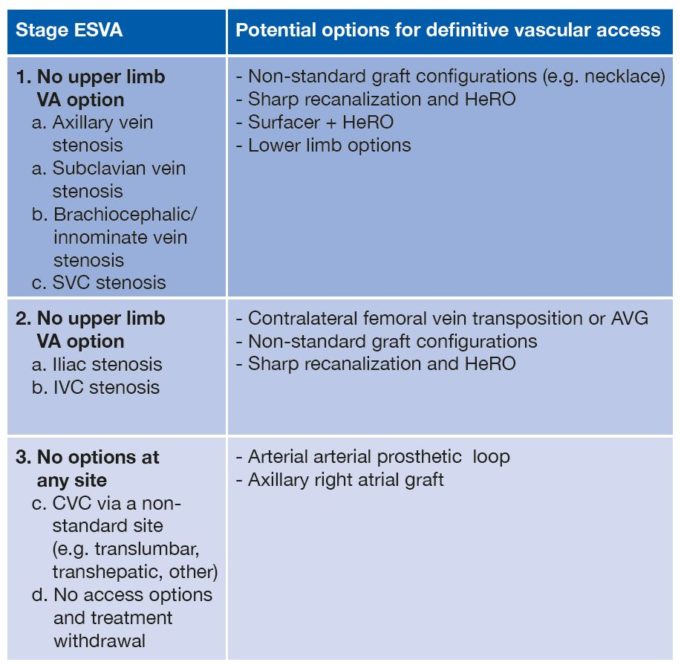
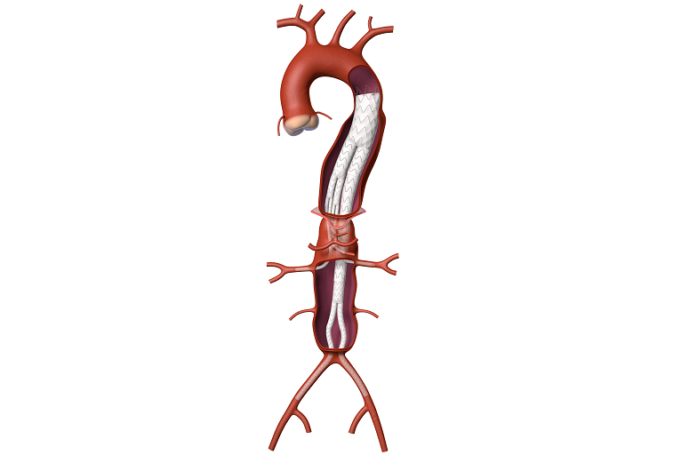
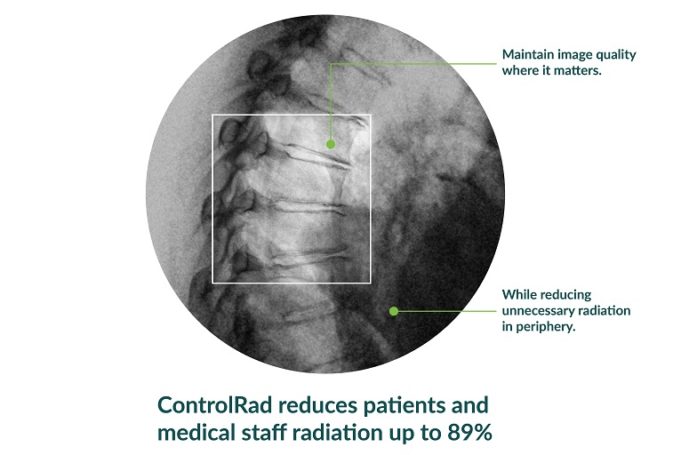
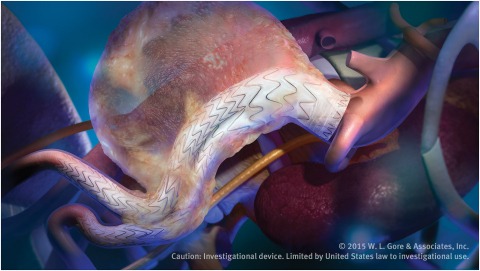
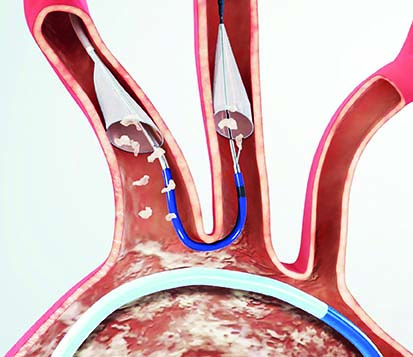
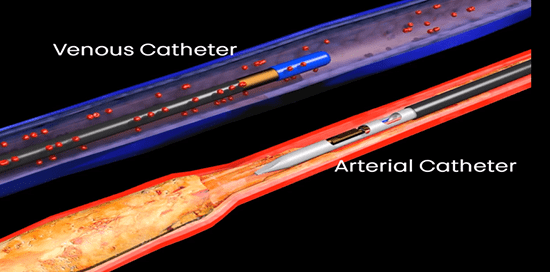
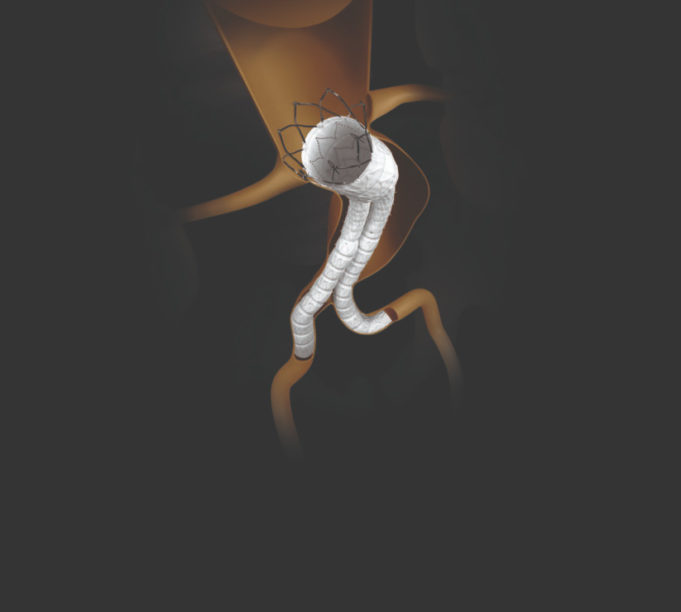The American College of Cardiology (ACC) and Society for Vascular Surgery (SVS) are collaborating on a single vascular registry, according to a press release, to harness the strengths of both organisations in improving care and outcomes of patients with...
MTF Biologics has announced that new positive coverage decisions by Aetna, Centene Corporation, WellCare and Tufts Health Plan will provide greater access to its human placental and dermal allografts, AmnioBand and AlloPatch, for the treatment of diabetic foot ulcers (DFUs).
In...
Rexgenero, a regenerative medicine company developing advanced cell therapies to treat chronic limb-threatening ischaemia (CLTI), has announced the acquisition of all the key assets of aratinga.bio SAS Group, a preclinical-stage immunotherapy biotechnology company based in Paris, France.
The acquisition, which...
For the CX 2020 LIVE Aortic Podium 1st Session, 489 participants from 62 countries tuned in to hear a series of experts examine technological and procedural developments in the field. Among topics covered was the early, yet promising, clinical...
The Society for Vascular Surgery (SVS) has released new clinical practice guidelines on the appropriate care and treatment of aneurysms of the visceral arteries.
These SVS evidence-based practice guidelines offer recommendations to inform the diagnosis, treatment options, screening and follow-up...
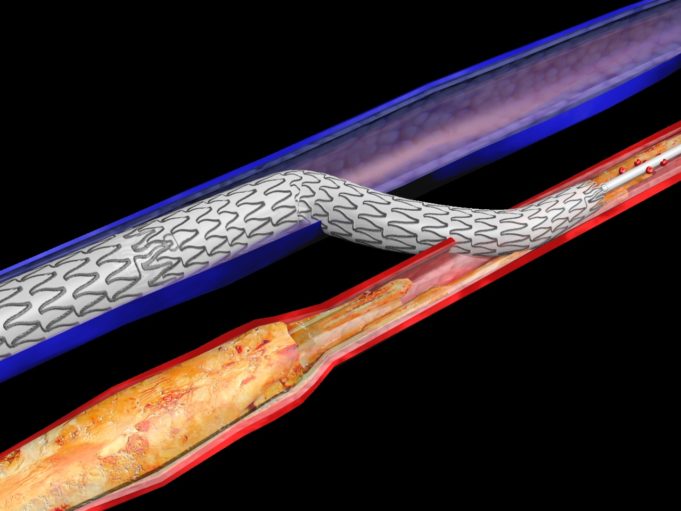
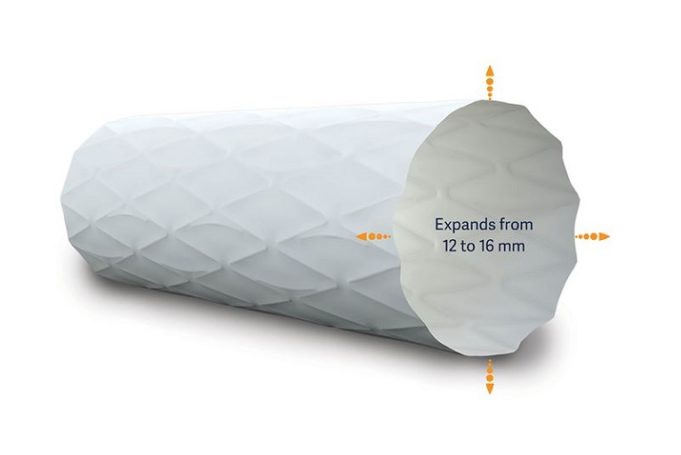
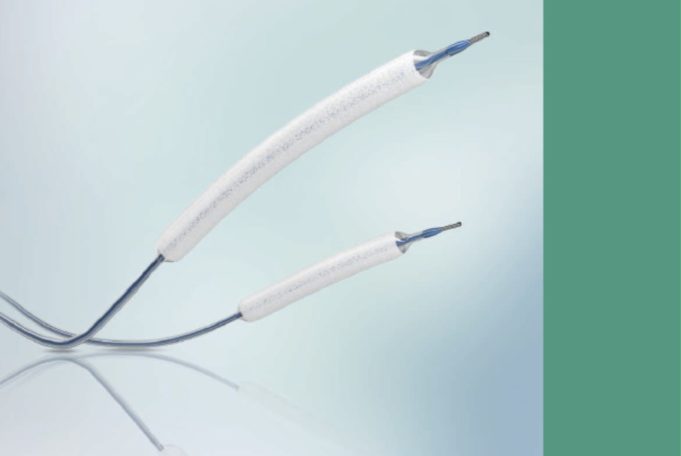
The Infinity Angioplasty Balloon Company has announced that its balloon platform—the Infinity angioplasty balloon catheter—has been cleared by the US Food and Drug Administration (FDA) for percutaneous treatment of peripheral arterial disease.
"The Infinity catheter represents a game changing...
In a new field safety notice, the UK Medicines and Healthcare products Regulatory Agency (MHRA) states that a warning and clinical summary section will be added to the instructions for use (IFU) of 12 paclitaxel-coated balloons and paclitaxel-eluting stents...
The first patients have been enrolled in the single-centre DEEPER LIMUS clinical trial, Reflow Medical has announced. The non-randomised pilot study will evaluate the Temporary Spur stent system for the treatment of lesions in the infrapopliteal arteries using a...
Surmodics recently announced it has received CE mark certification in the European Union for its SurVeil drug-coated balloon (DCB).
“I am excited about the potential of the SurVeil DCB to improve the treatment of PAD,” said Professor Marianne Brodmann, interventional...
The CX 2020 LIVE Vascular Access Consensus session sparked global interest, with chair Nick Inston (Birmingham, UK) and moderator Domenico Valenti (London, UK) taking questions from 17 countries, spanning South America to South East Asia. The session pinpointed the...
The Journal of Wound Care has republished a consensus document to tackle the rising rates of device-related pressure ulcers (DRPUs) among patients and now, due to COVID-19, health care professionals. The authors, Amit Gefen (Tel Aviv University, Tel Aviv, Israel) and Karen Ousey (University of Huddersfield, Huddersfield, UK) write that, amid...
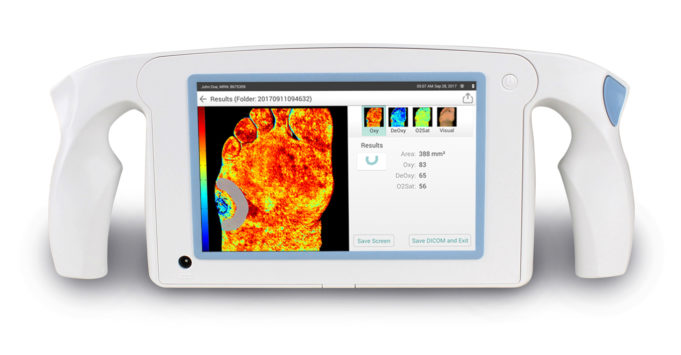
MolecuLight has announced that the Centers for Medicare & Medicaid Services (CMS) has assigned an Ambulatory Payment Classification (APC) code 5722 for the MolecuLight i:X imaging procedure for point-of-care real-time fluorescence wound imaging for bacterial presence, location, and load....
The 30th Annual Conference of the European Wound Management Association, EWMA 2020 will be a fully virtual conference, taking place on 18–19 November 2020.
The Association states: “The health and safety of our attendees, partners, vendors and staff is our...
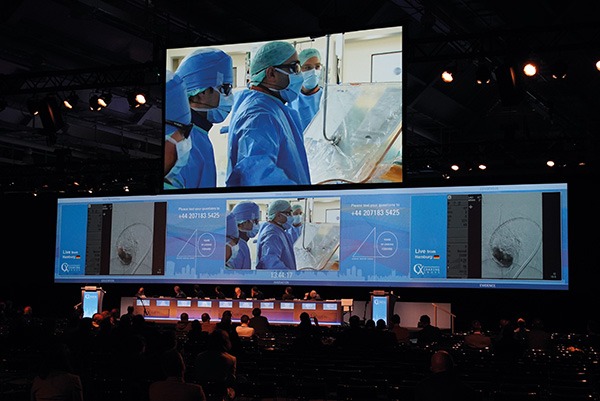
In a series of Podium 1st presentations from world-class faculty, CX 2020 LIVE attendees heard the latest data on peripheral arterial disease management and an evaluation of different types of stents, including covered and helical options for challenging lesions. In total, nearly 5,000 people, from 125 countries, have registered for the...
Vascular surgeons from across the USA are reporting health impacts on some patients, particularly elderly ones, in quarantine. The Society for Vascular Surgery (SVS) queried vascular surgeons regarding their concerns for patients’ health during the pandemic, and shared the findings...
Shockwave Medical announced today that the Centers for Medicare & Medicaid Services (CMS) has issued new codes for intravascular lithotripsy (IVL) procedures performed in peripheral arteries in both the hospital outpatient and inpatient settings.
The new Healthcare Common Procedure Coding...
Last week, the CX 2020 LIVE agenda turned to the technically challenging topic of aortic arch interventions. Through presentations, discussion, and polling, the session—chaired by Roger Greenhalgh (London, UK) and moderated by Stéphan Haulon (Paris, France)—aimed to tackle some...
In a boost for vascular education, the CX 2020 LIVE sessions have been granted European Union (EU) and US reciprocal Continuing Medical Education (CME) points in recognition of its highly interactive nature. In awarding this, the European Accreditation Council on CME (EACCME) is taking into account that this is...
Transit Scientific has received US Food and Drug Administration (FDA) clearance for the XO Score percutaneous transluminal angioplasty (PTA) scoring sheath platform for use in iliac, ilio-femoral, popliteal, infra-popliteal, and renal arterial plus synthetic and/or native arteriovenous hemodialysis fistula.
Angioplasty...
The US Food and Drug Administration (FDA) has designated Laminate Medical’s VasQ external support for the creation of arteriovenous fistulas (AVF) in haemodialysis patients as a breakthrough device.
According to a press release, the designation was based in part...
BD has announced the launch of the Halo One thin-walled guiding sheath, designed to perform as both a guiding sheath and an introducer sheath, for use in peripheral arterial and venous procedures requiring percutaneous introduction of intravascular devices.
According to...
Speaking from four separate rooms in four different corners of the world, William Ennis, Desmond Bell, Michael Edmonds, and William Li expounded over zoom on the myriad ways in which COVID-19 had impacted multidisciplinary wound care, both in terms...
Speaking from four separate rooms in four different corners of the world, William Ennis, Desmond Bell, Michael Edmonds, and William Li expounded over zoom on the myriad ways in which COVID-19 had impacted multidisciplinary wound care, both in terms...
VisionQuest Biomedical and the University of New Mexico School of Medicine have been awarded a three-year US$3 million grant from the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), part of the National Institutes of Health (NIH),...
Straub Medical has approved a full acquisition by Becton, Dickinson and Company (BD). The acquisition was completed on 20 April 2020. Straub Medical manufactures, develops, and sells medical devices for the treatment of arterial and venous diseases that are...
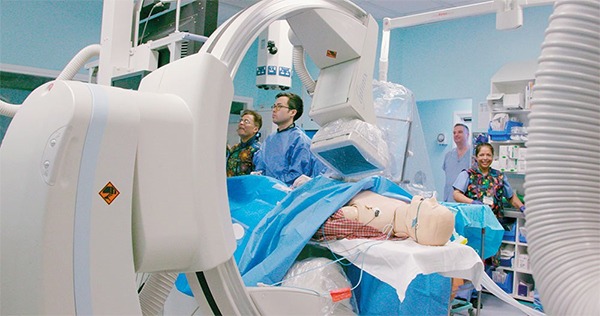
A global audience tuned into the second session and inaugural aortic offering of CX 2020 LIVE. With registrations now well over 4,000, the pioneering virtual vascular conference continues to attract delegates from far and wide. In this session, presentations,...
Mentice AB has announced the global introduction of its seventh-generation Vascular Interventional System Trainer (VIST) simulation platform.
According to the company, the VIST G7 includes artificial intelligence guidance, integration to robotics, and big data analytics. The newly redesigned platform of...
Merit Medical has received CE mark approval for the Wrapsody endovascular stent graft system from the British Standards Institution. The Wrapsody system is a flexible self-expanding endoprosthesis indicated for use in haemodialysis patients for the treatment of stenosis or...
Tokai Medical Products announced CE mark approval for its Rescue balloon occlusion catheter, a low-profile device designed for resuscitative endovascular balloon occlusion of the aorta (REBOA).
According to Tokai, REBOA uses a balloon catheter for temporary control of non-compressible torso...
A new study by researchers at the University of Maryland School of Medicine (UMSOM; Baltimore, USA) found that patients with an abdominal aortic aneurysm (AAA) received no benefits from taking a common antibiotic drug to reduce inflammation.
Patients who took...
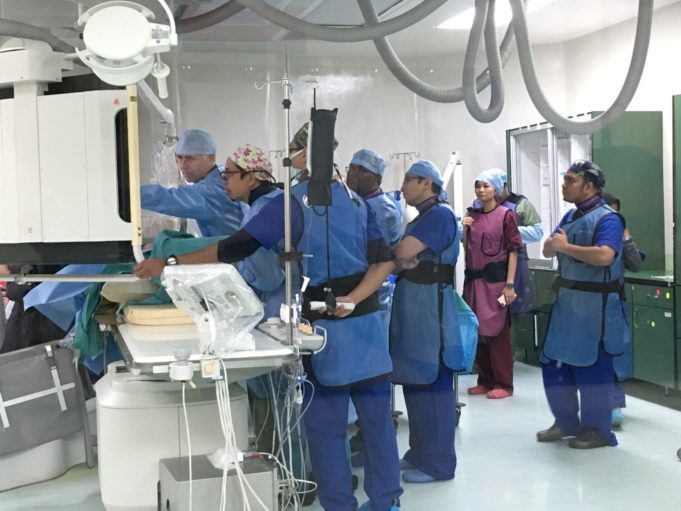
CX 2020 LIVE came to life online—despite COVID-19—using state-of-the art broadcast technology to bring together more than 1,000 vascular specialists, live, from 95 countries across the world. The first session of the highly interactive conference, designed to deliver global vascular...
New research published in the New England Journal of Medicine shows that the respiratory virus SARS-CoV-2, which causes COVID-19, causes severe damage to blood vessels, leading to widespread thrombosis, a press release by the Angiogenesis Foundation reports.
The study was...
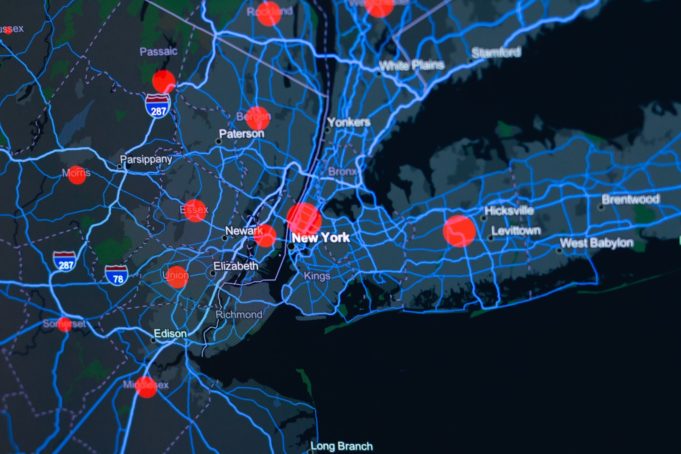
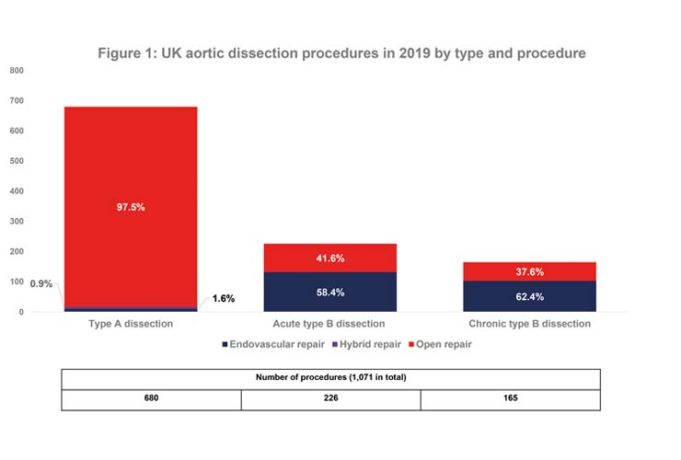
In a recently-published study, researchers found a significant drop in acute aortic dissection cases in New York City since the beginning of the COVID-19 pandemic.
The study—"The COVID-19 pandemic and acute aortic dissections in New York: A matter of public health"—has...
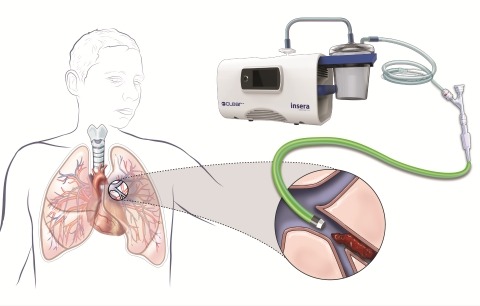
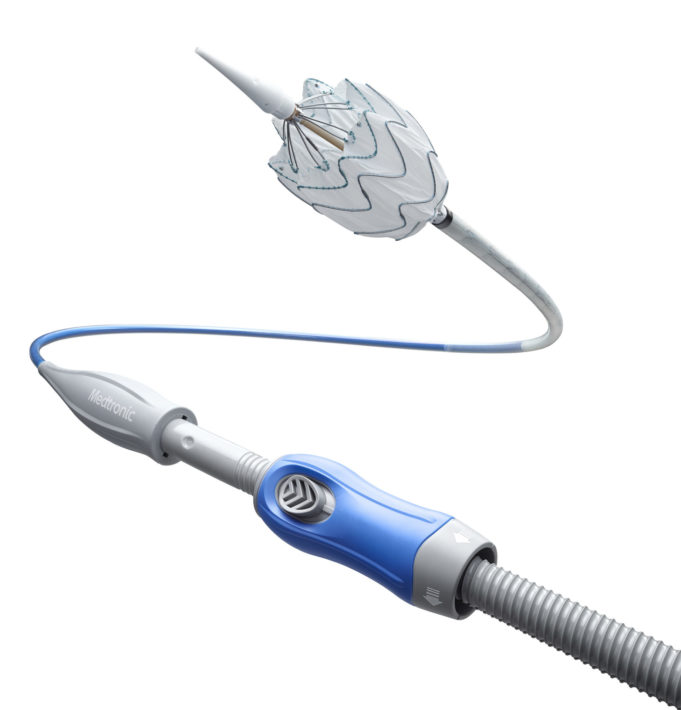
LimFlow SA today announced publication of positive two-year data from the ALPS registry of the LimFlow percutaneous deep vein arterialisation (pDVA) system. Results were published online yesterday in the Journal of Endovascular Therapy and will also appear in the...
LimFlow SA today announced publication of positive two-year data from the ALPS registry of the LimFlow percutaneous deep vein arterialisation (pDVA) system. Results were published online yesterday in the Journal of Endovascular Therapy and will also appear in the...
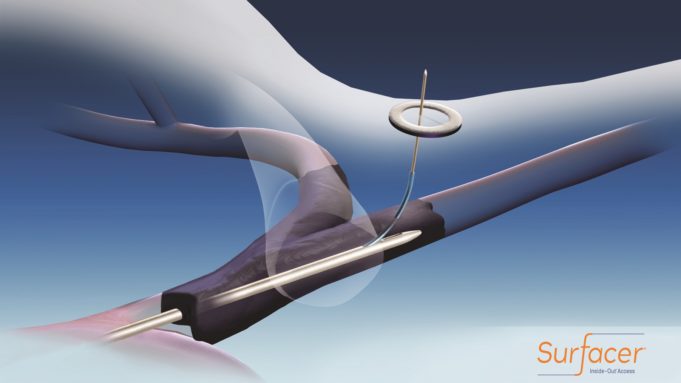
Bluegrass Vascular Technologies has announced that the first US commercial cases using the Surfacer Inside-Out access catheter system were completed at Santa Clara Valley Medical Center in San Jose, California.
The procedures were performed by Ehab Sorial and Ajit...
Getinge recently announced the launch of its Advanta V12 large diameter stent.
According to a press release, Getinge’s Advanta V12 stent has been shown to consistently improve patient outcomes by restoring iliac patency, reducing restenosis and re-intervention rates, improving ankle...
Older age, being male, deprivation, living in a densely populated area, ethnicity, obesity, and chronic kidney disease are associated with a positive test for COVID-19, according to results from 3,802 people tested for SARS-CoV-2 in the UK. The observational...
Older age, being male, deprivation, living in a densely populated area, ethnicity, obesity, and chronic kidney disease are associated with a positive test for COVID-19, according to results from 3,802 people tested for SARS-CoV-2 in the UK. The observational...
Roberto Chiesa (Milan, Italy), who was at the frontline of the COVID-19 pandemic in the Lombardy region of Italy, shares his team’s experience in tackling vascular and aortic emergencies during this time.
Chiesa outlines the response in the region to...
While more than 60% of medical students are female, the number of women in endovascular medicine is low, report Bella Huasen and Agnieszka Solberg. Here, for Vascular News, they discuss the reasons behind this under-representation and consider the importance...
Intact Vascular has announced the peer-reviewed publication of its Tack optimised balloon angioplasty (TOBA) III 12-month clinical trial results in the Journal of Vascular Surgery.
The multi-centre, single-arm, prospective study investigated the combination of the Tack endovascular system with Medtronic’s IN.PACT Admiral drug-coated balloon as...
Rebeca Reachi Lugo is a vascular surgeon in Mexico. Based on her own experiences, as well as the responses of 150 other Mexican vascular surgeons to a 10-question survey she conducted, Reachi Lugo tells Vascular News what life is...
The Charing Cross (CX) Symposium, with its world-class faculty and unique focus on live audience participation, has announced the launch of a not-to-be missed and timely vascular and endovascular education virtual event: CX 2020 LIVE.
Mark your calendars for this...
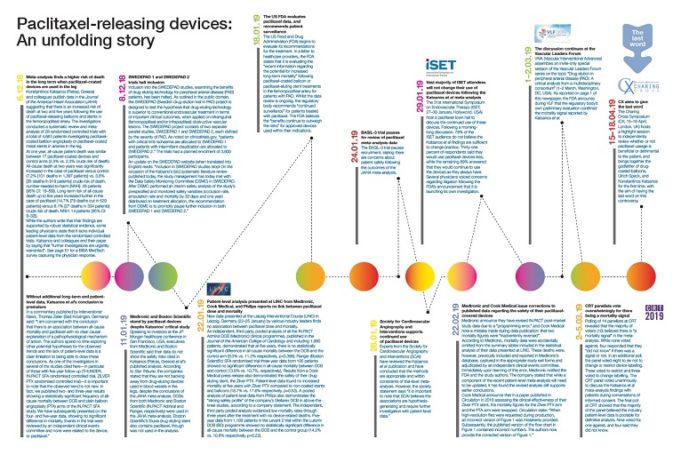
An individual patient-level data (IPD) analysis of the safety of paclitaxel-containing devices (PTXD), conducted by VIVA Physicians, identified an absolute 4.6% increased mortality risk associated with PTXD use.
The analysis, based on the most complete available data set of mortality...
Thomas Maldonado and Michael Barfield, both of NYU Langone Health (New York, USA), share their experiences of the coronavirus outbreak and discuss how the skills of vascular surgeons can be best utilised during the COVID-19 pandemic.
Maldonado says it is...
Endologix recently announced that it has completed enrolment in the EVAS2 confirmatory clinical study to evaluate the Nellix endovascular aneurysm sealing (EVAS) system.
“The completion of enrolment in our EVAS2 study is an exciting milestone for Endologix and for the...
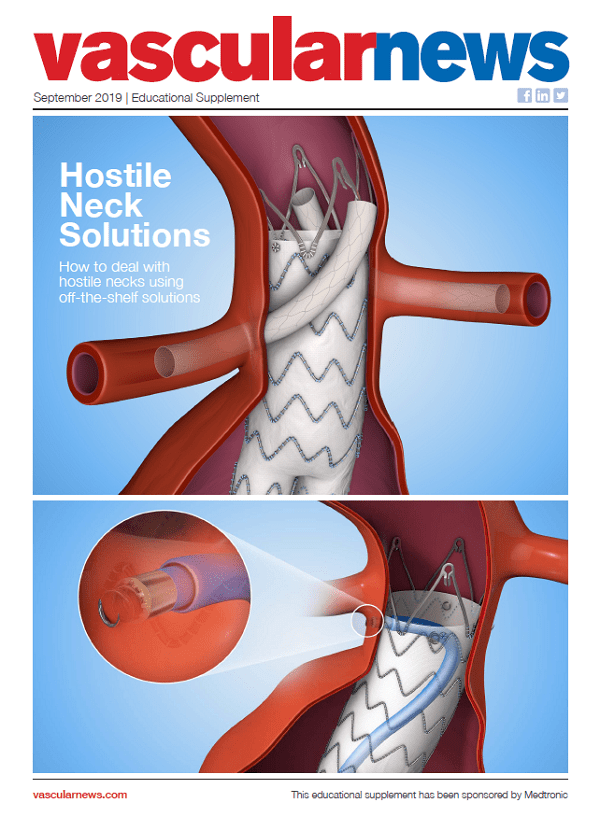
NOTE: This supplement is only available to read in selected countries and geographies
In this supplement, sponsored by Medtronic:
Fabio Verzini discusses his clinical experience with the new Valiant Navion™ thoracic stent graft, one year after its launch
Santi Trimarchi...
Therapeutic biologics company MiMedx has announced the appointment of William L Phelan as senior vice president and chief accounting officer, effective 1 May, 2020. Phelan is an accounting professional with more than 25 years of experience building, leading, and advising...
The Journal of Vascular Surgery (JVS) has introduced the COVID-19 Collection, a compilation of fast-tracked articles designed to proactively provide solutions to deliver the best care in the current COVID-19 environment.
JVS has dramatically shifted communication with vascular surgeons and vascular specialists...
Reviewing the available evidence for below-the-ankle interventions in the treatment of critical limb-threatening ischaemia (CLTI), Srini Tummala proposes that pedal artery intervention “should be an integral part of the armamentarium” when physicians are faced with patients with both below-the-knee...
The COVID-19 vascular service (COVER) study, a multinational, prospective, observational study that is aimed at capturing the impact of the pandemic on global vascular surgery, has gained ethical approval in the UK and is now recruiting worldwide.
An international Vascular...
Control Medical Technology has announced that the US Food and Drug Administration (FDA) has cleared its Aspire MAX 7 – 11F mechanical thrombectomy platform to remove blood clots from peripheral vessels.
"This FDA clearance quadruples our product offering and...
APERTO, Cardionovum’s paclitaxel-coated balloon for arterio-venous access, has received market approval for China, making it the first high pressure drug-coated balloon (DCB) available in China to treat obstructive lesions of native arterio-venous dialysis fistulae (AVF).
The approval was given based...
Terumo Aortic has announced that the US Food and Drug Administration (FDA) has granted approval of the Treo abdominal aortic stent-graft system for sale in the USA for the treatment of patients with abdominal aortic aneurysms (AAA).
According to a...
Cook Medical and Surmodics have announced an agreement in which Cook Medical will distribute two new Surmodics products, hydrophilic percutaneous transluminal angioplasty (PTA) balloon catheters that can be used over 0.014-inch and a 0.018-inch wire guides.
“We have had a...
The venous space continues to evolve in the United States, and many physicians are learning how to navigate through the challenges of the venous anatomy from their international peers. As a well-respected expert, the Galway-based interventional radiologist Dr. Gerard...
Vascular News talks with Gerard O'Sullivan, FSIR, FEBIR, University College Hospital, Galway, Ireland. This article is supported by BD Interventional.
Disclosures: Dr. Gerard O'Sullivan reports he is a consultant/speaker for BD Interventional, Aspirex, Boston Scientific Corporation, Cook Medical, Marvao Medical,...
Surgical navigation startup Centerline Biomedical, a Cleveland Clinic spin-off company, has announced the successful completion of the first in a series of clinical cases in the USA as it begins limited launch of its IOPS (Intraoperative positioning system) technology...
Prism Schneider (Cumming School of Medicine, University of Calgary, Calgary, Canada) and others write in a commentary in the Canadian Medical Association Journal—because of an increase in domestic violence during the pandemic—healthcare providers should be aware of the signs...
Prism Schneider (Cumming School of Medicine, University of Calgary, Calgary, Canada) and others write in a commentary in the Canadian Medical Association Journal—because of an increase in domestic violence during the pandemic—healthcare providers should be aware of the signs...
On 18 March, the Centers for Medicare & Medicaid Services (CMS) recommended “limiting non-essential care and expanding surge capacity into ambulatory surgical centres and other areas” to conserve resources and staff for managing COVID-19 patients. However, in a statement...
On 18 March, the Centers for Medicare & Medicaid Services (CMS) recommended “limiting non-essential care and expanding surge capacity into ambulatory surgical centres and other areas” to conserve resources and staff for managing COVID-19 patients. However, in a statement...
Terumo Aortic today announced that the US Food and Drug Administration (FDA) has granted breakthrough device designation for its Thoraflex Hybrid stented device for complex aortic arch repair.
The purpose of the FDA’s breakthrough device designation programme is to fast-track...
Results from a real-world experience over more than 10 years using the Endurant stent graft (Medtronic) demonstrate that it is safe and effective, with excellent long-term outcomes for abdominal aortic aneurysm (AAA) anatomy that falls both inside and outside...
Last week, the Charing Cross (CX) Symposium 2020 would have taken place in London, UK. To mark the occasion virtually, experts from across the globe took part in a wide-ranging, informal chat to share the impact of the COVID-19...
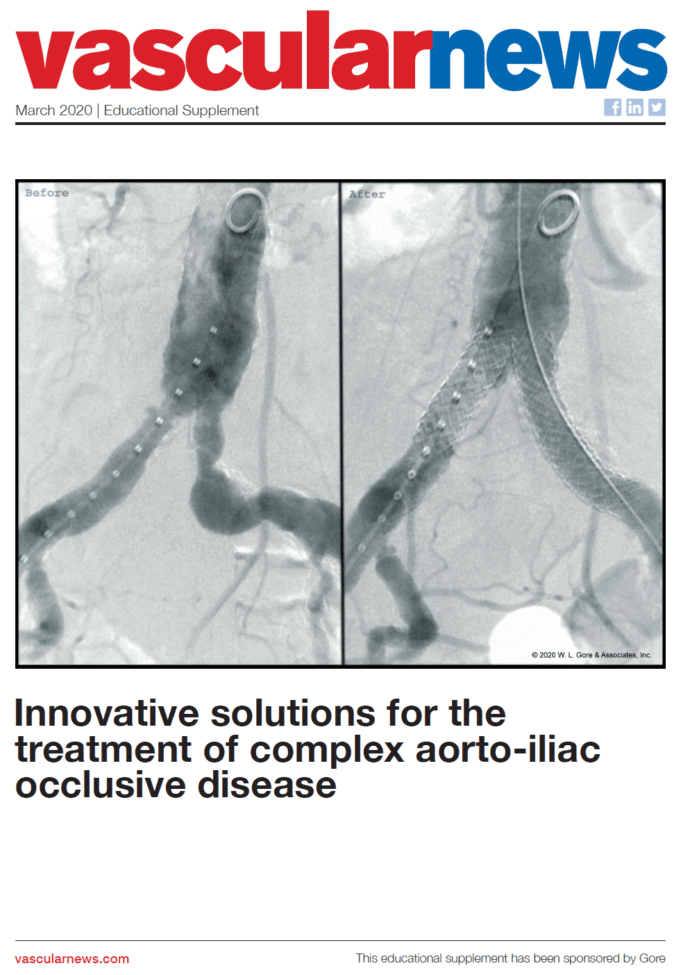
In this supplement, sponsored by Gore:
Michele Antonello considers the complexity of aorto-iliac occlusive disease, treatment gaps and current options
Jean Bismuth discusses results from the VBX FLEX study, considering the technical success of the data in comparison to...
Data from an unselected, real-world cohort of 64,000 claimants of the German BARMER Health Insurance reveals no signal of increased long-term mortality when paclitaxel-based devices are used in the femoropopliteal arteries, Eva Freisinger (Münster, Germany) told BLearning Peripheral at...
Since the outbreak of the COVID-19 pandemic, AOTI has been focusing all its resources on supporting a growing number of wound care patients being treated at home by the company’s multi-modality topical wound oxygen (TWO2) therapy.
In a press release,...
Intact Vascular has announced the first commercial use of its Tack Endovascular System (4F) in multiple sites across the USA. Notably the first FDA-approved vascular implant for below-the-knee (BTK) post-angioplasty dissection repair, Tack implants are designed to optimise balloon...
Intact Vascular has announced the first commercial use of its Tack Endovascular System (4F) in multiple sites across the USA. Notably the first FDA-approved vascular implant for below-the-knee (BTK) post-angioplasty dissection repair, Tack implants are designed to optimise balloon...
Endospan was recently granted breakthrough device designation from the US Food and Drug Administration (FDA) for the Nexus aortic arch stent graft system.
The FDA’s breakthrough device designation programme is intended to provide timely access to medical devices that have...
Several of the world’s ventilator manufacturers announce a newly formed Ventilator Training Alliance (VTA) to support frontline medical providers to access a centralised repository of ventilator training. The content can be found in a mobile app managed by Allego.
“Supporting...
On 15 April 2020, the Vascular Society of Great Britain and Ireland (Vascular Society) issued a comment on the recently published UK National Institute of Health and Care Excellence (NICE) guidelines on abdominal aortic aneurysm (AAA) diagnosis and management.
The...
i:X
MolecuLight has announced the integration of its MolecuLight i:X device with Tissue Analytics’ interconnected platform for seamless integration with leading electronic medical record (EMR)/electronic health record (EHR) platforms. According to a press release, Tissue Analytics is an artificial intelligence...
In a brief report in Annals of Internal Medicine, Seongman Bae (Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea) and colleagues report that neither surgical nor cotton masks appear to be an effective approach for preventing the dissemination of SARS-CoV-2 from...
In a brief report in Annals of Internal Medicine, Seongman Bae (Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea) and colleagues report that neither surgical nor cotton masks appear to be an effective approach for preventing the dissemination of SARS-CoV-2 from...
George Adams, MD (Raleigh, USA) talks to Vascular News at VIVA 2019 (Vascular InterVentional Advances; 4–7 November, Las Vegas, USA) about the six-month data from the TOBA II BTK pivotal study immediately after the late-breaking clinical trial session in which these data were...
George Adams, MD (Raleigh, USA) talks to Vascular News at VIVA 2019 (Vascular InterVentional Advances; 4–7 November, Las Vegas, USA) about the six-month data from the TOBA II BTK pivotal study immediately after the late-breaking clinical trial session in which these data were...
Intact Vascular has received US Food and Drug Administration (FDA) approval for the Tack Endovascular System (4F), a novel, minimal metal implant for precision dissection repair in the mid/distal popliteal, tibial and/or peroneal arteries.
Regulatory approval was based on data...
The Society for Vascular Surgery (SVS) Vascular Annual Meeting (VAM), which had been due to take place 17–20 June in Toronto, Canada, has been cancelled due to COVID-19.
The SVS posted the following statement on its website:
"After continuing to monitor...
Darren B Schneider is a vascular surgeon in New York—the US state with the highest number of COVID-19 confirmed cases and deaths at present. Here, for Vascular News, Schneider details how the role of a vascular surgeon has changed...
MedAlliance has announced enrolment of the first patient in the SAVE (Use of the Selution sirolimus-eluting balloon for dysfunctional AV access treatment indications) study utilising their novel sirolimus-eluting balloon, for the treatment of failed arteriovenous (AV) fistulae in renal...
Royal Philips has announced that the US government and Philips agreed to team up to increase the production of hospital ventilators in its manufacturing sites in the USA.
Philips plans to double the production by May 2020 and achieve...
Royal Philips has announced that the US government and Philips agreed to team up to increase the production of hospital ventilators in its manufacturing sites in the USA.
Philips plans to double the production by May 2020 and achieve...
VIVA Physicians recently hosted a virtual roundtable to address COVID-19. Tony Das (Connected Cardiovascular Care Associates, Dallas and BSW Heart Hospital, Plano, USA), one of the VIVA Board Directors, lead the discussion, addressing topics such as resource management, triage,...
Medtronic has announced updates regarding its efforts to increase ventilator production around the globe. The company is announcing progress in the ramp-up of its ventilator production, as well as collaborating with technology partners and governments to drive new ventilator...
Sebastian Debus (University Heart Center, Hamburg-Eppendorf, Germany) is joined by Birgitta Sigvant (Uppsala University, Uppsala, Sweden) and Rupert Bauersachs (Clinic Darmstadt, Darmstadt, Germany) for a special ESVS Educational Webinar: “From Science to Practice: Impact of the VOYAGER PAD trial...
The European Commission (EC) has adopted a proposal to postpone by one year the date of application of the new Medical Devices Regulation (MDR), which was due to come into force on 26 May this year. The postponement, a press...
The European Commission (EC) has adopted a proposal to postpone by one year the date of application of the new Medical Devices Regulation (MDR), which was due to come into force on 26 May this year. The postponement, a press...
Data, gathered in a study on the impact of aortic neck dilation after fenestrated endovascular aortic aneurysm repair (FEVAR), show that the process does indeed occur after the procedure—but with minimal clinical implications.
For principal evidence, the researchers pointed out,...
In a new statement published on 2 April 2020, all 45 societies represented by the US Council of Medical Specialty Societies (CMSS)—over 800,000 physicians—emphatically declare their belief that all frontline healthcare professionals must have access to personal protective equipment...
In a new statement published on 2 April 2020, all 45 societies represented by the US Council of Medical Specialty Societies (CMSS)—over 800,000 physicians—emphatically declare their belief that all frontline healthcare professionals must have access to personal protective equipment...
In response to the COVID-19 national crisis, Healogics has announced the launch of a Telehealth Programme. The programme is available immediately to Healogics' 600-plus wound care centres and 4,000 associated wound care providers, providing a means and set of...
The Society for Vascular Surgery (SVS) has collated a number of resources to provide information on COVID-19 and its impact, including advocacy, clinical practice guidelines, and a letter from the SVS president. The resource page can be found here.
The...
The American College of Surgeons (ACS) today leapt to the defense of healthcare workers who are heading to work in the face of increasing concerns over personal protective equipment (PPE) shortages as the COVID-19 pandemic cuts trails across the...
The American College of Surgeons (ACS) today leapt to the defense of healthcare workers who are heading to work in the face of increasing concerns over personal protective equipment (PPE) shortages as the COVID-19 pandemic cuts trails across the...
According to a statement, soft tissue repair company Aroa Biosurgery is stepping up to support clinicians and their patients who are battling to treat wounds, amid disruption to many existing US facilities for wound care due to COVID-19.
Aroa vice...
In the face of increasing pressure on healthcare systems, caused by the COVID-19 pandemic, the International Working Group on the Diabetic Foot (IWGDF)—in collaboration with D-Foot International and Diabetic Foot Australia—has set out to answer clinicians’ questions, particularly on...
The imperatives of preserving personal protective equipment (PPE) in the throes of the COVID-19 pandemic were discussed in depth during a Society for Vascular Surgery (SVS) Town Hall on the speciality’s early experiences with the coronavirus strain.
Some of the...
The Imperial College Network of Excellence in Vascular Science at Imperial College London (UK) is to run a COVID-19 Cardiovascular Conference as a live webinar on Thursday 2 April for all healthcare professionals battling COVID-19.
The webinar aims to share...
“Our goal is to reduce the burden on the healthcare system by keeping patients safe, functional, and at home during the COVID-19 pandemic,” state the authors of a new viewpoint published in the Journal of the American Podiatric Medical...
Medtronic has announced it is publicly sharing the design specifications for the Puritan Bennett 560 (PB 560) to enable participants across industries to evaluate options for rapid ventilator manufacturing to help doctors and patients dealing with COVID-19. This decision...
According to a statement, minor cuts and lacerations can be safely, quickly, and effectively treated at home with Clozex Medical innovative wound closures, reducing the risk of exposure to COVID-19 and other contagions.
"Stay safe and keep our medical professionals...
“More bleeding with background clopidogrel, even if not severe by adjudication, may be associated with broad consequences, including discontinuation of therapies. In the absence of clear benefit, clopidogrel exposure along with aspirin and rivaroxaban should be minimised or avoided...
Three months after undergoing renal denervation, patients with untreated high blood pressure had statistically significant reductions in average blood pressure over 24 hours compared with patients who underwent a sham procedure according to results from the SPYRAL-HTN OFF MED pivotal...
In stable atherosclerosis, a combination of aspirin plus rivaroxaban provided a similar relative degree of benefit on coronary, cerebrovascular, and peripheral endpoints in patients with and without diabetes, a prespecified analysis of the COMPASS trial has shown. It also...
Treatment with aspirin plus rivaroxaban following lower extremity revascularisation in patients with peripheral arterial disease (PAD) leads to a 15% reduction in the risk of major adverse limb and cardiovascular events when compared with aspirin alone.
The VOYAGER PAD study...
During a Q&A section of a European Commission (EC) college meeting on 25 March, EC spokesperson Stefan de Keersmaecker stated that the commission were looking to delay the “entry into force” of the new European medical device regulations (MDR) because of...
During a Q&A section of a European Commission (EC) college meeting on 25 March, EC spokesperson Stefan de Keersmaecker stated that the commission were looking to delay the “entry into force” of the new European medical device regulations (MDR) because of...
Sophie Renton (London, UK) moderates a session with Michel Makaroun (Pittsburgh, USA) and Armando Mansilha (Porto, Portugal) where the trio discuss some of the challenges relating to recruitment within the field of vascular surgery.
Makaroun explains that there are two...
NOTE: This supplement is only available in selected countries and geographies
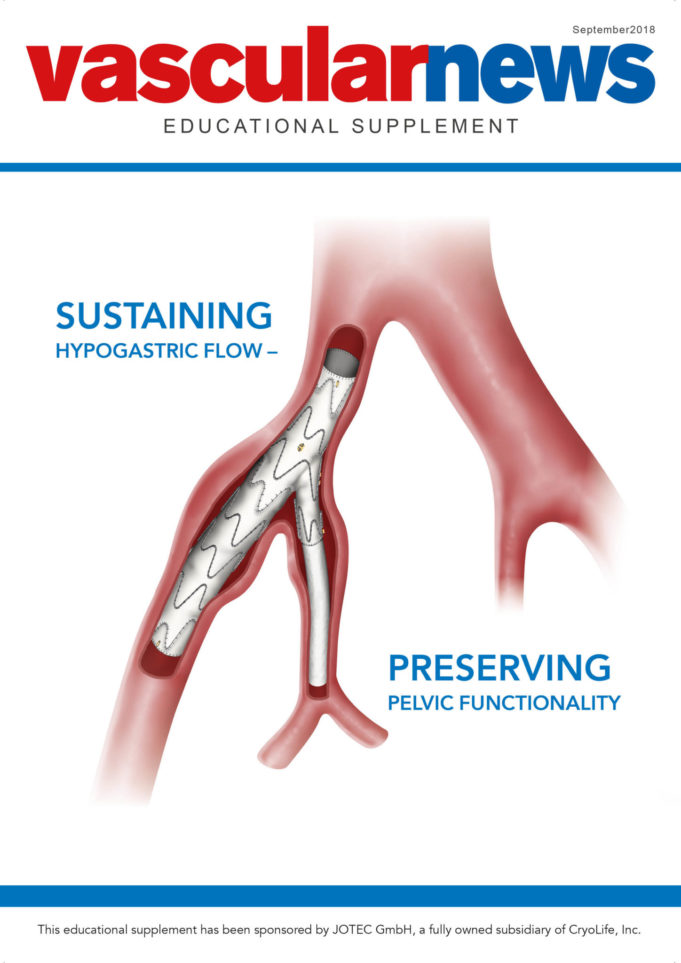
In this supplement, sponsored by CryoLife:
Gottfried J Mommertz outlines the benefits of JOTEC's E-tegra for the treatment of complex aneurysms and details a case study using the device
...
Thirty-day and one-year data from the STABLE II study have been published in the April 2020 issue of the Journal of Vascular Surgery (JVS) by Joseph V Lombardi (Cooper University Hospital, Camden, USA) and colleagues. The results demonstrate favourable...
Wound dressings in the UK are changed on average every three days, according to a new survey, conducted by the Journal of Community Nursing on behalf of Mölnlycke, of over 300 healthcare professionals, including tissue viability nurses, district nurses,...
A viewpoint in the Journal of the American Medical Association (JAMA) has offered potential solutions to modifying ongoing randomised clinical trials during the COVID-19 pandemic. It aims to “minimise disruption and preserve integrity”, while still ensuring participant health and safety.
Authors Mary...
A viewpoint in the Journal of the American Medical Association has offered potential solutions to modifying ongoing randomised clinical trials during the COVID-19 pandemic. It aims to “minimise disruption and preserve integrity”, while still ensuring participant health and safety.
Authors...
Use of autologous skin cell suspension (ASCS) in addition to compression has been shown to accelerate healing in large venous leg ulcers (VLUs), according to the results of a multicentre randomised trial conducted in the UK. Writing in the...
The American College of Surgeons (ACS) has announced the release of new clinical guidance for surgeons to curtail recommendations for elective surgical procedures, with the aim of preserving necessary resources for the care of critically ill patients during the...
The American College of Surgeons (ACS) has issued guidance for surgeons to curtail recommendations for elective surgical procedures to preserve the necessary resources for care of critically ill patients during the COVID-19 pandemic.
To assist in surgical decision-making in curtailing...
In a statement from the Alliance of Wound Care Stakeholders, a multidisciplinary trade association of physician specialty societies and clinical and patient associations, it has been emphasised that wound care is an essential—not elective—service that prevents admissions to hospital...
The 2020 Charing Cross (CX) Symposium that was due to take place next month in London, UK, has been cancelled due to COVID-19.
In full, the statement from the CX Symposium team, reads:
"The CX Symposium team has made the difficult...
The 2020 Charing Cross (CX) Symposium that was due to take place next month in London, UK, has been cancelled due to COVID-19.
In full, the statement from the CX Symposium team, reads:
"The CX Symposium team has made the difficult...
The US Food and Drug Administration’s (FDA) Center for Devices and Radiological Health (CDRH) has written to the medical device industry to outline its response to the COVID-19 public health emergency in its day-to-day operations with industry.
The letter from...
The president of the Vascular Society of Great Britain and Ireland, Chris Imray (University Hospitals Coventry and Warwickshire NHS Trust, Coventry, UK), has issued a letter to Vascular Society members on the COVID-19 virus and vascular surgery, offering “general...
A new Heart & Stroke guideline published today in the Canadian Medical Association Journal does not recommend taking ASA (Acetylsalicylic acid) as a preventive measure for those who do not have a history of stroke or heart or vascular disease,...
With healthcare systems around the world facing the most severe global health crisis in a generation, wound care is one of many areas that could be seriously affected. As iWounds News editor-in-chief William J Ennis explains, the commonality between...
Amid the COVID-19 pandemic, Benjamin W Starnes and Niten Singh urge service chiefs across the USA to "act this minute" in order to conserve resources and save lives.
We live in unprecedented times. On 19 January, patient zero arrived in...
A five-year institutional population health review has revealed that there is a rising trend in the clinical and economic burden of wound care in the Tropics. Published in the International Wound Journal (IWJ), the study was conducted after two...
In this issue:
CX 2020 highlights
A year in profiles
New meta-analysis finds “no observed difference” in mortality between paclitaxel and uncoated device use in CLTI patients (p. 20)
Centralisation of aortic surgery is “imperative” (p. 24)
In this issue:
CX 2020 highlights
A year in profiles
New meta-analysis finds "no observed difference" in mortality between paclitaxel and uncoated device use in CLTI patients (p. 20)
Centralisation of aortic surgery is "imperative" (p. 24)
Straub Medical (United States), the US direct-sales subsidiary of Straub Medical AG (Switzerland), has announced the launch and first intervention performed with the Rotarex S atherectomy device designed for treating occlusive peripheral vascular disease (OPVD).
Baljeet Uppal (Pinnacle Vein and Vascular Center, Sun City, USA), who...
The UK National Institute for Health and Care Excellence (NICE) has today published guidelines on abdominal aortic aneurysm (AAA) diagnosis and management. Discussion between physicians and patients to consider the risks of any intervention versus no intervention is encouraged,...
A retrospective analysis of patients diagnosed with lower extremity lymphoedema, and treated in a cancer-affiliated physical therapy department, has found that chronic venous insufficiency (or phlebolymphoedema) is the “predominant cause of lower extremity lymphoedema”. Published online in the Journal...
President-elect of the Society for Vascular Surgery, Ronald Dalman, talks to Vascular News about various aspects of his career. He also considers future development in the field and offers advice to aspiring surgeons.
What led you to become a vascular...
In this issue of iWounds News:
How wound care specialists can improve microcirculation
Multicentre RCT shows superiority of PICO NPWT system
Updated guidelines reveal pressure ulcer damage cascade
Thomas Serena: Wound care cooperation
Michael Edmonds: Avoiding amputation
Profile: Una Adderley
In this issue of iWounds News:
How wound care specialists can improve microcirculation
Multicentre RCT shows superiority of PICO NPWT system
Updated guidelines reveal pressure ulcer damage cascade
Thomas Serena: Wound care cooperation
Michael Edmonds: Avoiding amputation
Profile: Una Adderley
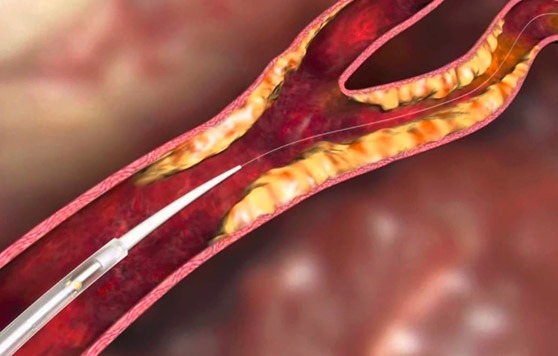
Endologix recently announced that it has received approval from the US Food and Drug Administration (FDA) for the Alto abdominal stent graft system (Alto).
The company received approval based on its regulatory submission that includes the ELEVATE investigational device exemption...
The Lindsay Leg Club Foundation has launched a new fundraising campaign in partnership with the Aviva Community Fund, in a bid to raise money for a new Leg Club. There are currently 44 Leg Clubs operating throughout the UK,...
The American Journal of Kidney Diseases (AJKD) has published the National Kidney Foundation's KDOQI Clinical Practice Guideline for Vascular Access: 2019 Update, a completely revamped set of 26 individual guidelines for clinicians that focuses on dialysis access choices tailored...
The 32nd International Symposium on Endovascular Therapy (ISET; 22–25 January, Hollywood, USA) had a particular focus on treating critical limb-threatening ischaemia (CLTI). In the session “Improving outcomes and prognosis for CLI”, conference attendees learnt about the global scale of...
The 32nd International Symposium on Endovascular Therapy (ISET; 22–25 January, Hollywood, USA) had a particular focus on treating critical limb-threatening ischaemia (CLTI). In the session “Improving outcomes and prognosis for CLI”, conference attendees learnt about the global scale of...
The early months of 2020 have seen the emergence of new technologies in the intraoperative navigation space. Here, Vascular News considers Philips’ Fiber Optic RealShape (FORS) technology and Centerline Biomedical’s IntraOperative Positioning System (IOPS)—data on which have been presented...
At this year’s Leipzig Interventional Course (LINC 2020; 28–31 January, Leipzig, Germany), William Gray (Main Line Health, Philadelphia, USA) considered the future of drug-coated devices and what to expect over the next five years. He suggests that while the...
“Venous leg ulcers (VLUs) secondary to deep venous stenosis represents a distinct class of patients who require a unique treatment paradigm,” stated Abhisekh Mohapatra (University of Pittsburgh Medical Center, Pittsburgh, USA), presenting results of a multicentre retrospective study of...
Reflow Medical has announced that they have received US Food and Drug Administration (FDA) clearance for an expanded indication for the Wingman crossing catheter after completing the Wing-IT clinical trial. The Wingman catheter crosses peripheral chronic total occlusions (CTOs)...
Okami Medical has announced the successful completion of the first cases with the Lobo vascular occlusion system. The first offering in the company's product portfolio, Lobo-3, recently received 510(k) clearance from the US Food and Drug Administration (FDA) for...
CryoLife has announced that it has received CE mark for the E-vita Open NEO, a hybrid stent graft system for the treatment of aortic arch disease.
Many patients with either an aneurysm or dissection in the aortic arch also present...
A short film (“Today is a Good Day”), which showcases the work of Aortic Dissection Awareness UK & Ireland, has won the Rare Disease Film Festival’s Peoples’ Choice award. According to Rare Disease UK, the organisers of the event, the film festival...
AVITA Medical has announced the initiation of a study into soft tissue reconstruction with the enrolment of the first patient at the Arizona Burn Center (Valleywise Medical Health Center, Phoenix, USA). According to a statement, this study will evaluate...
NOTE: This video is ONLY available to watch in selected countries and geographies
Andrew Holden (Auckland, New Zealand), Alexandros Mallios (Paris, France), Robert Lookstein (New York, USA) and Tobias Steinke (Dusseldorf, Germany) talk to BLearning Peripheral about the 12-month results...
MolecuLight has announced the publication of "In vitro detection of porphyrin-producing wound bacteria with real-time fluorescence imaging" in Future Microbiology. The peer-reviewed paper describes the results of an extensive in vitro validation study using the MolecuLight i:X fluorescence imaging system to detect common wound pathogens.
In this...
Smith+Nephew has announced the US launch of the new PICO 14 Single Use Negative Pressure Wound Therapy System (sNPWT), which has a pump duration of up to 14 days. According to a statement, the new pump builds on the...
Edward Choke (Singapore) talks to BLearning Peripheral at LINC 2020 (Leipzig Interventional Course; 28–31 January 2020, Leipzig, Germany) about the first-in-man clinical study—XTOSI—which looked at the safety and efficacy of a sirolimus drug-coated balloon (DCB) in the treatment of peripheral arterial disease.
The interim...
While drug-coated balloons (DCBs) have been “consistently efficacious” across various studies, the mortality signal has not, Peter Schneider (University of California San Francisco, San Francisco, USA) noted at this year’s Leipzig Interventional Course (LINC 2020; 28–31 January, Leipzig, Germany)....
The Charing Cross Symposium (CX; 21–24 April, London, UK) is the longest-running vascular and endovascular global symposium, and an important source of Education, Innovation, and Evidence as the vascular field in 2020 seeks clarity and truth. This year, the...
Ra Medical Systems has announced enrolment of the first patient in its pivotal study to evaluate the safety and effectiveness of the DABRA excimer laser system for use as an atherectomy device for the treatment of peripheral vascular stenoses.
“My...
Cerebrospinal fluid (CSF) drainage is a key component in the treatment of spinal cord injury but the evidence that prophylactic drainage decreases such injury during endovascular repair (EVAR) is “at best weak,” Gustavo Oderich (Mayo Clinic, Rochester, USA) told...
CryoLife has announced it has received CE mark for the On-X Ascending Aortic Prosthesis (AAP), allowing resumed distribution in the European Union (EU). The On-X AAP is indicated for the treatment of diseased, damaged or malfunctioning native or prosthetic...
Biotronik has announced three-year data from the Bioflow-V trial, which was presented recently at the 2020 Cardiovascular Research Technologies (CRT) conference. The three-year follow-up data demonstrates consistently lower clinical event rates and improvement in patient outcomes, reinforcing the differentiation...
Alberto Muñoz (Bogota, Colombia) talks to BLearning at CX 2019 (Charing Cross Symposium; 15–18 April, London, UK) about how the meeting provides a “great opportunity” to update attendees on the latest in vascular and endovascular surgery.
Muñoz goes on to highlight the international flavour...
Pharmaceutical company PolyPid has announced that the first patient has been enrolled and randomised in a phase 3 clinical trial called SHIELD (Surgical site Hospital-acquired Infection prEvention with Local D-plex). SHIELD will evaluate PolyPid's D-PLEX100 plus standard of care,...
A combined literature review and retrospective study has confirmed that the use of a dermal regenerative template (DRT) is “a viable option for providing vascularised coverage over wounds with avascular components”. Moreover, it was also found by Ronald N...
A combined literature review and retrospective study has confirmed that the use of a dermal regenerative template (DRT) is “a viable option for providing vascularised coverage over wounds with avascular components”. Moreover, it was also found by Ronald N...
As director of the National Wound Care Strategy, a programme that aims to improve the quality of wound care across the UK, Una Adderley is one of several figures in the nursing community leading the drive for change. Speaking...
Described as a “Cinderella condition”, Charcot foot is little understood within the non-specialist medical community. In this article, Benjamin Bullen (Cardiff Metropolitan University, Cardiff, UK) presents a multidimensional conceptualisation of health literacy and underlines the importance of developing knowledge...
Investigators at the University of Southern California, Los Angeles, USA, have developed a severity adjustment model for predicting the healing trajectory of chronic wounds within 12 weeks. The research, published online ahead of print in Advances in Wound Care,...
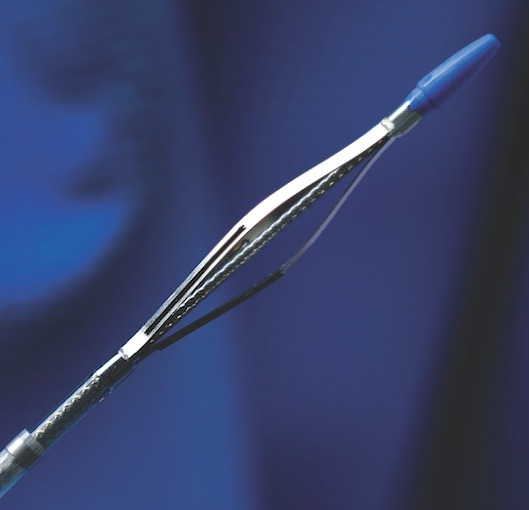
Fist Assist Devices has received CE mark and has begun marketing the Fist Assist device for vein enhancement and enlargement in the European Union and other CE mark countries. Fist Assist is the only wearable, patent protected, intermittent compression...
Results of a retrospective cohort study, published online in Wounds, have demonstrated that a combination of advanced treatment modalities—such as negative pressure wound therapy (NPWT)—and the use of oxidised regenerated cellulose (ORC), collagen or silver-ORC dressings, contributes to an...
MolecuLight has announced the release of a "significant upgrade" to its i:X digital wound measurement feature that enables clinicians to more accurately and quickly capture digital wound area measurement. According to a statement, clinicians can now reliably detect more complex...
Ablative Solutions has announced that positive six-month results from the Peregrine post-market study demonstrating the safety and efficacy of the company’s CE-marked Peregrine System infusion catheter were published in the Journal of the American College of Cardiology: Cardiovascular Interventions.
The Peregrine...
February 2020 brings another paclitaxel device meta-analysis of randomised controlled trials in chronic limb-threatening ischaemia (CLTI) patients. Krystal Dinh (Westmead Hospital, Sydney, Australia) et al report online ahead of print that the risk of all-cause mortality after treatment with...
For the first time, faculty in the biomedical engineering department, University of Connecticut, Mansfield, USA, have designed a wirelessly-controlled, or “smart”, bandage and a corresponding smartphone-sized platform that can precisely deliver different medications to the wound with independent dosing.
Chronic...
Peter Schneider (San Francisco, USA) talks to BLearning Peripheral at LINC 2020 (Leipzig Interventional Course; 28–31 January 2020, Leipzig, Germany) about the relevance of dose relationship and geographical data when discussing the controversy surrounding the use of paclitaxel devices in peripheral arterial disease...
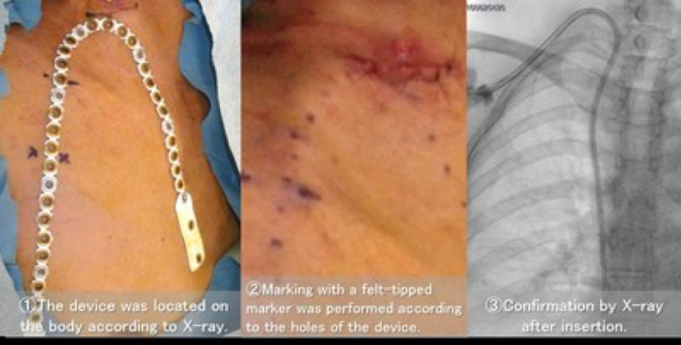
Robert Shahverdyan, head of the Vascular Access Center at Asklepios Klinik Barmbek, Hamburg, Germany, has recently published a retrospective analysis of his first 32 consecutive radiocephalic (forearm) VasQ External Support procedures in the Journal of Vascular Access. The paper...
Cerus Endovascular has received CE mark approval for its lead product, the Contour Neurovascular System, for the treatment of intracranial aneurysms. The system incorporates a fine mesh braid that is deployed across the neck of the aneurysm sac and...
Use of incisional negative pressure wound therapy (NPWT) does not change the rate of deep surgical site infection, for patients undergoing surgery for major trauma-related lower limb fractures, according to results of the WHIST randomised clinical trial. Published online...
President of the German Wound Council, Martin Storck, considers how to improve the treatment of chronic wounds. While the road to better treatment “may still be long,” he says, “it is a road worth going down”.
“On average, a patient...
President of the German Wound Council, Martin Storck, considers how to improve the treatment of chronic wounds. While the road to better treatment “may still be long,” he says, “it is a road worth going down”.
“On average, a patient...
Preliminary experience with left subclavian artery (LSA) branched devices indicates low morbidity and stroke rates with high patency rates. This was the conclusion of Gustavo Oderich (Mayo Clinic, Rochester, USA) at this year’s Controversies and Updates in Vascular Surgery...
At this year’s Critical Issues America annual meeting (Coral Gables, USA), Bijan Modarai (Guy’s and St Thomas’ NHS Foundation Trust and King’s College London, London, UK) gave a progress update on forthcoming European Society for Vascular Surgery (ESVS) radiation...
Aurealis Therapeutics has announced that the first diabetic foot ulcer (DFU) patient has been dosed with AUP-16, its three-in-one combination biologic drug for chronic non-healing wounds and regenerative diseases. The trial, currently being performed in non-healing DFU patients, is...
University Hospitals Ahuja Medical Center (UH) has been revealed as the first clinical setting in Ohio using special technology to identify harmful bacteria, leading to healing for patients suffering from chronic wounds.
Windy Cole, medical director of the UH Ahuja...
Bluegrass Vascular Technologies (Bluegrass Vascular) announced today that the US Food and Drug Administration (FDA) has granted a de novo classification order for its Surfacer Inside-Out access catheter system. The Surfacer system is intended to obtain central venous access...
Healogics has announced the newest release of WoundSuite Navigator, a physician relationship management mobile app. WoundSuite Navigator is Healogics' proprietary tool and is available on the WoundSuite Center Management platform.
According to a statement, the latest release includes optimisations...
Rist Neurovascular recently announced that it has received US Food and Drug Administration (FDA) 510(k) clearance to market the Rist Cath Radial Access Long Sheath (Rist Cath) for the introduction of interventional devices into the peripheral, coronary, and neurovascular...
Dierk Scheinert (Leipzig, Germany) moderates a panel with Eva Freisinger (Münster, Germany) and Thomas Zeller (Bad Krozingen, Germany) to discuss the developments over the past 12 months surrounding the safety of paclitaxel.
Zeller notes that further analyses of the randomised...
XableCath has announced that its XableCath Crossing catheters have received CE mark for peripheral use. Its crossing devices will be available for sale in Europe in two versions, a blunt tip and an abrasion tip, in a variety of...
Venous News filmed a panel discussion with Alun Davies (London, UK), Manj Gohel (Cambridge, UK) and Daniel Carradice (Hull, UK); who discussed treatment of venous ulceration and the randomised EVRA trial which, Carradice says, showed that early endovenous ablation of superficial venous...
Auckland-based Aroa Biosurgery has announced the launch of a large-format version of its Endoform Natural product. The largest of Aroa’s wound care products, this 8.5 by 6-inch device has been launched in the US and Canadian markets, following its...
Paul Hayes (Cambridge, UK) talks to BLearning Wounds at LINC 2020 (Leipzig Interventional Course; 28–31 January 2020, Leipzig, Germany) about the challenges of wound care, in particular assessing the success or failure of endovascular treatment of peripheral artery disease,...
The first head-to-head randomised controlled trial to directly compare two different drug-coated balloons—the lower-dose density (2μg/mm2) Ranger and higher-dose density (3.5μg/mm2) IN.PACT—showed similar results with regard to effectiveness and safety through 12 months for femoropopliteal interventions, Dierk Scheinert (Leipzig, Germany)...
Kenneth Ouriel (New York, USA) talks to BLearning Peripheral at LINC 2020 (Leipzig Interventional Course; 28–31 January 2020, Leipzig, Germany) about a recent independent analysis to assess safety outcomes of femoropopliteal drug-coated balloon (DCB) angioplasty using patient-level data from the Lutonix clinical programme–and...
Medtronic has announced that it will begin enrolment in a pilot study evaluating the safety and efficacy of the Symplicity Spyral renal denervation (RDN) system using a targeted procedural approach with a reduced number of radio frequency (RF) ablations....
At the Leipzig Interventional Course 2020 (LINC; 28–31 January, Leipzig, Germany), Alvimedica held a Lunch Symopsium in which they introduced a new in-stent treatment of superficial femoral artery (SFA) lesions—Nitides—a technology based on the sustained release of Sirolimus by...
According to the findings of a European multicentre retrospective analysis, the use of drug-coated balloons (DCBs) for the treatment of symptomatic central venous stenosis in dialysis patients has been demonstrated as safe. Presented at the Leipzig Interventional Course 2020...
Laminate Medical Technologies has announced the completion of enrolment into the VasQ external support US pivotal study. The study was conducted at 17 sites across the USA and prospectively enrolled 144 male and female patients in need of an...
Recently appointed chair of the Circulation Foundation, Rachel Bell (London, UK), talks to BLearning at VSASM 2019 (Vascular Societies’ Annual Scientific Meeting; 27–29 November, Manchester, UK) about the role of the vascular charity and her plans for its future.
Bell, who spoke on both aortic...
Enrico Ascher (New York, USA) interviews Juan Parodi (Buenos Aires, Argentina) for VEITHtv at VEITHsymposium 2019 (19–23 November, New York, USA), about his research on claudication therapy.
Parodi explains how he was able to avoid amputating a patient’s extremity by making the patient drink...
Sahil Parikh (New York, USA) talks to Brian DeRubertis (Los Angeles, USA) at VEITHsymposium 2019 (19–23 November, New York, USA) about the future of below-the-knee (BTK) interventions, with the latter noting that there is “a lot of excitement” around the...
Nick Cheshire (London, UK) interviews Hazim Safi (Houston, USA) for VEITHtv at VEITH 2019 (19–23 November, New York, USA) about his early training, which Safi describes as being “like the wild west”.
He argues arduous training prevents trainees from learning and believes it is...
Biolitec has added another laser fibre to its ELVeS Radial 2ring laser fibre family: the ELVeS Radial 2ring Pro enables the minimally invasive endovenous laser treatment of severely tortuous veins.
According to a press release, the ELVeS Radial 2ring Pro...
In this issue:
“Do not be an island”: Teamwork prescribed to tackle major demographic trends in vascular disease (p. 1)
Hotly-contested meta-analysis suggests a higher risk of death or amputation at one year when paclitaxel-coated balloons are used in...
In this issue:
"Do not be an island": Teamwork prescribed to tackle major demographic trends in vascular disease (p. 1)
Hotly-contested meta-analysis suggests a higher risk of death or amputation at one year when paclitaxel-coated balloons are used in...
Profusa has announced research findings that suggest the company's Lumee oxygen platform may help improve the clinical management of patients with critical limb ischaemia (CLI) who are undergoing endovascular revascularisation treatment (EVT). The data, from a recent post-market clinical...
Merit Medical Systems has announced that both its EmboCube (syringe-loaded embolisation gelatin foam) and Torpedo (uniform, preshaped gelatin foam loaded into a cartridge with optional blunt stylet) devices are now US Food and Drug Administration (FDA)-indicated for the embolisation...
MedAlliance has announced the award of its first CE mark approval: for SELUTION SLR, a novel sirolimus drug-eluting balloon (DEB), for the treatment of peripheral arterial disease. This approval applies to sizes from 2x20mm to 7x150mm.
This award was supported...
At this year’s Controversies and Updates in Vascular Surgery annual meeting (CACVS; 23–25 January, Paris, France), Dittmar Böckler (University Hospital Heidelberg, Heidelberg, Germany) made a case for the centralisation of aortic surgery, detailing 10 reasons why this is “imperative”...
New arteriovenous (AV) access results were presented at the 2020 Leipzig Interventional Course (LINC; 28–31 January, Leipzig, Germany), including data on percutaneous AV fistula creation and also the use of drug-coated balloons (DCBs) in AV access interventions. While Robert...
Intact Vascular has announced US Food and Drug Administration (FDA) pre-market approval (PMA) for the expansion of its Tack Endovascular System (6F) portfolio. The new approved device size is indicated for repair of post-angioplasty dissections in superficial femoral and...
Profusa has announced research findings that suggest the company's Lumee oxygen platform may help improve the clinical management of patients with critical limb ischemia (CLI) who are undergoing endovascular revascularisation treatment (EVT). The data, from a recent post-market clinical...
A study that aimed to evaluate the sensitivity and specificity of subepidermal moisture (SEM)—a biomarker employed for the early detection of pressure ulcers—has demonstrated that SEM biocapacitance measures “can complement clinical skin and tissue assessments (STAs)”. Published in Wound...
BandGrip has announced a partnership with Rush University Medical Center, Chicago, USA, to improve patient care and efficiencies in the operating room. According to a statement, Rush has recognised the benefits of the product and knows it would be...
NOTE: This video is ONLY available to watch in selected countries and geographies
The one-year IN.PACT AV trial results, first presented at LINC 2020 (28–31 January, Leipzig, Germany), reveal that the target lesion primary patency in the patient group treated with the...
At the 2020 Leipzig Interventional Course (28–31 January, Leipzig, Germany), Sabine Steiner (University of Leipzig, Leipzig, Germany) presented one-year results of the COMPARE-RTC of low-dose versus high-dose paclitaxel balloons. Both types of balloons exhibited comparable results with excellent effectiveness...
Today at the 2020 Leipzig Interventional Course (LINC; 28–31 January, Leipzig, Germany), BD announced that interim findings from the Lutonix below-the-knee (BTK) investigational device exemption (IDE) trial show positive safety results at three years for freedom from mortality,...
Profusa has announced that it has received Conformité Européenne (CE) mark approval to market its Wireless Lumee Oxygen platform for continuous, real-time monitoring of tissue oxygen in patients with potential acute and chronic changes in tissue oxygen levels. The...
Sky Medical Technology has announced that the geko device was presented as part of the didactic program, and at a corporate-sponsored symposium, at the Orthopaedic Summit 2019: Evolving Techniques (OSET 2019) annual meeting (December 11–14, Las Vegas, USA).
Jason Snibbe, a...
Diabetic foot ulcers frequently occur in different locations to previously-healed wounds for patients in diabetic foot remission, according to a secondary analysis of trial data. “Given the significant morbidity, mortality, and resource utilisation associated with foot ulcer recidivism, quality...
Philips today announced four-year results from the randomised controlled ILLUMENATE pivotal trial in the USA. The data show similar mortality rates through four years for patients treated with Stellarex compared to those treated with percutaneous transluminal angioplasty (PTA). The...
First revealed at the International Symposium on the Diabetic Foot (22–25 June 2019, The Hague, The Netherlands), new guidelines from the International Working Group on the Diabetic Foot (IWGDF) have updated recommendations for the practice of diabetic foot care...
The Society for Vascular Surgery (SVS) and Society of Thoracic Surgeons (STS) are releasing new Reporting Standards to ensure patients with Type B aortic dissections (TBAD) receive appropriate treatment and care.
The care of patients with Type B dissections has...
Acceleron Pharma today announced that the PULSAR Phase 2 trial of sotatercept met its primary and key secondary endpoints in patients with pulmonary arterial hypertension (PAH).
In patients on stable background PAH-specific therapies, sotatercept demonstrated a statistically significant reduction in...
Repair-related reinterventions and readmissions are lower after retroperitoneal compared with transabdominal open abdominal aortic aneurysm (AAA) repair, according to a large multicentre retrospective review published in the January edition of the Journal of Vascular Surgery.
For those patients undergoing open...
In this supplement, sponsored by Philips:
Hans-Henning Eckstein and Heiko Wendorff discuss their experience with various elements of the new hybrid OR system
“Integral in building my practice”: Adam Beck on VesselNavigator in complex vasculature
“The best of both worlds”: Ramon Varcoe on embracing the hybrid OR
Joss...
Ra Medical Systems has announced approval from the US Food and Drug Administration (FDA) that the company has provided sufficient data to support initiating an investigational device exemption (IDE) to evaluate the safety and effectiveness of the DABRA excimer...
MTF Biologics, a global nonprofit organisation, has announced a partnership with Net Health, a provider of cloud-based software for specialised care. According to a statement, the collaboration allows MTF Biologics to offer its wound care solutions through Net Health’s...
This article is an advertorial sponsored by Lombard Medical.
One of the novel applications of the Altura Endograft System is use in the revision of previously placed endovascular grafts or previous abdominal aortic surgery. Jörg Tessarek (Bonifatius Hospital Lingen, Lingen,...
Sean Lyden (Cleveland, USA) talks to VEITHtv at the VEITHsymposium 2019 (19–23 November, New York, USA) about the “tumultuous” year for the industry following the publication of the 2018 Katsanos meta-analysis which showed an increased mortality risk with the use of paclitaxel-coated balloons at five-years in...
Juan Granada (New York, USA) chats to Sahil Parikh (New York, USA) for VEITHtv about below-the-knee (BTK) intervention and innovation and how the paclitaxel controversy has influenced the treatment of BTK disease.
Parikh says: “Frankly there is a lot of dampened enthusiasm”...
“When it comes to complicated wounds, it is important to acknowledge that we cannot do it alone,” argued David Davidson (Buffalo Medical Group, Buffalo, USA), before emphasising that “we need a truly multidisciplinary limb preservation team” during his presentation...
Keith Calligaro (Philadelphia, USA) interviews Vikram Kashyap (Cleveland, USA) for VEITHtv at VEITHsymposium 2019 (19–23 November, New York, USA) about whether transcarotid artery revascularisation (TCAR) might replace carotid endarterectomy (CEA) in the future. Kashyap says that although he has become an “enthusiast” of...
Ali AbuRahma (Charleston, USA) moderates a panel for VEITHtv where he is joined by Mahmoud Malas (San Diego, USA) and Enrico Ascher (New York, USA), to discuss the differing strengths and downsides of transcarotid artery revascularisation (TCAR) and carotid endarterectomy (CEA).
Malas...
3M has announced that the UK National Institute for Health and Care Excellence (NICE) has published a Medtech Innovation Briefing (MIB) regarding the use of VAC VERAFLO Therapy to promote wound healing in chronic and acutely infected wounds.
The...
George Adams (Raleigh, USA) talks to VEITHtv at the VEITHsymposium 2019 (19–23 November, New York, USA) about the benefits of using easy-to-use Shockwave IVL (Intravascular Lithotripsy) in calcified below-the-knee vessels, which he says, results in “great outcomes”.
Adams explains that calcific plaque, particularly medial...
AngioDynamics has announced the launch of the PATHFINDER I: Post-Market Registry (PATHFINDER I-Registry, EX-PAD-05), a pilot study to evaluate the safety and efficacy of the company’s AURYON atherectomy system in the treatment of de novo, re-stenotic, and in-stent restenosis...
Smith+Nephew has announced the publication of results from a new randomised controlled trial (RCT) demonstrating that the use of the PICO single-use negative pressure wound therapy system (sNPWT) significantly reduces wound area, depth and volume, compared with traditional negative...
Enrico Ascher (New York, USA) interviews Mark Farber (Chapel Hill, USA) for VEITHtv at VEITHsymposium 2019 (19–23 November, New York, USA) about the differences between off-the-shelf (OTS) devices and custom manufactured devices (CMD) for complex aneurysm repair.
Farber says physicians are faced with a...
Healthy.io has announced the launch of a digital wound management solution, which is an extension of Healthy.io's clinical grade colour recognition products. According to a statement, this solution will help healthcare professionals objectively assess chronic wounds and track their...
A new meta-analysis, just published in the Journal of Vascular and Interventional Radiology (JVIR), suggests significantly worse amputation-free survival at one year with the use of paclitaxel-coated balloons in arteries below the knee in the treatment of chronic limb-threatening...
Eminent vascular surgeons and interventional cardiologists including Kim Hodgson (Springfield, USA), Ramon Varcoe (Sydney, Australia) and Gary Ansel (Columbus, USA) give their thoughts on the recently released meta-analysis in JVIR, which has suggested an increased risk of death or...
A range of interventional radiology experts including Michael Dake (Tucson, USA), John Kaufman (Portland, USA), Jim Reekers (Amsterdam, The Netherlands) and Gunnar Tepe (Rosenheim, Germany) give their thoughts on the recently released meta-analysis in JVIR, which has suggested an...
MedAlliance, the first drug-eluting balloon (DEB) company in the world to receive US Food and Drug Administration (FDA) breakthrough device designation status for a sirolimus DEB, has now been awarded breakthrough status for SELUTION SLR, its sustained limus release...
In the Aortic session at this year’s Controversies and Updates in Vascular Surgery meeting (CACVS; 23–25 January, Paris, France), Stéphan Haulon and Dominique Fabre will discuss the rise of artificial intelligence (AI), informing delegates how this technology could be...
Wound Care Advantage has announced the release of FlightPlan 2.0, an algorithm-based referral programme which is designed to provide flexibility and increase efficiency for both physicians and programme directors working in the field of wound care and healing.
According to...
There is no sign of increased all-cause mortality following the use of paclitaxel-coated devices for the treatment of symptomatic peripheral arterial occlusive disease, a new study based on German claims data attests in the European Journal of Vascular and...
The US Food and Drug Administration (FDA) has released draft guidance detailing the information device-makers should include in 510(k) submissions of catheter-based devices intended to treat peripheral vasculature lesions.
The guidelines, which cover class II peripheral percutaneous transluminal angioplasty (PTA)...
The application of new global vascular guidelines for chronic limb-threatening ischaemia (CLTI) provides an opportune and worthwhile window to revisit the concept of critical limb ischaemia (CLI), along with its implications as a worldwide healthcare issue. In this article,...
Watch Andrew Holden (Auckland, New Zealand) present the six-month results of the IN.PACT AV Access trial. The multicentre, prospective, randomised controlled trial shows that treatment with the IN.PACT AV drug-coated balloon (DCB) resulted in 56% fewer target lesion revascularisation procedures...
Alucent Biomedical has received US Food and Drug Administration (FDA) approval to proceed with a phase 1 clinical trial to evaluate the safety and efficacy of its Natural Vascular Scaffolding (NVS) technology. The therapy is designed to treat peripheral...
Penumbra has announced US Food and Drug Administration (FDA) 510(k) clearance for expanded indication of the Indigo Aspiration System. As part of the system, Indigo Aspiration Catheters and Separators are indicated for the removal of fresh, soft emboli and...
MedAlliance has announced completion of patient enrolment in the PRESTIGE below-the-knee (BTK) clinical trial. The objective of this clinical investigation is to evaluate the six-month safety and performance outcomes of SELUTION SLR, MedAlliance’s novel sirolimus-eluting balloon, for the treatment...
Reflow Medical announces that the Temporary Spur stent system, a novel retrievable stent technology intended for the treatment of below-the-knee (BTK) peripheral arterial disease, has been designated for the breakthrough devices programme by the US Food and Drug Administration...
With the level of spending on wound care in the billions of pounds, both the financial and health-related impact of venous leg ulcers cannot be understated. Dan Carradice (Hull York Medical School, Hull, UK) speaks to iWounds News about the...
Researchers at Lund University, Lund, Sweden, have developed a new hydrogel based on the body’s natural peptide defence, which has been shown to prevent and treat infections in wounds. The formulation kills multi-resistant bacteria, something that is “increasing in...
Ronald Dalman (Stanford, USA) and Kak Khee Yeung (Amsterdam, The Netherlands) talk to VEITHtv at the VEITHsymposium 2019 (19–23 November, New York, USA) about how the use of metformin may help suppress the progression of abdominal aortic aneurysms (AAA).
Dalman points out that initial...
Matt Thompson (Irvine, USA) talks to VEITHtv about his role, as chief medical officer at Endologix, in assessing the diverse portfolio of endografts and their performance.
Thompson notes that because different endograft designs have different strengths and complications, “it is important to...
In the area of lower limb amputation, core outcome sets “should be adopted on all future studies recruiting patients” said Graeme Ambler (University of Bristol, Bristol, UK), who was speaking at the annual meeting of the Vascular Societies' Annual...
Frank Arko (Charlotte, USA) talks to VEITHtv at the VEITHsymposium 2019 (19–23 November, New York, USA), about the benefits of using Shockwave Intravascular Lithotripsy (IVL) when performing EVAR and TEVAR through calcified iliac arteries.
Arko, who shares some of the downsides of traditional procedures...
Serena Auñón-Chancellor, clinical associate professor of medicine at Louisiana State University Health, New Orleans School of Medicine’s branch campus, in Baton Rouge, USA, is the lead author of a paper which describes a previously unrecognised risk of spaceflight discovered...
Christopher Kwolek (Boston, USA) talks to VEITHtv about the “truly revolutionary” LEOPARD trial which evaluated the AFX EVAR AAA Graft System (Endologix) with anatomical fixation against a range of other approved endovascular systems with proximal fixation. The three-year data show that...
The use of a new tissue allograft, which can be injected into areas of high pressure in the foot, has demonstrated clinical improvement in a number of real-life cases, according to a presentation delivered at the Desert Foot Conference...
“When it comes to complicated wounds, it is important to acknowledge that we cannot do it alone,” argued David Davidson (Buffalo Medical Group, Buffalo, USA), before emphasising that “we need a truly multidisciplinary limb preservation team” during his presentation...
Following their publication in the American Diabetes Association (ADA) journal Diabetes Care, results of a double-blinded, randomised controlled trial have been announced, showing that at both 12 weeks and 12 months, adjunctive cyclical pressurised topical oxygen wound therapy (TWO2)...
Bluegrass Vascular Technologies has announced the publication of positive results associated with a clinical study involving the use of the Surfacer Inside-Out access catheter system in the peer-reviewed American Journal of Kidney Diseases.
The publication highlights the first-ever independent, multicentre...
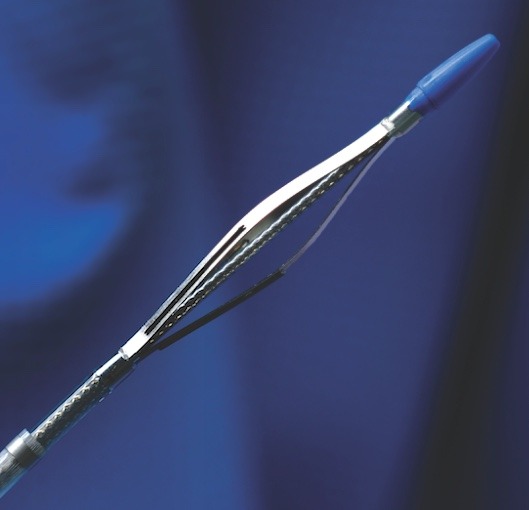
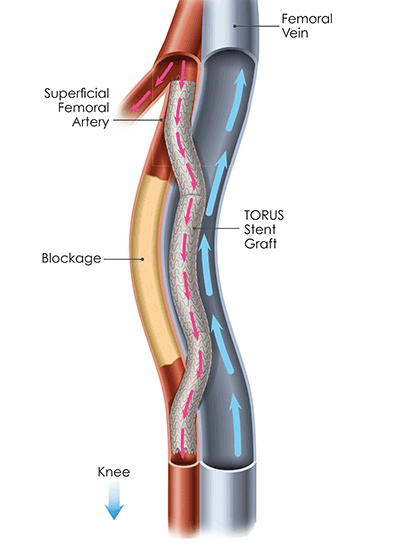
PQ Bypass announced today that it has received approval for coverage from the Centers for Medicare and Medicaid Services (CMS) for the Investigational Device Exemption (IDE) study titled TORUS 2. This approval will affect coverage across all Medicare administrative...
“The goal needs to remain on improving healing, but also reduced recurrence in the long term,” said Stephen Black (Guy’s and St Thomas’ Hospital, London, UK), during his presentation of results from the Leg Ulcer Pathway Audit (LUPA) study...
In this issue:
“Beware vulture capitalists”: Renowned inventor Thomas Fogarty outlines keys to success as medical device innovator and entrepreneur (p. 1)
New patient-level data “likely inadequate” to determine paclitaxel mortality signal (p. 1)
Celia Riga gives an update...
In this issue:
“Beware vulture capitalists”: Renowned inventor Thomas Fogarty outlines keys to success as medical device innovator and entrepreneur (p. 1)
New patient-level data “likely inadequate” to determine paclitaxel mortality signal (p. 1)
Celia Riga gives an update...
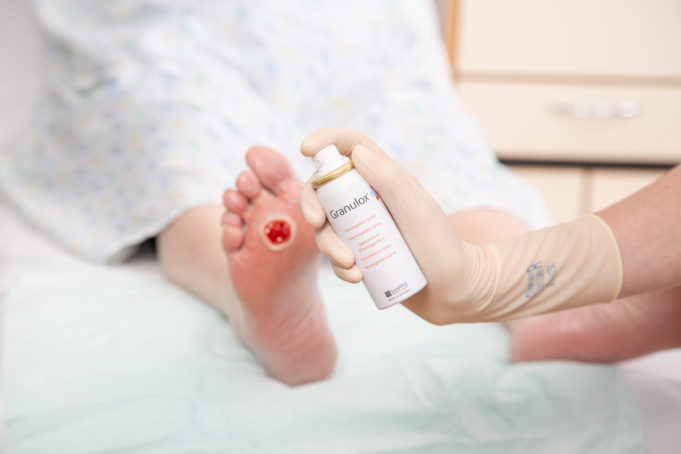
A controlled evaluation of 100 UK National Health Service (NHS) patients has demonstrated savings in chronic wound care costs with the use of Granulox, a haemoglobin spray that binds oxygen from the atmosphere and facilitates its diffusion to the...
Enrico Ascher (New York, USA) interviews endovascular pioneers Juan Parodi (Buenos Aires, Argentina) and Frank Veith (New York, USA) about the early days of endovascular aortic repair (EVAR) for VEITHtv. While Parodi recounts his early quest for a less invasive, less morbid,...
Jan Blankensteijn (Amsterdam, Netherlands) talks to BLearning Aortic at VSASM 2019 (Vascular Societies’ Annual Scientific Meeting; 27–29 November, Manchester, UK) following his Kinmonth Lecture presentation titled ‘The current status of EVAR—Two decades of daydreams and nightmares’.
Blankensteijn talks about the highs and lows associated...
Nick Cheshire (London, UK) interviews Andrew Bradbury (Birmingham, UK) for VEITHtv at VEITH 2019 (19–23 November, New York, USA) about the draft National Institute for Health and Care Excellence (NICE) guideline on abdominal aortic aneurysm (AAA) repair. This guideline has been in the centre...
The US Preventive Services Task Force (USPSTF) has released a new recommendation statement on screening for abdominal aortic aneurysms (AAA) in asymptomatic adults 50 years or older. The recommendation, published in the Journal of the American Medical Association (JAMA),...
Cardiovascular Systems announced today that the first patient in Europe has been treated with its Stealth 360 peripheral orbital atherectomy system (OAS) 1.25mm Micro Crown with GlideAssist (1.25 Micro Crown).
According to a press release, the 1.25 Micro Crown is...
Dominic Foy (Bournemouth, UK), President of the SVT (Society for Vascular Technology of Great Britain & Ireland), talks to BLearning at VSASM 2019 (Vascular Societies’ Annual Scientific Meeting; 27–29 November, Manchester, UK) about the role of the SVT and its aims to promote...
Laminate Medical Technologies (Laminate) has announced the completion enrolment of the VALUE study for the VasQ External Support. The postmarket study enrolled 80 patients (50 upper arm and 30 forearm fistulas) for sites across Germany, France, Spain, and the...
Described as “deliberately challenging”, the new time-to-treatment targets published by the Vascular Society of Great Britain and Ireland (VSGBI), as part of the Peripheral Arterial Disease Quality Improvement Framework, have prompted a number of different measures in the UK...
LimFlow, a specialist in minimally-invasive technology for the treatment of chronic limb-threatening ischemia (CLTI), a severe form of peripheral artery disease (PAD), has announced that the first patient has been treated in its PROMISE II US pivotal trial of...
A real-world study investigating the luminor drug-coated balloon (DCB; iVascular) in Brazil has started, following approval for commercialisation of the device by ANVISA, the Brazilian regulatory authority. LUMBRA (LUMinor balloon in a BRAzilian registry for real-world lesions) aims to...
Narayan Karunanithy outlines the benefits of intravascular ultrasound (IVUS) during haemodialysis access intervention.
Thoracic central veins include intra-thoracic segments of the internal jugular, subclavian, brachiocephalic veins and superior vena cava. These veins are located central to the superior thoracic aperture...
Michael Jenkins talks to Vascular News about the Circulation Foundation, outlining the history of the charity and giving an update on recent activities.
The history of the Circulation Foundation (CF) dates back to the original British Vascular Foundation, the...
A new initiative supported by both the Association for the Advancement of Wound Care (AAWC) and the Wound Healing Society (WHS)—the “Wound Care Expert/FDA-Clinical Endpoint Project” (WEF-CEP)—will take an updated list of primary endpoints for wound care trials to...
Joost van Herwaarden (Utrecht, The Netherlands) and Tilo Kölbel (Hamburg, Germany) discuss the current challenges of image-guided therapy—primarily regarding the limiting factor of having 2D imaging of a 3D environment, including of the intra-body devices and about the concerns...
Inpatient wound services are an essential component of care that can improve the likelihood of patients returning as outpatients, thereby reducing readmissions, Christi Cavaliere (Cleveland Clinic, Cleveland, USA) told the annual meeting of the American College of Wound Healing...
There is no significant benefit to the use of nanocrystalline-coated silver dressings over standard wound dressings, following coronary revascularisation, in patients with type 2 diabetes, according to the results of a study conducted with 194 patients. Results of the...
Internal BIBA MedTech data show that perceptions on paclitaxel devices have shifted considerably in the twelve months that have elapsed since Konstantinos Katsanos (Patras, Greece) et al published their meta-analysis in the Journal of the American Heart Association (JAHA)...
A new study reports high levels of patient satisfaction with a minimally invasive approach to creating vascular access for kidney patients who require haemodialysis. In addition, the researchers found that nearly all the dialysis access sites, or fistulas, were...
Using an implant containing gentamicin in the groin area following vascular surgery does not significantly reduce the incidence of infected wounds during the postoperative course, according to a new study presented at the European Society for Vascular Surgery annual...
MolecuLight, a specialist in handheld fluorescence imaging for real-time visualisation of fluorescence in wounds, has received FDA 510(k) clearance for its i:X handheld fluorescence imaging device for use in the detection of wounds containing bacteria. This FDA 510(k) clearance...
Orpyx has announced the launch of its Orpyx SI Sensory Insoles with remote patient monitoring for the prevention of diabetic foot ulcers (DFU) and neuropathy-related ulcers. By monitoring pressure, temperature, and movement, the Orpyx SI Sensory Insole system provides the wearer with...
In the UK, the lower limb amputation rate has not improved significantly since 2012, and is contributed to heavily by the high incidence of infection and ischaemia in patients with diabetic wounds. Michael Edmonds (King’s College Hospital, London, UK),...
The first UK clinical trial site for the treatment of diabetic patients with chronic limb-threatening ischemia (CLI) using a novel patient-specific regenerative therapy has opened for patient recruitment at the University Hospital of Wales in Cardiff, UK. The site...
CryoLife recently announced that it has received CE mark for the E-nside thoracoabdominal aortic aneurysm (TAAA) multibranch stent graft system for the endovascular treatment of TAAAs.
Approximately 20,000 patients with thoracoabdominal disease are treated annually worldwide, which places the overall...
With the rising importance of high-quality research in the field of wound care and healing, there is a need for key players from all disciplines across the wound care spectrum to collaborate. Writing for iWounds News, editorial board member...
Wound care specialists can improve microcirculation by maximising “the overall vascular health of patients” It is the refrain William Ennis (University of Illinois, Chicago, USA) hears continually: “I cannot do anything about the microcirculation!” His swift response however, at...
Insera Therapeutics announced recently that the first patient with acute pulmonary embolism has been treated with its flagship cyclical aspiration system, CLEAR. Earlier last month, the first two stroke patients were treated using the same platform. The company received...
CryoLife recently announced it has received CE mark for the E-nya thoracic stent graft system for the minimally invasive repair of lesions of the descending thoracic aorta, including thoracic aortic aneurysms and dissections. The E-nya thoracic stent graft will...
Paul Chrisp, the director of the Centre for Guidelines at the National Institute for Health and Care Excellence (NICE), spoke at the annual meeting of the Vascular Society for Great Britain and Ireland (VSGBI; 27–29 November) about what the...
New guidance on monitoring patients after revascularisation for peripheral arterial diseases (PAD) has been produced by the European Society of Cardiology (ESC) Working Group on Aorta and Peripheral Vascular Diseases and the European Society for Vascular Surgery (ESVS) to...
Endovascular arteriovenous fistulas (endoAVF) platforms add additional fistula options for patients and physicians, argues Dheeraj Rajan (University Health Network, University of Toronto, Toronto, Canada, and a paid consultant for BD) at the 2019 Vascular and InterVentional Advances meeting (VIVA;...
Merit Medical Systems recently announced that it has been granted Breakthrough Device Designation by the US Food and Drug Administration (FDA) for the Merit WRAPSODY endovascular stent graft system. The WRAPSODY system is a flexible, self-expanding endoprosthesis for which...
Silk Road Medical announced the presentation of real-world data for the treatment of patients with carotid artery disease at risk for stroke at the VEITHsymposium in New York, USA (19–23 November). In a headline presentation, Mahmoud Malas of the...
Medtronic has announced one-year outcomes for the Valiant Navion thoracic stent graft system for thoracic endovascular repair (TEVAR) and four-year clinical outcomes from the ANCHOR registry evaluating the Heli-FX EndoAnchor system in patients with hostile neck abdominal aortic aneurysms (AAA)....
Implementation of an evidence-based pathway for leg ulcer care in community nursing has resulted in a significant reduction in healing times, according to research conducted by the Manchester University NHS Foundation Trust, Manchester, UK. Although quality of life scores...
Medtronic today announced US Food and Drug Administration (FDA) approval of the IN.PACT AV drug-coated balloon (DCB), a paclitaxel-coated balloon indicated for the treatment of failing arteriovenous (AV) access in patients with end-stage renal disease (ESRD) undergoing dialysis.
AV fistulae...
Initial findings of an early feasibility trial have demonstrated that the LimFlow System does represent a safe and reproducible method of treating patients with no-option critical limb ischaemia (CLI). The results of PROMISE I, which were described by chief...
At this year’s VEITHsymposium (19–23 November, New York, USA), Marc Bosiers presented data that show that patient treatment with the Zilver PTX stent has several benefits when compared to traditional bypass surgery. The data, which were gathered from a...
Terumo Corporation today announced that it has decided to acquire Aortica Corporation, a US-based company dedicated to advancing the science of personalised vascular therapy.
Aortica has developed an automated case planning software known as AortaFit, designed to precisely match fenestrations...
Aiming for full revascularisation “may not be the best solution” for treating patients with critical limb ischaemia (CLI) according to Thomas Zeller (Bad Krozingen, Germany), who advises that it is favourable to pursue a strategy offering the “best chance...
Terumo Aortic has announced the launch of the company’s global endovascular registry known as TiGER (Terumo Aortic Global Endovascular Registry).
TiGER is a global, prospective, multi-arm, multicentre registry evaluating more than 1,000 patients over a period of five years. The...
Clinically relevant outcomes are needed to measure catheter-directed therapy (CDT) against anticoagulation alone to treat submassive pulmonary embolism (PE) patients. Further, CDT’s ability to affect short-term and long-term outcomes such as cardiopulmonary health after PE needs “better characterisation”, Akhilesh...
As part of newly published international guidelines on the prevention and treatment of pressure ulcers, it has been revealed that these wounds occur, primarily, due to the deformation and distortion of cells and tissues. Previously, it was understood that...
The 2019 Prevention and Treatment of Pressure Ulcers/Injuries: Clinical Practice Guideline, published on 15 November 2019, has introduced new evidence-based recommendations for using a "subepidermal moisture (SEM)/oedema measurement device as an adjunct to routine clinical assessment."
"The days of subjective...
At the 2019 Vascular Interventional Advances (VIVA) conference in Las Vegas, USA (4–7 November), Misti Malone (US Food and Drug Administration Office for Cardiovascular Devices, Silver Spring, USA), gave an update on the regulatory landscape, outlining various recent...
Daniel Clair (Columbia, SC, USA), presented positive six-month data from the full patient cohort of the PROMISE I early feasibility study of the LimFlow percutaneous deep vein arterialisation system (LimFlow) as a late-breaking trial at VIVA 2019 (Vascular InterVentional...
Curiously, the mortality risk for patients with critical limb ischaemia (CLI) has not changed over the last few decades, despite advances in cardiovascular medicine that treat myocardial infarction and stroke, the two biggest causes of death in these patients,...
Ian Loftus talks to Vascular News about various aspects of his professional life, from a surgical placement that inspired his career path through to professorship at St George’s Hospital in London, UK. The current president of the Vascular Society...
“Our aim is to establish the underlying economic and clinical case for change, identify what needs to happen and describe how we make that change happen” argued Una Adderley, director of the National Wound Care Strategy Programme, who was...
“Caring for vascular patients is going to take a team,” concluded Marc Bonaca (Boston, USA) at the 2019 Vascular Interventional Advances (VIVA) conference in Las Vegas, USA (4–7 October). Bonaca opened the conference with a presentation on the major...
US wound care service providers operating in skilled nursing facilities were urged to follow a new set of steps when attending to patients amid a new billing system adopted by the Centers for Medicare and Medicaid Services (CMS), according...
UK-based Sky Medical Technology has received US Food and Drug Administration (FDA) 510(k) clearance for its "geko" device for stimulation of the calf muscles to prevent venous thrombosis in non-surgical patients at risk for venous thromboembolism (VTE).
This adds...
Bruce Gray (Greenville, USA) talks to Vascular News after winning the LIVE (Leader In EndoVascular Education) award at VIVA 2019 (4–7 November, Las Vegas, USA), which he says was “very humbling” and “an honour”.
Gray speaks to the challenge of...
Nick Hopkins (Buffalo, USA) talks to BLearning Neuro at VEITH Symposium 2018 (13–17 November 2018, New York, USA) about the DAWN trial (LINK), which showed that in carefully selected acute ischaemic stroke patients that “intervention out to 24 hours is viable”.
“Stroke...
The National Pressure Ulcer Advisory Panel (NPUAP) has announced that it has changed its name to the National Pressure Injury Advisory Panel (NPIAP). According to a statement, the new name reflects the future direction of the organisation and reaffirms...
The first patient has been enrolled in the TORUS 2 multicentre, clinical trial, evaluating the use of the self-expanding Torus stent graft system (PQ Bypass), a novel technology designed for the treatment of peripheral arterial disease (PAD) in the...
At this year’s Vascular Interventional Advances (VIVA) conference (4–7 November, Las Vegas, USA), Michael D Dake presented data on Zilver PTX that supports the device’s benefits across different patient groups.
Dake presented the latest data regarding Zilver PTX, Cook Medical’s...
Abdominal aortic aneurysm (AAA) diameter has important implications for operative and long-term outcomes for patients, based on a study of 22,975 patient records in the Vascular Quality Initiative.
Using historical data and more recent large randomised controlled trials, international societies...
William Ennis is currently Catherine and Francis Burzik professor of Wound Healing and Tissue Repair, and chief of Wound Healing Section, University of Illinois, Chicago, USA. With his 25 years of experience practising wound care, Ennis speaks about what...
The efficacy of the Kerecis fish skin treatment for diabetic lower-extremity wounds has been affirmed in a new peer-reviewed article published in the journal Wounds.
The paper, “Acellular Fish Skin Graft Use for Diabetic Lower Extremity Wound Healing: a Retrospective Study...
Penumbra today announced that the EXTRACT-PE trial successfully met the primary endpoints, demonstrating the safety and efficacy of the Indigo Aspiration system for aspiration mechanical thrombectomy in patients with acute pulmonary embolism (PE). The data were presented in a...
Today Medtronic shared a six-month subgroup analysis from the IN.PACT AV Access trial, which evaluated how the IN.PACT AV DCB—an investigational device not currently approved for use in the USA—performed in different target lesion types, arteriovenous fistula (AVF) types,...
Low-frequency, high-intensity ultrasound energy delivery followed by percutaneous transluminal angioplasty (PTA) may improve clinical outcomes in below-the-knee (BTK) lesions without the need for a drug-eluting device, but a larger randomised study is needed to confirm these results. This was...
It was announced today that results of the TANGO low-dose cohort compared to controls are positive, both for primary and secondary endpoints. These latest findings were presented by Ehrin Armstrong at the 2019 Vascular Interventional Advances conference (VIVA) in...
LimFlow SA today announced the presentation of positive six-month data from the full patient cohort in its PROMISE I early feasibility study of the LimFlow percutaneous deep vein arterialisation system. Results were presented on the podium at the 2019...
Ramon Varcoe today announced positive five-year results of the ABSORB BTK trial at the 2019 Vascular Interventional Advances conference (VIVA) in Las Vegas, USA (4–7 November). Primary patency of the Absorb bioresorbable vascular scaffold (BVS) from Abbott achieved a...
It was announced today at the 2019 Vascular Interventional Advances conference (VIVA) (4–7 November, Las Vegas, USA) that, through six months, TOBA II BTK met all endpoints in a 100% dissected vessel population.
This prospective, multicentre, single-arm global pivotal study...
Imbed Biosciences, a biotech company which specialises in the development of advanced therapies for burns, chronic ulcers and surgical wounds, has announced that it has closed its Series A Preferred Stock equity financing, which was expanded to US$4 million...
Today at the 2019 Vascular Interventional Advances conference (VIVA; 4–7 November, Las Vegas, USA), Thomas Zeller (Bad Krozingen, Germany) gave an update on the DEEPER OUS trial. He concluded that the Temporary Spur stent system (Reflow Medical) appears to...
One-year results of the MIMICS-3D study were announced today at the 2019 Vascular Interventional Advances conference (VIVA; 4–7 November, Las Vegas, USA). Freedom from clinically-driven target lesion revascularisation (CD-TLR) was 89%, which aligns with results from the MIMICS RCT...
Findings from the REVEAL study confirm a favourable safety and effectiveness profile through six months for the Revolution peripheral atherectomy system from Rex Medical. The results were presented today at the 2019 Vascular Interventional Advances conference (VIVA) in Las...
Positive two-year results from a prospective multicentre premarket study including 25 patients treated with the Nexus aortic arch stent graft (Endospan) were presented today at the 2019 Vascular Interventional Advances conference (VIVA) in Las Vegas, USA (4–7 November).
Technical success...
Two-year data confirm that common femoral artery (CFA) disease can be treated endovascularly with the Supera stent (Abbott). Results of the VMI-CFA study were presented today at the 2019 Vascular Interventional Advances conference (VIVA) in Las Vegas, USA (4–7...
Boston Scientific today announced positive data for two devices within the peripheral drug-eluting product portfolio during separate late-breaking clinical trial presentations at the 2019 Vascular Interventional Advances conference (VIVA) in Las Vegas, USA (4–7 November).
Data presented included a 12-month...
Medtronic today announced four-year data from the IN.PACT Global study, which reinforce the long-term durability and safety of the IN.PACT Admiral drug-coated balloon (DCB) in patients with peripheral artery disease (PAD) in the superficial femoral (SFA) and popliteal arteries....
The SELUTION SFA trial met its primary endpoint and demonstrated an late lumen loss of 0.19mm. Furthermore, excellent freedom from clinically driven TLR was achieved through 24 months in 87.5%, with no primary TLR event observed after month 11....
CereVasc announced today that Argentina's National Administration of Drugs, Foods and Medical Devices (ANMAT) approved the company's request to conduct a clinical study titled "Endovascular Treatment of Communicating Hydrocephalus with the eShunt System" (ETCHES I). The study will be...
Okami Medical today announced 510(k) clearance by the US Food and Drug Administration (FDA) and initial launch of the LOBO vascular occlusion system.
The company further announced that US patent number 10,376,267, entitled Vascular Occlusion Device and Methods, has been...
Shockwave Medical today announced commercial availability of the Shockwave S4 peripheral intravascular lithotripsy (IVL) catheter in select approved geographies. Shockwave S4 is a low-profile catheter specifically designed to access and treat challenging calcified lesions in below-the-knee (BTK) arteries that...
MolecuLight Inc, a provider of handheld fluorescence imaging for real-time visualisation of bacteria for chronic wounds, has been informed by the American Medical Association (AMA) that in its summary of panel actions September 2019 meeting, the CPT Editorial Panel...
Lars Norgren talks to Vascular News about therapeutic angiogenesis in the treatment of peripheral arterial disease (PAD). He points out that further evaluation in this area is of great importance and that biomarkers may assist in the understanding of...
Medtronic today announced Shonin approval from the Ministry of Health, Labour and Welfare (MHLW) and the launch of the Valiant Navion thoracic stent graft system in Japan for the minimally invasive repair of thoracic descending aortic aneurysms (TAA) and...
In a session focused on new technologies at this year’s London Aorta meeting (10–11 October, London, UK), Celia Riga discussed the rise of robotics, artificial intelligence (AI), and wearables, informing delegates about the “merits and responsibilities” of being either...
The superiority of fish-skin xenografts over dehydrated human amnion/chorion allografts (dHACM) for the treatment of acute wounds has been established in a randomised controlled trial published in Wound Repair Regeneration. Conducted by Robert Kirsner (University of Miami Miller School...
Surmodics has announced that the US Food and Drug Administration (FDA) has designated its Sundance sirolimus-coated balloon catheter as a breakthrough device under the agency’s breakthrough devices programme.
The programme, launched in December 2018, is designed to streamline the market...
The first patient in the United Arab Emirates has been treated with the Stealth 360 peripheral orbital atherectomy system (Cardiovascular Systems), intended for the treatment of patients with peripheral and coronary artery disease.
The first peripheral patient in the Middle...
A systematic review and meta-analysis has found that frailty is an independent risk factor in vascular surgery patients, but there is no evidence to deny intervention in any patient based on frailty score alone. This was the main conclusion...
Intact Vascular has announced the peer-reviewed publication, “Practical applications of Tack implants for infrainguinal dissection repair: A single-centre experience” currently available online, with the article scheduled to be published in the February 2020 issue of Journal of Endovascular...
Masters Speciality Pharma, a UK-headquartered pharmaceutical company "specialising in the global distribution of high-quality, life-changing medicines", has announced the launch of Neuropad, a rapid and reliable screening test for diabetic foot syndrome, in Brazil.
According to a statement, diabetes is...
Endologix have announced a response to the US Food and Drug Administration (FDA) update regarding Type III endoleaks with the AFX Endovascular AAA System.
Matt Thompson, chief medical officer of Endologix, commented: “On September 28, 2017, the FDA issued a...
At the first London Aorta meeting, a collaborative association between the London Aorta Masterclass and the London Aortic Symposium, held 10–11 October in London, UK, Peter Holt (St. George’s Vascular Institute in London, UK) told delegates that “patients will...
The US Food and Drug Administration (FDA) has issued a new safety communication on the risk of Type III endoleaks with use of the Endologix AFX endovascular graft systems for the treatment of abdominal aortic aneurysms (AAA).
The safety communication,...
Improvements that enable better miniaturisation of technology leads to higher-quality wearable health solutions, including digital devices that were “never before possible’, said Breanne Everett (Orpyx Medical Technologies, Calgary, Canada) at the American College of Wound Healing and Tissue Repair...
Philippe Kolh (Liege, Belgium) talks to BLearning Peripheral at ESVS 2019 (European Society for Vascular Surgery; 24–27 September; Hamburg, Germany) about how the European Society for Vascular Surgery’s (ESVS) plans to disseminate the recently published Global Vascular Guidelines for Chronic Limb Threatening...
The GORE® TAG® Conformable Thoracic Stent Graft with ACTIVE CONTROL System was launched in the USA at the 2019 SVS Vascular Annual Meeting (VAM; 12–15 June 2019, National Harbor), just weeks after the device obtained FDA approval.
Dittmar Böckler (Heidelberg,...
Microbion Corporation has announced results from a Phase 1b trial of its microbial bioenergetic inhibitor, pravibismane, in patients with moderate to severe infected diabetic foot ulcers (DFUs). The findings of the trial were presented at the Symposium on Advanced Wound Care...
It has been found that the application of dehydrated human amnion and chorion allografts (dHACA) results in a better venous leg ulcer (VLU) healing rate than the use of standard multilayer compression therapy, following a randomised controlled study comparing...
Christian-Alexander Behrendt (Hamburg, Germany) talks to BLearning Peripherial at ESVS 2019 (European Society of Vascular Surgery; 24-27 September 2019; Hamburg, Germany) about an international study that underlines the stark difference between the number of major amputations in countries like Hungary and Slovakia and others,...
The Solaris (Scitech Medical) is a flexible self-expanding endograft, comprised of a thin multi-direction durable electrospinning polytetrafluoroethylene (PTFE) membrane encapsulating a Nitinol stent structure. The device has been engineered to effectively cover and instantaneously seal off diseased tissue with...
Lorenzo Patrone talks to Vascular News about retrograde access to lower limb arterial chronic total occlusions (CTOs).
What
The retrograde approach to lower limb arterial chronic total occlusions was originally described more than thirty years ago1–3 but is still sometimes...
The US Food and Drug Administration (FDA) has granted Breakthrough Device Designation to Medtronic for its novel Valiant thoracoabdominal aneurysm (TAAA) stent graft. A press release reports that the device is designed to offer an off-the-shelf solution for complex...
Kevin Mani (Uppsala, Sweden) talks to BLearning Aortic at ESVS 2019 (European Society for Vascular Surgery; 24–27 September; Hamburg, Germany) about a recently published epidemiological analysis of ruptured abdominal aortic aneurysms (AAAs) in Sweden over a twenty-year period.
The analysis indicated that the epidemiology of ruptured AAAs is...
There is a growing need for wound care amid higher rates of chronic wounds—somewhere in the vicinity of five to seven million in the United States per year—at a cost level in excess of US$25 billion, Venita Chandra (Stanford...
Concept Medical received CE certification for their MagicTouch group of products on 16 October 2019.
The CE certified products/brands are as listed below:
MagicTouch—PTA (sirolimus-coated percutaneous transluminal angioplasty balloon catheter) for peripheral arterial disease
MagicTouch—ED (sirolimus-coated balloon catheter) for...
A report titled ‘Venous Leg Ulcers: A Silent Crisis’ has been published by the All-Party Parliamentary Group on Vascular and Venous Disease (VVAPPG) at an event held at the Palace of Westminster, highlighting the opportunities available to help thousands...
Scientists of Jena University Hospital, Germany, conducted a meta-analysis sought to evaluate benefit and risk of paclitaxel-coated balloon angioplasty compared to plain old balloon angioplasty as therapy of intermittent claudication. The study confirms an increased all-cause mortality, which has...
Based on the results of the INVIPS trial, incisional negative pressure wound therapy (INPWT) has been shown to decrease the risk of surgical site infections (SSIs) following vascular surgery with inguinal incision. Conducted at Skane University Hospital (Malmӧ, Sweden),...
The National Institute for Health and Care Excellence (NICE) has published the final appraisal determination (FAD) recommending the use of Xarelto (rivaroxaban) by the National Health Service (NHS) in England at a dose of 2.5mg twice daily combined with...
WoundVision, the manufacturer of the industry's first long-wave infrared thermography (LWIT) solution for mitigating risk associated with hospital-acquired pressure injuries (HAPIs) and other wounds, has announced the expansion of its product offering with the debut of the Scout Mobile...
In this issue of iWounds News:
Clinical trials confirm efficacy of TLC-NOSF dressing
IWGDF unveils new infection and PAD guidelines
Michael Edmonds: A new dawn in diabetic foot care
Karen Imma Gellada: Wound care specialisation
Profile: William Ennis
In this issue of iWounds News:
Clinical trials confirm efficacy of TLC-NOSF dressing
IWGDF unveils new infection and PAD guidelines
Michael Edmonds: A new dawn in diabetic foot care
Karen Imma Gellada: Wound care specialisation
Profile: William Ennis
LifeNet Health's Dermacell AWM, an advanced acellular dermal matrix (ADM) for wound treatment, is the first human ADM available in India. According to a statement, this introduction will provide access to proven therapy for millions of patients in India suffering...
Alan Lumsden is chair of the Department of Cardiovascular Surgery and Medical Director at the Houston Methodist DeBakey Heart and Vascular Center in Houston, USA. He spoke to Vascular News about his current research interests and the three rules...
Michael R Jaff is to be vice president, clinical affairs, innovation and technology, peripheral interventions of Boston Scientific by January 2020. In this role, a press release reports, Jaff will lead clinical and medical affairs strategies to support the...
Philips has received US Food and Drug Administration (FDA) approval for two Stellarex 0.035” low-dose (200mm and 150mm) drug-coated balloons for the treatment of de novo and restenotic lesions in native superficial femoral or popliteal arteries.
According to a press...
PolarityTE, a company specialised in regenerative tissue products and biomaterials, has announced findings from an open-label, single-arm pilot study, which examined the impact of SkinTE in closing venous stasis leg ulcers (VLUs) following the failure of conventional treatments.
The clinical outcomes...
PQ Bypass recently announced it has received full approval of its investigational device exemption (IDE) trial of the company’s TORUS stent graft, a novel stent graft platform designed for the treatment of peripheral artery disease (PAD) in the superficial...
It was recently announced that luminor 18 and luminor 35 DCBs (iVascular), are now listed on the French LPPR list for de novo lesions of the femoro-popliteal arteries.
Luminor is a paclitaxel coated balloon specifically designed for dilatation of stenosis...
Advanced fluorescence imaging has the potential to decrease bioburden and significantly improve the healing rates of chronic wounds, according to the results of a case series investigation conducted in Cleveland, USA. As well as targeting debridement to promote wound...
Admedus has sold the distribution rights to its CardioCel and VascuCel product range for $A36.2 million to US-based LeMaitre Vascular. A press release from Admedus announces that it will focus on advancing its transcatheter aortic valve implantation (TAVI) programme...
A pilot evaluation of a new multilayer wound dressing has confirmed its efficacy for transporting exudate containing toxic matrix metalloproteases (MMPs) away from chronic venous leg ulcers (VLUs). Conducted by Oscar M Alvarez (University Hospital; Rutgers New Jersey Medical...
For patients with emergent large vessel occlusion (LVO) eligible for endovascular therapy, prehospital triage to a more distant comprehensive stroke centre (CSC) compared with a closer primary stroke centre (PSC) was associated with significantly shorter time to thrombectomy, better...
Bijan Modarai (London, UK) talks to BLearning Peripheral at ESVS 2019 (European Society for Vascular Surgery; 24–27 September; Hamburg, Germany) about the HT-CLI trial, which will look at the efficacy of a novel blood filtration technology that is aimed at using cells that are...
Turn Therapeutics, a Los Angeles-based pharmaceutical and medical device company, has announced that it has received FDA clearance for its Protego antimicrobial wound dressing, which is described as a disruptive innovation to traditional petroleum saturated gauze dressings.
Protego wound...
3M has announced that it has completed the acquisition of Acelity and its KCI subsidiaries from a consortium comprised of funds advised by Apax Partners (the Apax Funds), together with controlled affiliates of the Canada Pension Plan Investment Board...
VEITHsymposium and BLearning, the digital education division of BIBA Medical, are pleased to announce a collaboration on the creation and dissemination of digital education video content, which will be filmed at the 46th Annual VEITHsymposium in New York, USA...
A real-world safety analysis of paclitaxel-based devices in peripheral arteries, recently published in the European Heart Journal, showed no evidence for increased mortality over a period of 11 years. “In summary,” the authors remark, “our analysis found the use...
Insera Therapeutics has announced that the first two ischaemic stroke patients have been treated with its flagship cyclic aspiration system, CLEAR. The company received CE mark approval for its flagship product, the CLEAR Aspiration System, in March 2019.
“We used...
Medtronic today announced it has received breakthrough device designation from the US Food and Drug Administration (FDA) for its Valiant TAAA stent graft system for minimally invasive repair of thoracoabdominal aortic aneurysm (TAAA).
The FDA breakthrough device programme is intended...
Source: CRF
At the 2019 Transcatheter Cardiovascular Therapeutics (TCT) meeting (25–29 September, San Francisco, USA), InterVene won the TCT-Shark Tank Innovation Competition for its novel endovenous system (BlueLeaf) for forming valves in patients with deep vein reflux and/or chronic venous insufficiency. The award,...
At the 2019 Transcatheter Cardiovascular Therapeutics (TCT) meeting (25–29 September, San Francisco, USA), InterVene won the TCT-Shark Tank Innovation Competition for its novel endovenous system (BlueLeaf) for forming valves in patients with deep vein reflux and/or chronic venous insufficiency. The award, which...
Klox Technologies Inc. (Klox), a regenerative medicine company focused on the development of fluorescent light energy biomodulation systems for applications in dermatology and wound care, has announced that it has completed the acquisition of FB Dermatology, a specialized dermatology...
In this supplement:
Defining the boundaries of the hostile neck
Limitations of custom-made solutions and the added value of off-the-shelf treatment options
Securing the hostile neck: Benefits of ESAR
New in vitro study confirms that the degree of aortic...
Following the 2019 International Vascular Access Symposium (i-VAS 2019; 13 September, Paris, France), current president Alexandros Mallios talks to Vascular News about his highlights of this year’s meeting, what we can look forward to next year, and what makes...
Jos van den Berg (Lugano, Switzerland) tells Vascular News at CIRSE 2019 that the results of the APERTO AVF China trial provide strong evidence of the benefits of using the APERTO drug-coated balloon (DCB; Cardionovum) in haemodialysis patients who had stenosis of, mainly, de novo...
In the largest prospective analysis of cerebrospinal fluid (CSF) in patients undergoing endovascular thoracoabdominal aneurysm repair (TAAA) to date, permanent paraplegia was found to be associated with shedding of bound ADVS-1 from the parenchymal cord into CSF and disruption...
According to a press release, AngioDynamics has acquired Eximo Medical and its proprietary 355nm wavelength laser-technology platform for US$46 million in up-front consideration with up to US$20 million of contingent consideration related to certain technical and revenue milestones. The...
Reoperations for bleeding are cut in half among patients who receive protamine during carotid endarterectomy, a new study has found. That was the main finding revealed by David Stone (Dartmouth-Hitchcock Medical Center, Lebanon, USA) at the 33rd Annual Meeting...
Caitlin Hicks (Baltimore, USA) talks to BLearning at SVS 2019 (Society for Vascular Surgery; 12–15 June; Washington DC, USA) about why it is so important to save limbs and the huge impact limb salvage can have on a patient’s...
Andrew Holden (Auckland, New Zealand) provides perspective on the first results from the Disrupt PAD III observational registry that were presented at the Charing Cross International Symposium 2019. This large registry is designed to evaluate the use of Intravascular...
BD has announced the publication of a company-initiated, independent analysis of the Lutonix 035 drug-coated balloon (DCB) femoropopliteal clinical programme in the Journal of the American College of Cardiology (JACC): Cardiovascular Interventions. These data were simultaneously presented in a...
Lorenzo Patrone (London, UK) shares with BLearning at CIRSE 2019 (7–11 September, Barcelona, Spain) the benefits of using the OUTBACK™ Elite Re-entry Catheter (Cordis, a Cardinal Health Company) in chronic total occlusions—in any arterial segment from the iliacs to...
Imbed Biosciences has announced that it has been awarded a US$1.5 million two-year Small Business Innovation Research (SBIR) grant from the National Institute for Arthritis and Musculoskeletal and Skin Diseases (NIAMS) at the National Institutes of Health (NIH). According...
Intact Vascular has announced the positive one-year results of its Tack Optimised Balloon Angioplasty (TOBA) III clinical trial, successfully achieving both primary and secondary endpoints. Marianne Brodmann, head of the Clinical Division of Angiology, Medical University of Graz, Austria...
Bluegrass Vascular Technologies (Bluegrass Vascular) has announced the publication of positive results associated with the clinical use of the Surfacer Inside-Out access catheter system in the peer-reviewed Journal of Vascular Access (JVA).
Results of an independent, single-centre study demonstrated successful use...
Julien Hasselmann (Malmö, Sweden) talks to iWounds News at ESVS 2019 (European Society of Vascular Surgery; 24–27 September; Hamburg, Germany) about the recent findings of the INVIPS trial which looked at the effect of negative pressure wound therapy (NPWT)...
Terumo Aortic has announced the completion of enrolment of the RelayPro pivotal study in the USA. RelayPro is a low profile, next generation thoracic stent graft device designed to expand the treatment of thoracic endovascular aortic repair (TEVAR) to...
NOTE: ONLY intended for healthcare professionals outside of the USA and France.
Zoom in on a tête-à-tete between Marianne Brodmann (Graz, Austria) and Giovanni Torsello (Munster, Germany) who talk through the latest clinical evidence that directly compares drug-eluting stents in...
Axxess is partnering with Swift Medical to provide thousands of home health agencies access to advanced wound care management.
"Swift Medical's wound management capabilities complement what Axxess provides within our solution and enables us to deliver even more value to...
Huntleigh has announced the UK release of an innovative technology that is designed to assist in the management of hard-to-heal leg ulcers. Developed at the Arjo R&D Faculty in Cardiff, the Wound Express Advanced Wound Therapy Device is designed...
NOTE: ONLY intended for healthcare professionals outside of the USA.
Ulf Teichgräber (Jena, Germany) and Koen Deloose (Dendermonde, Belgium) discuss the 24-month results of the EFFPAC trial and the six-month outcomes of the TINTIN trial. Both trials used the luminor...
EO2 Concepts has announced the publication of a post hoc analysis from its randomised, double-blinded, multi-centre study, published last year in the Journal of Wound Care. This latest study evaluates the positive impact which continuous diffusion of oxygen (CDO), with...
LimFlow has announced that the US Food and Drug Administration (FDA) has approved its investigational device exemption (IDE) for the PROMISE II pivotal study of the LimFlow Percutaneous Deep Vein Arterialisation (pDVA) System.
PROMISE II is a multicentre, prospective, single-arm study...
A recent analysis, with some of the longest follow up to date, suggests that patients undergoing endovascular aneurysm repair (EVAR) may experience a significant long term decrease in renal function. The authors, Edmund R Charles and colleagues at the...
LimFlow has announced that the US Food and Drug Administration (FDA) has approved its investigational device exemption (IDE) for the PROMISE II pivotal study of the LimFlow Percutaneous Deep Vein Arterialisation (pDVA) System.
PROMISE II is a multicentre, prospective, single-arm study...
In this issue:
Global vascular guidelines on chronic limb-threatening ischaemia: Redefining a critical disease (p. 1)
“Unprecedented” development of NICE aortic aneurysm guideline (p. 1)
Nick Inston argues that “more creative solutions” are needed for end-stage vascular access patients...
In this issue:
Global vascular guidelines on chronic limb-threatening ischaemia: Redefining a critical disease (p. 1)
"Unprecedented" development of NICE aortic aneurysm guideline (p. 1)
Nick Inston argues that "more creative solutions" are needed for end-stage vascular access patients...
New data presented at the 2019 American Society of Colon and Rectal Surgeon’s Annual Scientific Meeting by Justin Malek, Baylor Scott & White Health, reveal that the CleanCision System, an advanced irrigating wound protection therapy, was shown to reduce...
A recent study found that being a smoker, having diabetes, and also having a history of coronary heart disease or stroke increases the lifetime risk for PAD by as much as five times that of someone of the same...
The results of AVeNEW, the first level one clinical study dedicated solely to the use of a covered stent designed to treat stenosis in the arteriovenous (AV) fistula access circuit, show that the Covera vascular covered stent (Bard) demonstrates...
Orchestra BioMed, in partnership with Terumo Corporation, today announced that the company has secured breakthrough device designation by the US Food and Drug Administration (FDA) for its Virtue sirolimus-eluting balloon (SEB) in the treatment of below-the-knee (BTK) peripheral arterial...
The US Food and Drug Administration (FDA) has cleared the Artis icono, a family of angiography systems from Siemens Healthineers that permit a range of minimally invasive procedures to be performed in a single interventional suite.
The Artis icono biplane...
According to a press release, Proximo Medical has been announced as the commercial partner for Biotronik's peripheral vascular intervention (PVI) platform in select US markets.
Proximo Medical is a fractional commercial organisation for start-up medical device technologies and established medical device companies looking...
The National Alliance of Wound Care and Ostomy (NAWCO) has announced the first professional credential to recognize clinical expertise in advanced wound care at the 2019 Wild On Wounds (WOW) national conference in Las Vegas, USA.
According to a statement,...
MolecuLight Inc., a provider of handheld real-time fluorescence imaging for chronic wounds, has announced the publication of "Fluorescence imaging guided dressing change frequency during negative pressure wound therapy: a case series": a new paper published today in the Journal...
Two-year results of EffPac—a randomised controlled trial to assess the safety and efficacy of a novel drug-coated balloon (DCB) with a nanotechnology coating—have been announced. The investigators, Ulf Teichgräber (University Hospital Jena, Germany) and colleagues, report that use of...
According to a press release, Cryolife has entered into strategic distribution and credit facility agreements with Endospan. Under the terms of the agreement, CryoLife will have exclusive European distribution rights to Nexus, the first off-the-shelf endovascular stent graft system...
Rex Medical recently announced that it has received 510k clearance from the US Food and Drug Administration (FDA) for the Revolution Peripheral Atherectomy System.
Regulatory clearance was based on data provided by the REVEAL investigational device exemption (IDE) clinical trial....
Cook Regentec has announced enrolment of the first patient in an international clinical trial evaluating the HemaTrate Blood Filtration System to treat patients with critical limb ischemia (CLI) due to peripheral arterial disease (PAD). To treat CLI, the HemaTrate...
Twelve-month results of the BIOFLEX COF trial have demonstrated that the implantation of stents with low chronic outward force (COF) was associated with less neointimal hyperplasia, consequently resulting in less restenosis. Patients treated with Biotronik’s low COF, Pulsar stent,...
Smith+Nephew has announced the US launch of its new PICO 7Y Single Use Negative Pressure Wound Therapy System (sNPWT) with AIRLOCK technology. This is the first PICO sNPWT pump to include an integrated Y connector, enabling the utilisation of...
New data on the use of Zilver PTX, Cook Medical’s paclitaxel-coated stent for peripheral arterial disease (PAD), confirm the safety of the device. These data was presented at the Annual Scientific Meeting of the Cardiovascular and Interventional Radiological Society...
What do Formula 1 cars, space shuttles and airplanes have in common with a ruptured aneurysm? In this article, Colin Bicknell writes about the importance of a well-oiled response strategy for endovascular aneurysm repair (EVAR) of a ruptured aortic...
Statins are linked with reduced mortality in patients with peripheral arterial disease, even when started late after diagnosis, reports a study presented recently at ESC Congress 2019 (European Society of Cardiology; 30 August–4 September, Paris, France) together with the...
PIUR Imaging has launched a portable version of its tomographic ultrasound system to the European market. The new CE-marked PIUR tUS Infinity is a small, wireless version of its tomographic ultrasound solution that turns any standard ultrasound into a...
Vascular Therapies is developing Sirogen, a proprietary sirolimus formulation for intraoperative local, perivascular drug delivery focused on improving vascular access outcomes in patients with kidney disease. Daniel G Clair, vascular surgeon and chairman of the Department of Surgery at...
NOTE: This video is ONLY available to watch in selected countries and geographies
Jos van den Berg (Lugano, Switzerland) speaks to Andrew Holden (Auckland, New Zealand) to distil the headline findings from the first-ever results of the IN.PACT AV access...
The six-month results of TINTIN (Treatment with the lumINor DCB and The IVolutioN self-expanding stent) trial have been revealed at the annual meeting of the Cardiovascular and Interventional Radiological Society of Europe (CIRSE 2019; 7–11 September, Barcelona, Spain) by Koen Deloose.
TINTIN is...
Omar Ishrak, Medtronic’s chairman and CEO is to retire on 26 April 2020, following the end of the company’s current fiscal year. Also, the Medtronic Board of Directors has announced key leadership appointments as part of its multi-year, leadership...
Omar Ishrak, Medtronic’s chairman and CEO is to retire on 26 April 2020, following the end of the company’s current fiscal year. Also, the Medtronic Board of Directors has announced key leadership appointments as part of its multi-year, leadership...
A systematic review and meta-analysis, recently published in the Journal of Endovascular Therapy, found no difference in short- to midterm mortality among patients who underwent dialysis access treatment using a drug-coated balloon compared with percutaneous transluminal angioplasty with an...
Michael Dake (Tucson, Arizona) presented the two-year data from the VERNACULAR trial at CIRSE 2019 on 8 September in Barcelona, Spain.
Dake said: “The 24-month results from the VERNACULAR trial using the BD Venovo stent show absolutely sustained benefit in...
Medtronic has announced the first-ever results from the IN.PACT AV access clinical study comparing the investigational IN.PACT AV drug-coated balloon (DCB) to percutaneous transluminal angioplasty (PTA) in patients with de novo or non-stented restenotic arteriovenous (AV) fistulae lesions. The...
Beacon Tip catheters (Cook Medical) are once again available to physicians in Europe. As of September 2019, the catheters are available in the UK, Ireland, Norway, Sweden, Finland, Denmark, Germany, Poland, the Netherlands, Luxembourg, Belgium, France, Switzerland, Austria, Hungary,...
Concept Medical has been granted breakthrough device designation from the US Food and Drug Administration (FDA) for MagicTouch AVF, its sirolimus drug-coated balloon (DCB) catheter, for the treatment of stenotic lesions of arteriovenous fistulae or arteriovenous graft in haemodialysis...
AstraZeneca has announced detailed results from the positive Phase III THEMIS trial, which showed BRILINTA (ticagrelor) plus aspirin reduced the relative risk for the composite of cardiovascular (CV) death, heart attack, or stroke by 10% (7.7% vs. 8.5%)...
Surmodics has completed enrolment in TRANSCEND, its pivotal clinical trial for the SurVeil drug-coated balloon (DCB).
“TRANSCEND is a rigorous level one, randomised controlled trial that comes at a very important time for the vascular and interventional community,” said Kenneth...
Nicholas (Nick) Inston calls for “more creative solutions” to vascular access challenges, commending interventional radiologists’ innovative reputation as instrumental to increasing the number of treatment approaches available to physicians. Outlining the complexities of end-stage vascular access in an ever-increasing...
According to a recent study, ultrasound surveillance following carotid endarterectomy is “costly and unlikely to improve outcomes or affect management,” even if over 80% ipsilateral restenosis is detected. The investigators suggest that “further research should determine the utility of...
A recent study concludes that the interplay of anatomical and physiological factors in patients with complex aneurysms renders conventional risk stratification tools “unhelpful”, and that a multidisciplinary approach to assessment is mandated. Furthermore, the investigators suggest it is “unusual”...
Ali Azizzadeh (Los Angeles, USA) talks to BLearning at SVS 2019 (Society for Vascular Surgery, 12–15 June, Washington DC, USA) about his recent move from Houston, Texas to Los Angeles, California, and how the patient populations differ between these two areas.
Azizzadeh has observed...
Laminate Medical Technologies (Laminate) has announced that the American Journal of Kidney Disease has published the first multicentre, randomised controlled study of the VasQ External Support for newly created arteriovenous fistulas in haemodialysis patients. Arteriovenous fistulas are the standard...
Michael Edmonds, a pioneer in the treatment of the diabetic foot, has underlined the need for innovation and development in the wound care field. Today, the emergence of multiple randomised controlled trials (RCTs) is driving the effort to upgrade...
Siemens Healthineers will acquire all issued and outstanding shares of common stock of Corindus Vascular Robotics, Corindus announced this month.
The cash transaction is US$4.28 per share, resulting in an aggregate purchase price of US$1.1 billion by Siemens Medical Solutions,...
At the Charing Cross Symposium (London, UK), Ian Chetter (Hull, UK) presented on the pathway from assessment to treatment for patients with surgical wounds healing by second intention (SWHSI), discussing the impact this may have on such factors as...
Inhaling a vaporised liquid solution through an e-cigarette, also known as vaping, immediately impairs vascular function, even when the solution does not include nicotine, according to the results of a new study published in Radiology.
E-cigarette use is on the...
A study into the efficacy of early reflux elimination for venous leg ulcer (VLU) patients has revealed that the combination of ultrasound-guided foam sclerotherapy (UGFS) only, and a hybrid method of endovenous laser treatment (EVLT) with UGFS, could present...
MolecuLight Inc., a provider of handheld fluorescence imaging for real-time visualization of bacteria for chronic wounds, has announced it has secured a US$7.5 million term loan from Oxford Finance LLC, a specialty finance firm providing senior debt to life...
The impact of chronic wounds—a global “healthcare problem”—is felt by patients and the medical community, yet there is limited education on the management of wounds and no standard training programme currently in place. Karen Imma Gellada, a member of...
The BASIL-3 study, which was halted in response to findings reported in the Journal of the American Heart Association (JAHA) that paclitaxel-coated and –eluting devices were associated with an increased mortality signal when used in the femoropopliteal arteries, is...
International Cardio Corporation (ICC) announced that it has achieved the first human use of its high-intensity focused ultrasound (HIFU) system to designed to treat peripheral vascular disease (PVD).
The Minneapolis-based company said its HIFU Synthesiser system is undergoing a 15-patient...
Ascyrus Medical announced today that it has received breakthrough device designation from the US Food and Drug Administration (FDA) for its Ascyrus Medical Dissection Stent (AMDS) to treat acute Type A aortic dissections. This designation underlines the clinical importance...
Concept Medical has been granted breakthrough device designation from the US Food and Drug Administration (FDA) for its MagicTouch percutaneous transluminal angioplasty (PTA) sirolimus drug-coated balloon (DCB) catheter, used in the treatment of peripheral arterial disease (PAD) in infra-popliteal...
ZipLine Medical, Inc. has announced the publication of positive results from clinical studies in two peer-reviewed journals, demonstrating that the use of Zip Surgical Skin Closure leads to better clinical outcomes and healthcare cost savings than surgical staples.
A randomised,...
Engineers at Heriot-Watt University, Edinburgh, UK, are developing a sensor that will help patients, doctors and nurses manage how wounds heal. Wounds cost the NHS between £4.5 and £5.1 billion each year to manage, while burns, diabetic ulcers, caesarean...
A private equity firm, Palatine (Manchester, UK), has announced that it has made a “significant investment” into a UK centre—Veincentre—that provides endovenous laser ablation (EVLA) for the management of varicose veins. The investment, according to Palatine, is the fourth...
Ian Loftus (London, UK), president of the Vascular Society for Great Britain and Ireland, tells BLearning about the most recent developments in the UK’s NICE guidance on abdominal aortic aneurysm.
The draft version of the guideline created a furore, when...
Endologix has announced that the CE mark for the Nellix endovascular aneurysm sealing (EVAS) system has been reinstated. The CE mark for the system was suspended on 22 January and now it that has been reinstated, Nellix will be...
Results from two open prospective trials have revealed that the TLC-NOSF sucrose octasulphate dressing, with poly-absorbent fibres, represents an effective and safe treatment for the local management of leg ulcers. In addition, cost-effectiveness studies examining the economic impact of...
Endologix has announced the investigational device exemption (IDE) approval from the US Food and Drug Administration (FDA) to commence a new pivotal study to evaluate the safety and effectiveness of the Nellix Chimney EndoVascular Aneurysm Sealing System (chEVAS) for...
Speaking at the 2019 Society for Vascular Surgery (SVS) Vascular Annual Meeting (VAM; 12–15 June, National Harbor, USA), Samir Shah (Division of Vascular Surgery, Brigham and Women's Hospital, Boston, USA) reported that patients with dementia have worse outcomes after...
Medtronic has issued the following statement regarding the US Food and Drug Administration’s (FDA) updated letter to healthcare providers for paclitaxel-devices in patients with peripheral artery disease (PAD) in the superficial femoropopliteal artery (SFA):
“Medtronic would like to thank FDA...
Clinical studies of paclitaxel-coated balloons and paclitaxel-eluting stents “may continue and should collected long-term safety (including mortality) and effectiveness data”, says the US Food and Drug Administration (FDA) in a letter to healthcare providers on 7 August. This is...
The US Food and Drug Administration (FDA) have denied BD premarket approval (PMA) for the company’s Lutonix drug-coated balloon (DCB) in the treatment of peripheral arterial disease (PAD).
Speaking in the company’s third quarter conference call on 6 August, BD...
𝙏𝙝𝙞𝙨 𝙫𝙞𝙙𝙚𝙤 𝙞𝙨 𝙞𝙣𝙩𝙚𝙣𝙙𝙚𝙙 𝙛𝙤𝙧 𝙝𝙚𝙖𝙡𝙩𝙝𝙘𝙖𝙧𝙚 𝙥𝙧𝙤𝙛𝙚𝙨𝙨𝙞𝙤𝙣𝙖𝙡𝙨 𝙞𝙣 𝙀𝙪𝙧𝙤𝙥𝙚 𝙤𝙣𝙡𝙮.
Giovanni Torsello (Münster, Germany) and Mauro Gargiulo (Bologna, Italy) discuss the “very high” morbidity and mortality caused by open surgery when treating complex aortic aneurysms. This is why, they say,...
The US Food and Drug Administration (FDA) have denied BD premarket approval (PMA) for the company’s Lutonix drug-coated balloon (DCB) in the treatment of peripheral arterial disease (PAD).
Speaking in the company’s third quarter conference call on 6 August, BD...
Members of the International Working Group on the Diabetic Foot (IWGDF) presented up-to-date guidelines for the diagnosis, prognosis and treatment of diabetic foot ulcers at the 29th conference of the European Wound Management Association (EWMA; 5–7 June, Gothenburg, Sweden),...
Members of the International Working Group on the Diabetic Foot (IWGDF) presented up-to-date guidelines for the diagnosis, prognosis and treatment of diabetic foot ulcers at the 29th conference of the European Wound Management Association (EWMA; 5–7 June, Gothenburg, Sweden),...
Two-year results from the MOBILE (Marrowstim treatment of limb ischaemia in patients with severe peripheral arterial disease) trial indicate that injecting concentrated bone marrow aspirate (cBMA, Zimmer Biomet), vs. placebo, into patients with critical limb ischaemia is associated with...
Bentley has announced the company celebrated its tenth anniversary on June, 29th 2019. The announcement states that the story of the company is a story of remarkable global expansion driven by a dedicated workforce and an innovative product base...
The interim results of a multicentre, European retrospective analysis investigating the use of drug-coated balloons (DCBs) in symptomatic central venous stenosis (CVS) suggest that these devices could be a valid option for treatment, slowing down the effect of neo-intimal...
Two-year results from the MOBILE (Marrowstim treatment of limb ischaemia in patients with severe peripheral arterial disease) trial indicate that injecting concentrated bone marrow aspirate (cBMA, Zimmer Biomet), vs. placebo, into patients with critical limb ischaemia is associated with...
Jos van den Berg (Lugano, Switzerland), Antonio Micari (Bergamo, Italy) and Marianne Brodmann (Graz, Austria) sit down after a specially convened US FDA panel reviewed paclitaxel device data, with recommendations still to come. The physicians discuss the safety of...
The National Institute for Health and Care Excellence (NICE) in the UK has published a positive draft final appraisal determination recommending the use of rivaroxaban (brand name Xarelto, Bayer) by the National Health Service (NHS) in England. The NICE...
Reflow Medical, Inc announces that the first patients have been enrolled in its DEEPER OUS clinical trial using the Temporary Spur Stent System.
DEEPER OUS is a 100-patient prospective, non-randomised, multicentre trial designed to assess the safety and efficacy of...
GraftWorx has commenced a service evaluation in collaboration with the Academic Health Science Network (AHSN) and a National Health Service (NHS) Hospital Foundation Trust based in the North East of England.
After evaluating early data against standard measures, the...
In her presentation at the Royal Society of Medicine Venous Forum (RSMVF; 17–19 June, London, UK), consultant vascular surgeon Emma Wilton (Oxford University Hospitals, Oxford, UK) outlined the important process for identifying which patients with leg ulceration require deep...
Endovenous laser ablation (EVLA) of perforating veins has been established as an effective method of treating chronic venous ulcers in patients with diabetes mellitus, according to a new study conducted in Azerbaijan.
Results of the study, which involved 42 patients...
The first patient has been randomised in the TARGET BP I trial at the Piedmont Heart Institute in Atlanta, USA, to investigate the use of the Peregrine System Kit (Ablative Solutions) in renal denervation.
Target BP I is a blinded,...
A member of the UK parliament has emphasised the need for a national wound care strategy during a recent debate in the House of Commons, highlighting that “diagnosis is difficult, but rapid diagnosis is absolutely essential”. Ann Clywd, MP...
Laminate Medical Technologies has announced the enrolment of their first forearm fistula patients in their US pivotal trial of the VasQ device. VasQ is an implanted blood vessel external support intended to promote maturation and reduce the high primary...
Hoth Therapeutics, a biopharmaceutical company focused on targeted therapeutics for patients suffering from indications such as dermatological and chronic wound disorders, has announced the initial results of its pilot study on the application of the BioLexa Platform on diabetic...
NuCryo Vascular has announced that the company has signed a commercialisation agreement with Lokai Medical, a specialty distributor of coronary/peripheral and interventional devices, to distribute the PolarCath Balloon Dilatation System in the USA.
Balloon cryoplasty has been shown in clinical...
Karen Bauer (Toledo, USA) discusses the wound care limb salvage track at C3 which offered the “opportunity for clinicians from multiple disciplines to get together and talk about some really cutting-edge” topics.
Bauer, a wound specialist, says that at the...
Five-year data from the INSPIRATION US investigational device exception (IDE) pivotal trial confirms favourable safety and effectiveness of the INCRAFT device. Presenting the trial at the Society of Vascular Surgery (SVS) Vascular Annual Meeting (VAM; 12–15 June, National Harbor,...
Vascular malformations have perplexed a plethora of physicians from diverse specialties for decades, writes Jocelyn Brookes. The British Society of Interventional Radiology (BSIR) has formed a Vascular Anomalies Special Interest Group (VASIG), which met last year for a study...
The US Food and Drug Administration (FDA) has granted 510(k) clearance to Centerline Biomedical’s Intra-Operative Positioning System (IOPS), a device used in endovascular procedures to provide 3D visualisation.
The non-radiation-based surgical navigation system uses anatomical mapping algorithms and electromagnetic tracking...
Rajesh Dave (Camp Hill, USA) talks to iWounds News about his highlights from the 15th annual C3 conference, which included a special session on the paclitaxel controversy.
Dave talks about the “journey of 15 years” and how the meeting started...
What are the Top 10 calls a newly matched vascular surgery integrated resident is first likely to encounter in the real world? Patients are more likely to present with pain, nausea or swollen leg, rather than ruptured aneurysm. And...
When new rules capped training hours for medical residents at 80 hours per week in 2003, critics worried that the change would leave physicians-in-training unprepared for the challenges of independent practice. New research, published today in the British Medical...
Gore has announced the first US implant of its Gore TAG conformable thoracic stent graft with ACTIVE CONTROL system. The successful procedure was performed by William Jordan, chief of the Division of Vascular Surgery and Bradley Leshnower, cardiothoracic surgeon,...
A new visualisation method enables more precise catheter navigation through the vascular system. It is being developed at the Fraunhofer Institute for Digital Medicine (MEVIS) in Bremen, Germany, and one of its ambitions is to reduce physician radiation exposure.
The...
Mahmoud Malas (San Diego, USA) presented, for the first time, updated results from the TransCarotid Artery Revascularization (TCAR) Surveillance Project at the Society for Vascular Surgery (SVS) Vascular Annual Meeting (VAM; 12–15 June, National Harbor, USA).
Malas distilled the data...
Paul Hayes (Cambridge, UK), Frank Arko (Charlotte, USA) and Fabio Verzini (Turin, Italy) discuss the Valiant Navion™ (Medtronic) live thoracic endovascular aortic repair (TEVAR) case at Charing Cross 2019, which demonstrated the use of the new CoveredSeal proximal configuration.
Hayes...
Intraoperative perianastomotic delivery of sirolimus is feasible, according to Sriram Iyer (Lenox Hill Hospital, New York, USA). Based on preliminary data from 56 arteriovenous fistula (AVF) patients treated with intraoperative perivascular sirolimus, there are no safety concerns, and, using...
According to a recent study, an endovascular approach is “safe and effective” in the treatment of abdominal aortic aneurysms detected by the National Abdominal Aortic Aneurysm Screening Programme (NAAASP). Furthermore, the investigators found no aneurysm-related mortality in those treated...
The new West London Vascular and Interventional Centre at Northwick Park Hospital (Harrow, UK) has officially been declared open. As of the 25th June, 2019, patients who require vascular treatment will be able to benefit from the modern facilities...
Health technology solutions—such as mobile apps and wearables—can play a huge role in activity tracking and assessing the outcomes of peripheral vascular interventions, says Ronald L Dalman (Stanford, USA), president-elect of the Society for Vascular Surgery (SVS). Down the...
Micro Medical Solutions has received Investigational Device Exemption (IDE) approval from the US Food and Drug Administration (FDA). IDE approval allows the company to initiate a US pivotal clinical trial to evaluate MicroStent’s safety and efficacy. A press release...
Siemens Healthineers and Mentice AB have announced that they are collaborating to fully integrate Mentice’s VIST virtual patient into the Artis icono angiography system from Siemens Healthineers.
According to a press release, the VIST virtual patient thus becomes a fully...
In this issue:
CX audience deems paclitaxel not dangerous, while vascular pathologist says establishing the truth takes time (p.1)
EVAR funding threatened, while a strategy for safe, cost-effective repair is “just around the corner” (p.1)
Marianne Broadmann outlines follow-up...
In this issue:
CX audience deems paclitaxel not dangerous, while vascular pathologist says establishing the truth takes time (p.1)
EVAR funding threatened, while a strategy for safe, cost-effective repair is "just around the corner" (p.1)
Marianne Broadmann outlines follow-up...
The president-elect of the Society for Vascular Surgery (SVS), Kim Hodgson, begins his year as SVS president in 2019. In this profile interview with Vascular News, Hodgson discusses his career and research interests, how he came to be involved...
Both Medtronic and BD have released statements in response to the FDA circulatory system devices panel held last week (19–20 June, Washington, DC, USA). The companies reiterate the safety and efficacy of their own paclitaxel devices and state that...
Kim Hodgson (Springfield, USA), president of the Society for Vascular Surgery (SVS) for 2019–20 spoke to Vascular News at the organisation’s Vascular Annual Meeting in June to share the most influential data to have emerged from the Vascular Quality...
Dawn Coleman (Ann Arbor, USA) shares headline findings from a Society for Vascular Surgery (SVS) survey that found 41% of 960 respondents met validated metrics for burnout. “Burnout, depression and suicidal ideation represent a very real problem within our...
The US Food and Drug Administration (FDA) convened this week for a General Issues Panel Meeting on the late mortality safety signal associated with paclitaxel-coated products (i.e. paclitaxel-coated balloons and paclitaxel-eluting stents) used to treat peripheral arterial disease (PAD)...
A new Expert Panel Report, published in The Diabetic Foot Journal, recommends a new treatment algorithm for topical oxygen therapy in patients with diabetic foot ulcers (DFU). The new treatment algorithm recommends that topical oxygen therapy is considered as...
Ra Medical is conducting a study to demonstrate the DABRA Excimer Laser System delivers effective, long lasting results and a low reintervention rate for patients with peripheral arterial disease (PAD). The study will gather data two years post-treatment in...
The Society for Vascular Medicine (SVM) held its 30th Annual Scientific Sessions in McLean, USA earlier this month and announced its new leadership as well as presented its annual awards.
Raghu Kolluri of OhioHealth Heart & Vascular (Columbus, USA) was...
Vikram S Kashyap, co-principal investigator of the ROADSTER 2 trial, today presented the final 30-day outcomes of the post-market approval study in a late-breaking trial session at the Society for Vascular Surgery (SVS) Vascular Annual Meeting (VAM; 12–15 June,...
The OVER trial investigators, led by the late Frank Lederle, recently released the long-term results of endovascular aneurysm repair (EVAR) versus open repair. Janet Powell writes for Vascular News about the 14-year outcomes, which were published in the New...
The halted BASIL-3 study now plans to resume enrolment of patients into the randomised controlled trial, which is comparing balloon angioplasty versus stenting for severe ischaemia in the legs of patients with peripheral arterial disease (PAD). Two of the...
New research finds that a third of practicing vascular surgeons meet criteria for burnout, subsequently increasing the incidence of depression and suicide ideation within this cohort of physicians. Presenting these data at the Society of Vascular Surgery (SVS) Vascular...
The Society for Vascular Surgery (SVS) Vascular Quality Initiative (VQI), with its large multicentre registries has produced several datasets for analysis of vascular patient care in the USA. This year, at the SVS Vascular Annual Meeting (VAM; 12–15 June,...
The methodology underpinning the conclusion by Konstantinos Katsanos (Patras, Greece) et al that there is a positive dose-response relationship between paclitaxel and mortality is flawed, argues Andrew Holden (Auckland, New Zealand) and colleagues in a paper published in the...
A publishing agreement between the Society for Vascular Surgery (SVS) and BIBA Medical Ltd has been signed today by the respective directors at the Vascular Annual Meeting (VAM; 12–15 June, National Harbor, MD, USA). This collaboration makes BIBA Medical...
Orchestra BioMed and Terumo have formed a global strategic partnership for the development and commercialisation of Virtue sirolimus-eluting balloon (SEB) in the percutaneous coronary and peripheral interventions field.
Virtue SEB is the first and only non-coated drug-eluting angioplasty balloon that...
Andrew Holden (Auckland, New Zealand) and Peter Schneider (San Francisco, USA) discuss the Tack Endovascular System (Intact Vascular) and its impact on below the knee and above the knee procedures at the Charing Cross Symposium 2019. Schneider talks about...
Cardiovascular Systems recently announced that the first patient has been enrolled in the REACH PVI study. The purpose of this study is to prospectively evaluate acute clinical outcomes of orbital atherectomy via transradial access for the treatment of peripheral...
Cardiovascular Systems recently announced that the first patient has been enrolled in the REACH PVI study. The purpose of this study is to prospectively evaluate acute clinical outcomes of orbital atherectomy via transradial access for the treatment of peripheral...
New research from Finland found dramatic decreases in mortality from ruptured abdominal aortic aneurysms following a series of simulated staff training sessions on how to handle those life-threatening emergencies.
When haemodynamic compromise exists in this situation, a multitude of events...
Endologix recently announced that the EC Certificate of Conformity (CE mark) for the Nellix endovascular aneurysm sealing system (Nellix system) has been reinstated by GMED, the EU Notified Body for the Nellix system. The reinstatement followed an assessment of...
The TLC-NOSF (UrgoStart, Urgo Medical) advanced wound care dressing was recently recommended by the UK National Institute for Health and Care Excellence (NICE) for healing of venous and diabetic foot ulcers. Speaking at the European Wound Management Association annual...
The US Food and Drug Administration (FDA) has approved expanded sizes of the Lutonix 018 drug-coated balloon (DCB) to treat long superficial femoral artery lesions in patients with peripheral arterial disease.
According to a press release, the Lutonix 018...
Four years ago, vascular experts from around the world had a "quixotic quest", to get all surgeons and providers to agree on the best ways to treat a common and debilitating illness: chronic limb-threatening ischaemia. The goal has come...
Four years ago, vascular experts from around the world had a "quixotic quest", to get all surgeons and providers to agree on the best ways to treat a common and debilitating illness: chronic limb-threatening ischaemia. The goal has come...
Watch the video roundup from the CX 2019 Highlight Session, Paclitaxel: The last word.
Filmed onsite at the Charing Cross Symposium (15–18 April 2019, London, UK).
Ulf Teichgräber (Jena, Germany), talks to Vascular News at EuroPCR 2019 about the EFFPAC trial which compared the luminor (iVascular) drug-coated balloon (DCB) with plain old balloon angioplasty (POBA).
Teichgräber outlines the 24-month results from the trial specifically the secondary...
Do not use paclitaxel drug-coated balloons (DCBs) or drug-eluting stents (DESs) in the routine treatment of patients with intermittent claudication until further notice, a medical device alert (MDA) issued today by the UK Medicines and Healthcare products Regulatory Agency...
The SAVE-US (Surfacer system to facilitate access in venous occlusions—United States) pivotal trial has completed enrollment. The SAVE-US trial is evaluating the safety and efficacy of the Surfacer Inside-Out access catheter system (Bluegrass Vascular Technologies) in 30 patients across...
The UK Medicines and Healthcare products Regulatory Agency (MHRA) acted to limit the future use of paclitaxel-eluting stents and paclitaxel-coated balloons in routine clinical care following recommendations from the independent Expert Advisory Group (EAG) on the use of these...
Michael Edmonds is professor of Diabetic Foot Medicine, King’s College London, and consultant physician, King’s College Hospital, London, UK. A pioneer in many areas of the diabetic foot, Edmonds speaks about his interest in diabetes and its treatment, the...
Preliminary findings from an initial study with 25 patients have shown that measuring the changes in oxygen concentration at baseline, during, and after endovascular revascularisation, can predict wound healing and functional improvement in critical limb ischaemia patients.
Marianne Brodmann, Medical...
The opening of an outpatient wound centre has been associated with a significant increase in peripheral vascular practice and a significant decrease in amputation rate. Venita Chandra and colleagues Alyssa M Flores, Matthew W Mell and Ronald L Dalman...
The 2019 Charing Cross Symposium (CX; 15–18 April, London, UK) saw the introduction of a new strand of care with the debut of the iWounds Workshop to the exhibition hall. The iWounds Workshop aimed to promote early diagnosis of...
Keith Harding advocates for the increased involvement of medical staff in the treatment of patients with wound healing difficulties, as the number of chronic wounds seen in hospitals is on the rise. He argues that wounds “do not receive...
CX Executive Board member Una Adderley (Wakefield, UK), director of the National Wound Care Strategy Programme for the National Health Service (NHS) England, spoke recently at the 2019 Charing Cross Symposium (London, UK) on the challenge of ankle-brachial pressure...
There is a robust evidence base to support the use of compression therapy for healing venous leg ulceration and for preventing ulcer recurrence. The most recent version of the Cochrane Review1 on the effectiveness of compression and the relative effectiveness of different...
Speaking at the Leipzig Interventional Course (LINC; 22–25 January, Leipzig, Germany), Katja Mühlberg of the University of Leipzig, Germany, called for an improvement in the evidence base around the role of compression in healing venous leg ulcers (VLU). Mühlberg...
The UK's National Institute of Health and Care Excellence (NICE) has published new guidance recommending UrgoStart wound dressings (Urgo Medical) for treatment of diabetic foot ulcers and venous leg ulcers. In a press release, Urgo Medical states that use...
Healogics, a provider of advanced chronic wound care services in the USA, has announced they have added vein ablation to their in-house service offerings through a new programme called Healogics VeinCare. This programme allows the company to provide patients...
Despite what current guidelines denote, and in addition to the emergence of data supporting the early intervention of venous ulceration, Alun Davies (London, UK) put forward that the recommendations are consistently being ignored, and highlighted that: “Urgent action is...
“A lot more bacteria than anticipated” have been discovered on wounds in a patient cohort consisting of 89% venous leg ulcers, and using fluorescence imaging proved superior for detecting infection in wounds. This was the conclusion of Tomas Serena (Warren,...
Defining success in venous ulcer healing, and for all chronic wounds, remains a nebulous concept—with no consensus on what constitutes a successful outcome. Dave Bosanquet (Bristol, UK) outlined the difficulties at the Charing Cross Symposium (London, UK), arguing that...
Michael Edmonds (London, UK), Thomas Serena (Warren, USA), Keith Harding (Cardiff, UK), and Williams Ennis (Oak Lawn, USA) talk at Charing Cross Live about the first-ever iWounds workshop, the importance of wound management and what they hope to achieve.
Edmonds...
The US Food and Drug Administration have issued a Class I Recall of the SoloPath (Terumo Medical) ballon-expandable transfemoral system and re-collapsible balloon access system, due to malfunction. A total of 14 reports had flagged related incidents of a...
Investigators of SWEDEPAD have announced the conclusion of their safety committee analysis, which recommends the halted trials resume enrolment.
This decision to consider resuming enrolment in the SWEDEPAD trials came twelve days after a dedicated session called "Paclitaxel: The last...
Three-year results from the ILLUMENATE Pivotal trial and the ILLUMENATE European randomised controlled trial (EU RCT) have been presented in a late-breaking trial session at the New Cardiovascular Horizons Annual Conference (NCVH; 29–31 May, New Orleans, USA) by S...
It is imperative that every medical institution has an interdisciplinary CLI team, argues Jihad Mustapha. Here, he exhorts the benefits of working in such a multidisciplinary team, and outlines how the circle of care concept can be applied to...
ControlRad has announced that the US Food and Drug Administration (FDA) has granted 510(k) clearance for its ControlRad Trace. The company has initiated its commercial launch. The ControlRad Trace is the only technology that can be integrated into existing...
Proximo Medical has announced that it has been selected as the marketing and commercial partner for Walk Vascular, a medical device company that has developed JETi, a novel peripheral thrombectomy catheter.
Proximo Medical is a fractional commercial organisation for start-up...
Rexgenero, a regenerative medicine company developing advanced cell-based therapies to treat critical limb ischaemia (CLI), has announced that it has successfully expanded manufacturing of its lead development candidate REX-001 through a partnership with the German Red Cross Blood Donor...
A single-centre study of standard, fenestrated and branched endovascular aneurysm repair patients in Nürnberg, Germany, has found that outcomes for women are worse but the reasons are not yet clear, as literature on the topic is lacking. The results...
Eluvia DES is the only technology that uses polymer-based drug delivery to treat Peripheral Artery Disease (PAD). Its polymer ensures that the majority of drug is delivered to the tissue while minimizing downstream particulates. Boston Scientific has more than...
Data presented at EuroPCR today indicates that renal denervation with the Symplicity system (Medtronic) is associated with a reduction in subclinical atrial fibrillation in high-risk patients with hypertension over a median follow-up of two years.
In the study, 80 patients...
The UK National Institute for Health and Care Excellence (NICE) has released new recommendations for use of the PICO negative pressure wound dressing (Smith & Nephew) for closed surgical incisions, saying the negative pressure dressings are "associated with fewer...
The UK National Institute for Health and Care Excellence (NICE) has released new recommendations for use of the PICO negative pressure wound dressing (Smith & Nephew) for closed surgical incisions, saying the negative pressure dressings are "associated with fewer...
The long-awaited Global Vascular Guidelines for chronic limb-threatening ischaemia have been finalised, with the comprehensive guideline set to be published shortly. The news were announced by the Society for Vascular Surgery, one of the societies—alongside the European Society for...
Gore has announced that the US Food and Drug Administration (FDA) has granted regulatory approval for commercial distribution for the Gore TAG Conformable Thoracic Stent Graft with Active Control System, a unique thoracic endovascular aortic repair (TEVAR) solution combining...
Micro Medical Solutions (MMS), an innovator in the field of microvascular interventions that improve clinical outcomes and quality of life, announced today that John Fletcher has started as a strategic advisor to the company's senior management. Fletcher is the founder...
Until recently, creation, maintenance and restoration of vascular access function was entirely performed by open surgical methods. However, it is becoming apparent that endovascular management is a valid treatment option and a comparable alternative to the open surgical one,...
iD3 Medical has announced that it has received approval in Belgium to initiate a randomised controlled trial, BIOPACT-RCT: a physician-initiated multicentre trial evaluating the safety and efficacy of Biotronik’s Passeo-18 Lux drug-coated balloon (DCB) for the treatment of peripheral...
Intact Vascular has announced the first commercial use of its Tack Endovascular System in multiple sites across the USA. Recently FDA-approved for above-the-knee interventions, the first-of-its-kind dissection repair device is implanted postangioplasty to resolve dissections in patients with peripheral...
Misonix, Inc., a provider of minimally invasive therapeutic ultrasonic medical devices, has announced that it has entered into a definitive agreement with Solsys Medical, a regenerative medical company and allograft producer, to acquire Solsys in an all-stock transaction valued...
NuCryo Vascular, the manufacturer and marketer of the PolarCath Balloon Dilatation System, announced today that the company is raising a Series B round of financing and is launching its Extended PolarCath Balloon Dilatation Catheter.
The Series B financing will provide...
CX Executive Board member Una Adderley (Wakefield, UK), director of the National Wound Care Strategy Programme for the National Health Service (NHS) England, spoke recently at the 2019 Charing Cross Symposium (CX; 15–18 April, London, UK) on the challenge...
Keith Harding advocates for the increased involvement of medical staff in the treatment of patients with wound healing difficulties, as the number of chronic wounds seen in hospitals is on the rise. He argues that wounds “do not receive...
For the first time, the audience at the 2019 Charing Cross Symposium (London, UK) was able to experience an array of virtual reality live cases in both the peripheral and aortic programmes. The virtual reality live streaming in 360°...
The 2019 Charing Cross Symposium (London, UK) saw the introduction of a new strand of care with the debut of the iWounds Workshop to the exhibition hall. The iWounds Workshop aimed to promote early diagnosis of underlying conditions in...
Using EndoAnchors to fix and seal endovascular aortic grafts is safe and effective for short-neck abdominal aortic aneurysm (AAA) patients, two-year data from the ANCHOR global registry have shown. Frank Arko (Charlotte, USA) presented the findings at the 2019...
Implementing a method of heparinisation guided by activated clotting time (ACT), with a goal of 200–220 seconds, provides a “promising” increase in safety and may decrease risk of thromboembolic events, while not increasing bleeding complications. Presenting the findings of...
An off-the-shelf endograft for the treatment of thoracoabdominal aortic aneurysms (TAAA) provides a safe and straightforward solution for a wide range of patients—including emergency cases. Data for the Zenith T-branch endograft were presented by Martin Austermann (Münster, Germany) at...
At the 2019 Charing Cross Symposium (CX; 15–18 April, London, UK), Vincenzo Brizzi (Bordeaux, France) reported the 10-year results for late rupture after endovascular aneurysm repair (EVAR) at his centre (Groupe Hospitalier Pellegrin, Bordeaux, France). These showed that late...
In the Aortic Podium 1st session at the 2019 Charing Cross Symposium (CX; 15–18 April, London, UK), Fabio Verzini (Turin, Italy) reported the 30-day clinical results of the complete Valiant Navion (Medtronic) cohort. He told delegates that the acute...
Defining success in wound healing remains a nebulous concept, with no consensus on what constitutes a successful outcome. Dave Bosanquet (Bristol, UK) outlined the difficulties in a Charing Cross (CX) session yesterday on this topic, and said a “core...
At the 2019 Charing Cross Symposium (CX; 15–18 April, London, UK), there was a section of the programme on drug-coated balloon trial updates which ended with a panel discussion on whether peripheral vascular disease data is applicable to vascular...
As part of the CX Vascular Access Masterclass on Wednesday, Colin Deane (London, UK) considered the role of surveillance in thrombosis, asking whether there exists a test that is simple, safe, precise and timely. While it has been established...
Endovascular aneurysm repair (EVAR) using proximal polymer sealing does not seem to induce neck dilatation like other endografts, concluded Hence Verhagen (Rotterdam, The Netherlands) during the Aortic Podium 1st session at the 2019 Charing Cross Symposium (CX; 15–18 April,...
Coil embolisation for at-risk patients during endovascular aneurysm repair (EVAR) is a safe and reproducible procedure, Dominique Fabre (Le Plessis-Robinson, France) told delegates at the 2019 Charing Cross Symposium (CX; 15–18 April, London, UK).
Fabre presented the results of the...
Across the globe, vascular recruitment is struggling. This was the reason why delegates convened at the 2019 Charing Cross Symposium (CX; 15–18 April, London, UK) for a session dedicated to the topic, where representatives from three continents explained exactly...
Yesterday’s CX Highlight Session on the durability of endovascular aneurysm repair (EVAR) and the National Institute for Health and Care Excellence (NICE) aortic guidelines began with a live endovascular aneurysm repair performed by Stéphan Haulon (Le Plessis-Robinson, France). The...
Isaac Nyamekye (Worcester, UK) reported that compared with the Venefit and radiofrequency induced thermotherapy (RFITT) systems, endovascular radiofrequency ablation (EVRF) provided inferior truncal ablation at six months. He added this result indicated that operators should be more cautious about...
Xavier Berard (Bordeaux, France) gave delegates at the 2019 Charing Cross Symposium (CX; 15–18 April, London, UK) three fundamental principles to bear in mind when dealing with aortic graft infections using an open approach.
He advised the audience to first...
During the CX Peripheral and Aortic Workshops at the 2019 Charing Cross Symposium (London, UK), experts demonstrated a wide range of peripheral and aortic technologies and techniques. Throughout break times, supervised learning has been carried out at hands-on stations,...
The 2019 Charing Cross Symposium (London, UK) featured an Aortic Techniques & Technologies edited live cases session, which aimed to give delegates the opportunity to explore the application of different technologies in complex situations, which are perhaps not suitable...
Marianne Brodmann outlines follow-up and surveillance protocols after endovascular treatments for peripheral arterial disease (PAD), arguing for a standardised approach to ensure best practice for patients who have gone through peripheral endovascular procedures.
Endovascular procedures have become a key element...
Rexgenero, a regenerative medicine company working to develop cell-based therapies to treat critical limb ischaemia (CLI), has presented an update on its two Phase III SALAMANDER trials being conducted across Europe for its lead development programme REX-001 to treat...
In collaboration with the China Endovascular Course (CEC), at Charing Cross Symposium (CX; 15–18 April, London, UK) played host to CEC@CX. Wei Guo (Beijing, China), Daqiao Guo (China) and Sheng Wang (Beijing, China) co-chaired a session focused on endovascular...
“CX meets Latin America” is always an extremely popular and well attended programme, and this year was no exception. Charing Cross Symposium 2019 (CX; 15–18 April, London, UK) brought together practitioners from across the region, providing a forum for...
Cook Medical has released de-identifiable patient-level data from a clinical trial of its Zilver PTX peripheral paclitaxel-eluting stent.
The move comes a month after the US Food and Drug Administration (FDA) said that its preliminary review of long-term follow-up data...
Steven Rogers (Manchester, UK) presented the case for using contrast-enhanced tomographic 3D ultrasound imaging for peripheral arterial disease, telling the audience at Charing Cross Symposium (CX; 15–18 April, London, UK): “Our surgeons certainly prefer the 3D vein mapping to...
Surmodics has announced it has received US Food and Drug Administration (FDA) 510(k) clearance for its Sublime guide sheath.
The highly flexible, proprietary Xtreme braid-reinforced Sublime sheath is designed to resist kinking and maximise strength while retaining a low profile....
New evidence supports the utility of spatial frequency domain imaging (SFDI) for identifying compromised circulation in patients at risk of peripheral arterial disease (PAD). These data were published in the Journal of Vascular Surgery, in a study that describes...
European guidelines should do more to promote the use of ambulatory endovascular peripheral interventions and to support physicians wishing to set up an outpatient centre, said Yann Gouëffic (Nantes, France) at the Charing Cross Symposium (CX; 15–18 April, London,...
Novel data from the Stroke from Thoracic Endovascular Procedures (STEP) study has provided insight into current practice of thoracic endovascular aortic repair (TEVAR), and the future role of diffusion-weighted magnetic resonance imaging (DW-MRI) in reducing stroke from endovascular repair....
On Wednesday, during the Aortic Podium 1st session at Charing Cross (CX) Symposium, Fabio Verzini (Turin, Italy) reported that endovascular aneurysm repair (EVAR) with an iliac branch endoprothesis (Gore) is safe and effective. He added that the procedure was...
Following the latest information presented at the Charing Cross (CX) International Symposium related to the meta-analysis of paclitaxel-coated devices (Katsanos et al), hear from a multi-specialty panel of global experts who are discussing their current practice and the way they...
Office-based lower extremity arterial interventions are feasible, safe, and cost-effective, and provide high levels of patient satisfaction. Enrico Ascher (Brooklyn, USA) presented data that “challenge the dogma that high-risk patients should not be offered office-based endovascular infrainguinal arterial procedures”,...
At the Charing Cross International Symposium, Gary Ansel (Columbus, Ohio) moderates a global panel that includes Thomas Albrecht (Berlin, Germany), Peter Schneider (San Francisco, USA) and Eric Secemsky (Boston, USA). The panel outlines the value of paclitaxel devices in...
Despite what current guidelines denote, and in addition to the emergence of data supporting the early intervention of venous ulceration, Alun Davies (London, UK) put forward that the recommendations are consistently being ignored, and highlighted that: “Urgent action is...
The first-ever 60-month data on Medtronic’s VenaSeal closure system were presented on Tuesday, indicating that at five years, treatment with the cyanoacrylate adhesive for closure of diseased vein segments was not inferior to the alternative treatment arm of radiofrequency...
There were calls for greater transparency and sharing of the available randomised controlled trial data of paclitaxel-coated devices at the CX 2019 Highlight Session, Paclitaxel: The Last Word. The session also emphasised the need for a meta-analysis of individual patient-level data...
Delegates voted overwhelmingly against the notion that there was a demonstrable danger in any organ of the body attributed to circulating paclitaxel. These polling results, with a 67% majority, came after vascular pathologist Elena Ladich (Hollywood, USA) presented insights...
The durability of endovascular aneurysm repair (EVAR) is in the limelight as the UK vascular community awaits the final aortic aneurysm guidelines from the National Institute for Health and Care Excellence (NICE), set to go against widespread endovascular practice...
New data from the MIMICS-2 trial show the BioMimics 3D stent, which mimics natural vascular curvature, remains safe and effective for patients with symptomatic atherosclerotic disease of the femoropopliteal arteries two years after implantation.
Principal investigator Timothy Sullivan (Abbott Northwestern...
This post is sponsored by iVascular
Interview with Vicente Riambau (Hospital Clinic de Barcelona, Spain) about the paclitaxel matter. Are really all the drug-coated balloons the same?
This post is sponsored by iVascular
Interview with Koen Deloose (AZ Sint-Blasius, Belgium) about paclitaxel safety and the outcomes of Luminor in different types of lesions.
Surmodics has announced the company has now resumed patient enrolment into its TRANSCEND clinical trial, and is nearly 75% of the way to its goal of 446 randomised patients.
Patient enrolment in TRANSCEND, the pivotal clinical trial for the SurVeil...
Boston Scientific has announced results from sub-analyses of the IMPERIAL clinical trial for the Eluvia Drug-Eluting Vascular Stent System at the 41st Charing Cross (CX) Symposium (15–18 April, London, UK). Data demonstrated that the efficacy and safety performance of...
Intact Vascular has announced it received US Food and Drug Administration (FDA) approval for the Tack Endovascular System (6F), a purpose-built dissection repair device implanted post-angioplasty in patients with peripheral arterial disease (PAD).
Regulatory approval was based on data from Intact...
Avinger has announced that the company received 510(k) clearance from the US Food & Drug Administration (FDA) for its Pantheris SV (small vessel) image-guided atherectomy system.
Avinger is a developer of image-guided, catheter-based systems for the diagnosis and treatment of...
Reflow Medical has announced the completion of enrolment in its Wing-IT investigational device exemption (IDE) trial; a prospective, multicentre, nonrandomised study evaluating the ability of the Wingman catheter to cross chronic total occlusions (CTOs) in peripheral lesions after failed...
Biotronik has launched the Carnelian support catheter, designed to improve the access for treatment of tortuous and highly calcified lesions. Carnelian support is indicated for the exchange and support of guidewires into peripheral vasculature.
The launch of the Carnelian support...
The Cardiovascular and Interventional Radiological Society of Europe (CIRSE) has released a statement on the use of paclitaxel-coated balloons and stents in the treatment of peripheral arterial disease (PAD) following the paper published late last year by Konstantinos Katsanos...
Pursuit Vascular, Inc., manufacturer of ClearGuard HD Antimicrobial Barrier Caps, has announced that the company has received both (CE) Mark and Medical Device Single Audit Program (MDSAP) certificates for its haemodialysis catheter caps, paving the way for launch in...
Researchers’ hopes that metformin could reduce the growth of abdominal aortic aneurysms (AAA) recently got a substantial boost from a large study, which found that the diabetes drug reduced aneurysm growth by 23%. As reported in the March 2019...
Following the recent US Food and Drug Administration (FDA) approval of Cook Medical’s Zenith Dissection Endovascular System, Cooper University Health Care in Camden, USA, has treated the first patient in the US with the device as part of Cook’s...
BD has announced the launch of their UltraScore 014 Focused Force percutaneous transluminal angioplasty (PTA) Balloon. This marks the first scoring balloon with sheath compatibility as small as 4F on the US market.
The UltraScore balloon is intended to dilate...
In response to the updated letter to healthcare providers from the US Food and Drug Administration (FDA), the Society for Vascular Surgery (SVS) released a letter from SVS president Michel Makaroun and Kim Hodgson, chair of the SVS paclitaxel...
Jan Blankensteijn (Amsterdam, The Netherlands; principal investigator of the DREAM trial) tells Vascular News about what inspired him to design the DREAM trial, how its findings became particularly important with the long-term follow-up data for endovascular aneurysm repair (EVAR)...
Roger Greenhalgh (London, UK), was honoured at the 2018 VEITHsymposium alongside other endovascular aneurysm repair (EVAR) pioneers. He spoke to Vascular News about his conviction in the late 1990s that prospective randomised controlled trials were the way to properly...
The Medicines and Healthcare products Regulatory Agency (MHRA) in the UK and the Food and Drug Administration (FDA) in the USA have both recently issued new statements on the use of paclitaxel-eluting devices. While the FDA release preliminary analysis...
Endospan has announced that it has received CE mark for its Nexus stent graft system, to be used for branched endovascular repair in the aortic arch.
Diseases of the aortic arch present surgeons with challenging surgical treatment options and risks...
The US Patent and Trademark Office (USPTO) has granted the Ten Medical Design AB (10MD) patent application, Radiation Protective Material (US2016060791), a press release from the Sweden-based company states.
This patent describes ways for the surgical staff to stay protected...
A new paper looking specifically at mortality has found no difference in deaths between paclitaxel-coated balloons and uncoated balloons when used in the femoropopliteal artery. These data, a pooled analysis of four randomised controlled, multicentre trials, were published in...
"Paclitaxel kills the cells in the artery wall; sirolimus simply stuns them," says Peter Gaines (Sheffield, UK). Gaines commented at the 2019 Vascular Leaders Forum (VLF; 1–2 March, Washington, DC, USA) on the modes of action of these two...
Paclitaxel Timeline:
Meta-analysis finds a higher risk of death in the long term when paclitaxel-coated devices are used in the leg
Without additional long-term and patient-level data, Katsanos et al’s conclusion is premature
SWEDEPAD 1 and SWEDEPAD 2 trials...
In this issue:
CX 2019 highlights
A year in profiles
US FDA continues to investigate paclitaxel devices in the leg
European vascular community continues to dispute draft NICE AAA guidelines amidst publication delays
Krishna Rocha-Singh (Springfield, USA) tells Vascular News that plans after VLF 2019 include publishing a white paper and expeditiously pooling individual patient-level data from the industry randomised controlled trials for independent statistical analysis. This analysis would validate, or disprove,...
In this issue:
CX 2019 highlights
A year in profiles
US FDA continues to investigate paclitaxel devices in the leg
European vascular community continues to dispute draft NICE AAA guidelines amidst publication delays
BD has announced that the Covera vascular covered stent has received premarket approval by the US Food and Drug Administration (FDA) for use in treatment of end stage renal disease patients on haemodialysis. The company states their device is...
The National Kidney Foundation Kidney Disease Outcomes Quality Initiative (NKF KDOQI) provides evidence-based clinical practice guidelines for haemodialysis vascular access and related complications, and has done so since 1997. The latest guidelines, published in 2006, no longer reflect the...
With “an indisputable association between depression and peripheral arterial disease (PAD)”—nearly a third of PAD patients experience comorbid depression or depressive symptoms—Joel Ramirez and Marlene Grenon here call for vascular interventionalists to take a more active role in their...
The commissioner of the US Food and Drug Administration (FDA), Scott Gottlieb, has unexpectedly resigned after serving just shy of two years in the post.
Gottlieb announced his intention to stand down in a letter to Alex Azar II, the...
This article is an advertorial by Lombard Medical.
Endovascular repair presents a safe and effective treatment method, but late failure of endovascular aneurysm repair (EVAR) and endoleak both remain a problem for vascular specialists. A recent paper in the Journal...
Ra Medical Systems has announced study results from Athar Ansari, director of the California Heart & Vascular Clinic in El Centro, USA. The study examined the company's DABRA laser system for the endovascular treatment of blockages resulting from lower...
The Society for Vascular Surgery (SVS), the premier scientific organisation devoted to the treatment of vascular disease, is concerned about a recent article in the Journal of the American Heart Association (JAHA) by Konstantinos Katsanos et al suggesting higher...
Medtronic has issued the following statement regarding revised clinical study data:
On 15 February, 2019, Medtronic issued a statement regarding a programming error in the clinical data reporting isolated to the two and three year follow-up periods in our IN.PACT...
In the last 20 years, there have been major developments in the use of endovascular and hybrid haemorrhage control tools, especially in the treatment of ruptured abdominal aortic aneurysm (rAAA).
It has been shown that total endovascular aortic repair (EVAR)...
The Vascular Leaders Forum (VLF, 1–2 March, Washington DC, USA), hosted by the non-profit organisation VIVA Physicians, is a special consortium which was called in the wake of the meta-analysis published by Katsanos et al late last year, and...
“Drug-eluting stents (DES) are important additions to the armamentarium of devices used for peripheral artery revascularization, associated with decreased rates of restenosis and target vessel revascularization”, Eric Secemsky (Boston, USA) and colleagues write in the Journal of the American...
The new Simbionix ANGIO Mentor iCase (3D Systems) is a new endovascular simulation case editor which can be used to create endless training scenarios, a press release from the company states.
Virtual reality simulators are being used to help specialists...
The Charing Cross International Symposium (CX) continues its three-year cycle of raising vascular and endovascular controversies in order to challenge the available evidence and be able to reach a consensus after discussion with an expert audience.
CX will, as always,...
MedAlliance SA has been granted Breakthrough Device Designation from the US Food and Drug Administration (FDA) for SELUTION, its sustained limus release (SLR) drug-coated balloon (DCB) catheter, for the treatment of coronary disease.
The FDA Breakthrough Device Program is...
At the MWC (25–26 February, Barcelona, Spain), formerly the Mobile World Congress, Philips unveiled a unique mixed reality concept developed in partnership with Microsoft for the operating room of the future. Based on Philips’ Azurion image-guided therapy platform and Microsoft’s...
Gary Ansel (Columbus, USA) tells Vascular News at VEITHSymposium 2018 about how drug-based therapies have become a mainstay in femoropopliteal revascularisation and how interventionalists should be using best medical therapy “more than we do ”. He also shares his...
At the EKOS symposium at LINC 2019, Nils Kucher (Zurich, Switzerland), Houman Jalaie (Aachen, Germany) and Mert Dumantepe (Istanbul, Turkey) share their experiences in treatment of pulmonary embolism, acute and chronic deep venous thrombosis (DVT) using acoustic pulse thrombolysis.
This...
In this article, Konstantinos P Donas and Frank J Criado review the evidence for endovascular aneurysm repair using the chimney technique, and consider where current guidelines and research are leading the vascular community when it comes to endovascular strategies...
Marianne Brodmann (Graz, Austria) quizzes Michael Lichternberg (Arnsberg, Germany) at LINC 2019 on his choice of tools to identify and repair dissections in both the superficial femoral artery and below the knee.
If there is dissection that shows a haemodynamic...
France-based medtech company Affluent Medical has announced positive results from its SCOPE 1 clinical trial validating the efficacy of Kardiozis technology: an endoprosthesis using thrombogenic fibre embolisation of the aneurysm sac to prevent endoleak and reintervention.
The results were presented at the...
Philips Zenition, a new mobile C-arm imaging platform from Philips, has launched in the USA, Germany, Austria and Switzerland. It is being introduced into these countries over the next six months, and is set to penetrate other markets in...
Medtronic has issued the following statement regarding revised clinical study data:
Recently, Medtronic became aware of a programming error in the clinical data reporting isolated to the two- and three-year follow-up periods in our IN.PACT Global post-market study, part of...
BD has announced the 510(k) clearance from the US Food and Drug Administration (FDA) for the WavelinQ 4F endoAVF system earlier this month. The endoAVF system is the company’s most recent innovation in endovascular arteriovenous fistula (endoAVF) creation technology, which...
Even though the number of deaths due to ruptured aortic aneurysms (rAAA) has decreased in the USA by 68% in recent years, a significant number of deaths from ruptured aortic aneurysms occur in patients whose demographics exclude them from...
A recent US-wide, multicentre analysis has found no evidence of increased all-cause mortality associated with the use of paclitaxel-coated devices compared with non-drug-coated devices for femoropopliteal artery revascularisation. The study, published in JAMA Cardiology, sought to evaluate differences in...
Two-year data on the Passeo-18 Lux drug-coated balloon (DCB; Biotronik) continue to validate its safety and effectiveness in intra-inguinal arteries. Gunnar Tepe from the Klinikum Rosenheim in Germany presented 24-month results for the first time from the full cohort...
Nicholas Inston (Birmingham, UK) tells Vascular News at LINC 2019 about the exciting field of percutaneous arteriovenous fistula (AVF) creation, the main devices and trials in the space and the lack of long-term evidence showing AVFs created percutaneously “are still...
At a session dedicated to the draft abdominal aortic aneurysm (AAA) guidelines put forward by the UK’s National Institute of Health and Care Excellence (NICE) at Controversies and Updates in Vascular Surgery (CACVS; 7–9 February, Paris, France), the controversy...
Jihad Mustapha (Grand Rapids, USA) explains how the Temporary Spur stent system (Reflow), which is a spiked self-expanding stent is intended for use in conjunction with a drug-coated balloon, works to help deliver drug to the media or subintima...
Hans-Henning Eckstein (Technical University of Munich TUM, Munich, Germany)and Martin Storck (Klinikum Karlsruhe, Karlsruhe, Germany) show Vascular News the rubber duck that they give patients undergoing vascular and endovascular procedures under local anaesthetic. The patient holds the duck, which...
In the arterial space, VIVA 2018 focused on the fact that there is an increasing awareness of limb threatening ischaemia coupled with an increase in interventional tools, but no clear defined pathway for treatment. There is also uncertainty about...
Mario Lachat (University of Zürich, Zürich, Switzerland) presented mid-term results of an open-label, non-randomised single-arm investigational clinical study of the Nexus (EndoSpan) aortic arch stent graft system in 25 patients. Speaking at Controversies & Updates in Vascular Surgery (CACVS;...
In this issue:
Meta-analysis finds a higher risk of death in the long term when paclitaxel-coated devices are used in the leg (p.1)
Draft NICE aortic guidelines cause endovascular controversy (p.1)
Study finds increased risk of abdominal cancer associated...
In this issue:
Meta-analysis finds a higher risk of death in the long term when paclitaxel-coated devices are used in the leg (p.1)
Draft NICE aortic guidelines cause endovascular controversy (p.1)
Study finds increased risk of abdominal cancer associated...
The UK's National Institute of Health and Care Excellence (NICE) has published new guidance recommending UrgoStart wound dressings (Urgo Medical) for treatment of diabetic foot ulcers and venous leg ulcers. In a press release, Urgo Medical states that use...
Cook Medical has announced its recent approval from the US Food and Drug Administration (FDA) for its Zenith endovascular aortic dissection system.
The system, consisting of a proximal stent-graft component and a distal bare stent component, provides physicians a...
Teleflex has received premarket approval (PMA) from the US FDA for the Manta vascular closure device. A press release reports that the device is the first commercially available biomechanical vascular closure device designed specifically for large bore femoral arterial...
StimLabs has announced the first patient enrolment in the company's clinical trial assessing the safety and efficacy of full-thickness human placental graft Revita as a wound covering for diabetic foot ulcers.
The multicentre randomised, controlled study will evaluate the efficacy...
InspireMD has announced positive interim results from the SIBERIA trial, an investigator-initiated study of the CGuard embolic prevention system (EPS). The data were presented at the Leipzig Interventional Course (LINC; 22–25 January, Leipzig, Germany).
Andrey Karpenko and Pavel Ignatenko from...
The FDA has granted approval to include the use of atherectomy for lesion preparation in its ongoing US pivotal study of the Chocolate Touch drug-coated balloon (DCB; QT Vascular) and the addition of subgroup analysis related to the use...
The PreludeSYNC DISTAL compression device (Merit Medical) is now available in US, European, Middle Eastern, African, and Asia-Pacific markets. The device is the first product specifically designed to achieve haemostasis of the radial artery during procedures where access is...
Alberto Muñoz tells Vascular News about his 35 years in vascular surgery, including his involvement in the World Federation of Vascular Societies, and discusses the landmark advances made in these decades and what he hopes is yet to come.
Why...
Dierk Scheinert (Leipzig, Germany) talks to Vascular News at LINC 2019 about the importance of vessel preparation with adjunctive therapies such as the use of the dedicated scoring balloon, ULTRASCORE (BD), prior to the use of a drug-coated balloon...
Euphrates Vascular, a company focused on the treatment of vascular occlusions, recently received 510(k) clearance for sale of its ICHOR Percutaneous Reperfusion System.
ICHOR is a Percutaneous Reperfusion System designed to treat organised thrombus and embolic events in the peripheral...
XableCath, Inc. has announced that its blunt and abrasion tip catheters have been cleared by the US Food and Drug Administration (FDA) as peripheral crossing catheters. The catheters have been safely and successfully used to cross challenging lesions in...
Medtronic has released new data supporting the long-term durability, safety, and efficacy of the Valiant Captivia thoracic stent graft system for the treatment of blunt thoracic aortic injury (BTAI). Himanshu J Patel of the University of Michigan Department of...
Renowned experts expressed confidence in the continued use of paclitaxel-coated devices after independent patient-level data presented at LINC 2019 did not show correlation between paclitaxel exposure and mortality.
Findings from Baim (formerly Harvard Clinical Research Institute) are now published in...
Recent technological advancements, including the use of micromesh stents, greater adoption of proximal protection and new techniques such as transcarotid artery revascularisation (TCAR) are taking the field of carotid artery stenting (CAS) forward. A major advance will be getting reimbursement...
Israeli start-up Laminate Medical Technologies has announced a capital raising round of US$12 million. This round brings total investment in the company to date to US$24 million.
Participating in the current round is the leading dialyser manufacturer in Japan, Asahi...
Anthony Comerota, of the Inova Medical Group, Alexandria, USA, received the career achievement award at the 31st International Symposium on Endovascular Therapy (ISET; 27-30 January 2019, Hollywood, USA).
Speaking to Vascular News, Comerota expressed his gratitude: "This is one of the...
Sean Lyden (Cleveland, USA) and Fabrizio Fanelli (Rome, Italy) discuss the safety and future of drug-coated balloons (DCB) in light of the latest pooled analysis of patient-level data of over 2,300 patients treated with Philips’ Stellarex DCB in above-the-knee...
The editors-in-chief of major cardiovascular journals—of both US and European societies—have come together to “sound the alarm” about the dangers of medical misinformation that has been disseminated through the internet, social media, and other platforms. They claim that this...
Philips launches its Azurion with FlexArm to aid patient imaging and positioning during image-guided procedures.
During increasingly complex interventions, clinicians need to quickly and easily visualise critical anatomy and identify changes to the patient during the procedure. Azurion with FlexArm...
The discussion around the use of paclitaxel-coated and eluting devices continues at the 31st International Symposium on Endovascular Therapy (ISET; 27-30 January 2019, Hollywood, USA). Organisers hosted a paclitaxel town hall to consider whether or not the recent meta-analysis from Katsanos and...
Profusa has announced promising clinical data from two studies evaluating the company's Lumee Oxygen Platform, a tiny, injectable tissue-integrated biosensor and an intelligent data platform intended for continuous, real-time monitoring of tissue oxygen levels. The data, presented at the...
Eximo Medical have announced the first commercial use of their B-Laser atherectomy system for treatment of peripheral vascular disease. Barry Weinstock of Orlando Heart & Vascular Institute, director of the catheterisation laboratory at the Center for Cardiovascular Excellence in Orlando, USA used the B-Lase...
Experts from the Society for Cardiovascular Angiography and Interventions (SCAI) have reviewed the recent meta-analysis by Katsanos and colleagues and have concluded that the methods are appropriate and within constraints of trial-level meta-analysis. However, the society statement says “it...
The CE Mark for the Nellix endovascular aneurysm sealing system (Endologix) has been suspended by its Notified Body, GMED, following a voluntary recall and Field Safety Notification (FSN) issued by Endologix on January 4, 2019.
“The notification from GMED is...
The IN.PACT Admiral drug-coated balloon (DCB; Medtronic) exhibits a consistent and durable treatment effect in “a more complex patient demographic than typically seen in other DCB pivotal trials”, Osamu Iida (Kansai Rosai Hospital, Hyogo, Japan) informed the audience at...
Biotronik has presented the company's Pulsar-18 T3 stent system for the first time during the Leipzig Interventional Course (LINC; 22–24 January, Leipzig, Germany).
The Pulsar-18 T3 system is a unique combination of three technologies for an improved implantation procedure, a...
New results suggest that the Tigris vascular stent (Gore) is a safe and effective device that can be incorporated into a modern “leave-nothing-behind” treatment algorithm that reserves stent usage for more challenging patients. These findings were recently presented at...
A new pooled analysis of patient-level data from Philips demonstrates the “strong safety profile” of the company's Stellarex drug-coated balloon (DCB) in above-the-knee studies, according to a company statement. The independent, third party pooled analysis evidences low mortality rates...
The findings published by Konstantinos Katsanos and colleagues in JAHA quickly sparked wide-spread debate across the vascular community, as the meta-analysis’ implications and questions raised for use of paclitaxel-coated devices are discussed. The data had a near-immediate impact on...
Martin T Rothman has been appointed as chief medical officer of Vascular Dynamics, effective 1 January 2019.
Rothman is a pioneer in interventional cardiology, as evidenced by his clinical practice spanning several decades, his extensive pre-clinical and clinical research presented...
African Americans who smoke cigarettes are more likely than those who do not smoke to develop peripheral arterial disease (PAD), according to new research published in the Journal of the American Heart Association, the open access journal of the...
African Americans who smoke cigarettes are more likely than those who do not smoke to develop peripheral arterial disease (PAD), according to new research published in the Journal of the American Heart Association, the open access journal of the...
New data on the IN.PACT Admiral drug-coated balloon (DCB; Medtronic) in patients with peripheral arterial disease (PAD) in the superficial femoral (SFA) and popliteal arteries finds no statistically significant difference in all-cause mortality when the DCB or plain balloon...
Findings presented at the Leipzig Interventional Course (LINC; 22–25 January 2019, Leipzig, Germany) contradict the results of a meta-analysis published in the Journal of the American Heart Association in December 2018 linking paclitaxel-coated devices and increased mortality. Several presentations...
A renowned panel, moderated by Dierk Scheinert (Leipzig, Germany), comprising Peter Schneider (Honolulu, USA), John Laird (St Helena, USA) and Thomas Zeller (Bad Krozingen, Germany) expressed confidence in the continued use of paclitaxel-coated devices after individual patient-level data were...
Gore has introduced reduced profiles for the most commonly used diameters of the GORE TAG Conformable Thoracic Stent Graft with ACTIVE CONTROL System. The reduced profile allows physicians to perform TEVAR in patients with smaller vessels where access is challenging and...
Enrolment of the first patients in the BELONG study—benefit of arterial vessel preparation by arterial by longitudinal micro-incisions before drug-eluting balloon angioplasty of the superficial femoral and popliteal arteries—has been announced by VentureMed Group.
The primary objective of this clinical...
The impact of the 2018 meta-analysis of randomised paclitaxel-device trials, published in the Journal of the American Heart Association by Konstantinos Katsanos et al, continues to travel through the vascular world. As industry starts to respond to the conclusions...
The US government shutdown means the country’s Food and Drug Administration (FDA) cannot accept new user fees, which means the agency cannot accept new medical product applications.
FDA commissioner Scott Gottlieb took to Twitter to highlight agency employees who are...
Medical device company PQ Bypass has entered into an agreement that provides for up to an aggregate of US$60 million in equity financing, led by Deerfield Management. Joining the round are existing investors, including Seroba Life Sciences and MedTech...
Philips launches its Azurion with FlexArm platform to aid patient imaging and positioning during image-guided procedures.
Azurion with FlexArm allows clinicians to intraoperatively visualise patient anatomy in both 2D and 3D. As the clinician moves the system, the image beam...
A population-based cohort study by Viknesh Sounderajah and colleagues at Imperial College in London, UK, found an increased risk of abdominal cancer associated with endovascular anurysm repair—irrespective of whether or not CT surveillance was included in follow-up post-EVAR. Presenting...
For the vascular community in the UK, discourse on guidelines in 2018 increasingly came to focus on the proposed guidelines document from the National Institute for Health and Care Excellence (NICE), which in a draft published in May outlined...
Vascular Therapies, a biotechnology company focused on improving outcomes for patients with advanced kidney disease, today announced the appointment of John McDermott as the company's chief executive officer.
Bob Croce, chairman of the Board of Directors of Vascular Therapies, Inc. comments, "We...
Speaking to investors at the JP Morgan healthcare conference in San Francisco, executives from Medtronic and Boston Scientific said their data do not show the safety risks cited in Katsanos (Patras, Greece) et al’s published analysis.
According to the Star...
This article is an advertorial by Lombard Medical.
The outcomes at five years of the Aorfix (Lombard Medical) PYTHAGORAS trial, published in the Journal of Vascular Surgery, provided the first long-term data from an endovascular aneurysm repair (EVAR) cohort to...
This article is an advertorial by Lombard Medical.
Endovascular aneurysm repair (EVAR) is an established treatment modality in vascular practice which continues to develop, as new devices and technology continuously provide a greater range of options for the treatment of...
This article is an advertorial by Lombard Medical.
Four-year results of a clinical trial experience of 90 endovascular aneurysm repair (EVAR) patients treated with the Altura endograft system (Lombard Medical) were presented at the 2018 VEITHsymposium (13–17 November, New York,...
The first commercial use of the Tack Endovascular System (Intact Vascular) has taken place in multiple hospitals within Germany. A novel therapy for dissection repair following balloon angioplasty, the Tack implant is a first-of-its kind device for patients with...
Unrestricted sales and use of the Nellix System (Endologix) will cease immediately, in order to ensure optimal outcomes for patients, according to Endologix. The product will only be available for use under clinical protocol with pre-screened patients that adhere...
Ablative Solutions has announced the first close of its US$77 million Series D funding round. The round was led by new investor Glide Healthcare, and co-led by exiting investor BioStar Ventures, as well as an undisclosed new strategic corporate...
The Tack Optimised Balloon Angioplasty II below-the-knee (TOBA II BTK) clinical trial has successfully completed enrolment, ahead of schedule. This study, sponsored by Intact Vascular, is the first pivotal trial investigating a permanent vascular implant in arteries below the...
The first patients in the United States have been treated using the OrbusNeich Teleport Microcatheter (Teleport; Cardiovascular Systems), which recently received US Food and Drug Administration (FDA) 510(k) clearance.
Microcatheters are used to provide support and safe guidewire exchange during...
A pooled analysis of four randomised controlled trials finds drug-coated balloon (DCB) angioplasty superior to plain angioplasty in patients with femoropopliteal artery disease, irrespective of their demographics, cardiovascular risk factors, comorbidities or lesion characteristics. This conclusion was recently published...
The Valiant NAVION stent graft system (Medtronic) has been approved by the US Food and Drug Administration (FDA) for inclusion in a physician-sponsored investigational device exemption (IDE) study.
The physician-sponsored IDE study is currently ongoing at the University of Washington...
Merit Medical Systems, Inc. has announced that it has acquired substantially all of the assets of Vascular Insights, LLC, based in Quincy, USA. Vascular Insights’ primary assets are the ClariVein IC and ClariVein OC specialty infusion and occlusion catheter...
Otsuka Medical Devices, a 100% subsidiary of Otsuka Holdings Co., Ltd., and Veryan Medical have announced the completion of the acquisition of Veryan by Otsuka Medical Devices through its UK subsidiary Otsuka Medical Devices UK Ltd.
Veryan’s BioMimics 3D peripheral...
Vascular News has learned that inclusion into the SWEDEPAD studies, that are examining benefits of drug-eluting technology for peripheral arterial disease patients, has been halted.
As outlined in the public domain, the SWEDEPAD (Swedish drug-elution trial in peripheral arterial disease)...
Abiomed is to invest US$15 million in Shockwave Medical and the two companies will collaborate on a training and education programme in the USA and Germany focused on the benefits of complementary use of their respective technologies.
The press release...
The first European Society for Vascular Surgery (ESVS) guideline on treatment of abdominal aortic aneurysm (AAA), published in the European Journal of Vascular and Endovascular Surgery in 2011,1 has had a major impact on clinical practice and research. However,...
In April 2018, Chinese medical device company MicroPort Scientific acquired UK-based Lombard Medical, a developer, manufacturer and marketer of endovascular aortic aneurysm repair devices. The two companies had previously collaborated in a strategic partnership. Vascular News speaks to MicroPort’s...
In this issue:
Societies' expert consensus document highlights aortic arch treatment needs
Drug-eluting stents go head-to-head in IMPERIAL trial
Kevin Mani: Essential radiation protection for the modern vascular surgeon
Profile: Gustavo Oderich
Contego Medical has announced that the US Food and Drug Administration (FDA) has granted 510(k) clearance for its Vanguard IEP Peripheral Balloon Angioplasty System with Integrated Embolic Protection. Contego Medical is a medical device company developing and commercialising a...
New data, just published in the Journal of the American Heart Association (JAHA), suggest that there is an increased risk of death at two and five years following the use of paclitaxel-coated balloons and stents in the femoropopliteal artery....
By Thomas Zeller
The Katsanos et al meta-analysis reports an increased risk of mortality beyond one year across randomised studies of paclitaxel-coated balloons (DCB) and paclitaxel-eluting stents (DES). These observations are of great interest to the vascular specialist community. Even...
The Chicago-based Society for Vascular Surgery (SVS) has announced it will relocate its headquarters to Rosemont, USA, near Chicago’s O’Hare International Airport, as of 1 March, 2019. The SVS is an international, not-for-profit professional medical society, serving specialty-trained vascular...
Cordis has announced that the US Food and Drug Administration (FDA) has approved its INCRAFT AAA Stent Graft System for use in complex access anatomies. The INCRAFT system is an ultra-low profile and flexible endovascular aneurysm repair (EVAR) system...
Ross Milner (Chicago, USA) spoke to Vascular News at the VEITHsymposium about the importance of long-term data for aortic endografts and the GREAT (Global Registry for Endovascular Aortic Treatment) study—a 5,000 patient real-world dataset that will follow patients out to...
Stéphan Haulon, member of the CX Aortic Executive Board looks ahead to CX 2019 and raises some of the key questions that will be addressed in the programme.
The US FDA has announced plans to modernise its 510(k) clearance programme for approving medical devices for the US market. Data show that about 20% of current 510(k) devices are approved on trials that compare novel devices to predicate...
CGuard Embolic Prevention System (EPS) (InspireMD) has been granted regulatory approval by the Therapeutic Goods Administration (part of the Australian Government’s Department of Health) for commercial sale and reimbursement in Australia.
The launch of the CGuard (EPS) will commence immediately...
More than ever before, arterial hypertension represents one of the greatest health threats and challenges for both patients and treating physicians. With the new adjustment and narrowing of blood pressures targets for antihypertensive treatment, the number of patients with...
Originally from Porto Alegre in the south of Brazil, Gustavo Oderich speaks to Vascular News about his journey to the Mayo Clinic in Minnesota, his career mentors who taught him the value of both open and endovascular treatment, as well as...
Patients treated with either the Roadsaver (Terumo) or the CGuard (InspireMD) micromesh carotid stents had similar low risk of embolisation following carotid artery stenting at one Italian centre. The results were presented by Maria Antonella Ruffino (San Giovanni Battista...
Real-world outcomes in patients from the ANCHOR registry at three years show safe and effective performance of the Heli-FX EndoAnchor system (Medtronic) in complex abdominal aortic aneurysm (AAA) anatomy—particularly in hostile aortic neck anatomies.
William Jordan presented three-year data from...
The draft Global Vascular Guidelines on chronic limb threatening ischaemia (CLTI) have just been posted for open public comment until 3 December 2018. Michael S Conte (San Francisco, USA), one of the co-editors of the guidelines, told Vascular News...
Artificial intelligence (AI) is already better than humans at reading specific radiographic findings. It will impact phycisians’ jobs, but because it can currently only do very specific things, it is not yet “that robot chasing you down a dark...
Michael Jaff (Newton, USA) outlines the major recent developments in best medical therapy that will impact the treatment of peripheral arterial disease (PAD) at VEITHsymposium 2018.
Patrick Geraghty (St Louis, USA) presented new datapoints from the six-month outcomes of the Lutonix BTK IDE trial at the 2018 VEITHsymposium (13–17 November, New York, USA). In addition to the positive safety and efficacy data that were presented...
Boston Scientific has announced it has reached an agreement on the terms of a recommended offer to acquire BTG, a company headquartered in the United Kingdom, which develops and commercialises products used in minimally-invasive procedures targeting cancer and vascular...
In this issue:
Societies' expert consensus document highlights aortic arch treatment needs
Drug-eluting stents go head-to-head in IMPERIAL trial
Kevin Mani: Essential radiation protection for the modern vascular surgeon
Profile: Gustavo Oderich
Tilo Kölbel (Hamburg, Germany, and co-director of Aortic Live) tells Vascular News about the recently installed fusion imaging system from Cydar Medical, which connects live images from the operating room to the power of cloud computing. Kölbel shares his...
iVascular has announced the completion of enrolment of patients in its T.I.N.T.I.N. (Treatment with the Luminor DCB and the iVolution self-expanding stent) study.
The effectiveness of drug-coated balloons (DCB) to inhibit restenosis in symptomatic superficial femoral artery (SFA) lesions has...
The first patients in Germany have been treated with the Stealth 360 Peripheral Orbital Atherectomy System (OAS; Cardiovascular Systems Inc). The German cases represent the first commercial use of Peripheral OAS in Europe.
Tobias Achenbach, head of the Radiology Department...
Six-month outcomes of the Lutonix BTK IDE trial, a multicentre randomised controlled trial using the Lutonix 014 drug-coated balloon (DCB; BD), has demonstrated that the DCB is safe and effective to use for the treatment of narrowed or obstructed...
The first US patient has been enrolled in the Stellarex ILLUMENATE below-the-knee (BTK) investigational device exemption (IDE) study, led by principal investigators Bill Gray and Mahmood K Razavi. This global, prospective, randomised, multicentre trial is designed to assess safety...
CE mark approval has been granted for the Valiant Navion thoracic stent graft system (Medtronic) for the minimally invasive repair of all lesions of the descending thoracic aorta, including thoracic aortic aneurysms (TAA), blunt traumatic aortic injuries (BTAI), penetrating...
Stéphan Haulon (Université Paris Sud, France) tells Vascular News at Aortic Live 2018 in Essen, Germany, about a niche group of patients, i.e., those that have had prior open repair of the ascending aorta following an acute type A dissection, that are...
The International Symposium on Endovascular Therapeutics (SITE) is an annual international symposium based in Barcelona, Spain, with its focus in the Spanish vascular world. Vincent Riambau, founder and chairman of SITE, spoke to Vascular News about founding the meeting,...
The FDA have given Medtronic approval to begin a clinical trial to evaluate the Symplicity Spyral renal denervation system in patients with hypertension who are already prescribed anti-hypertension medications. A press release states that the SPYRAL HTN-ON MED trial...
Vivasure Medical has announced the European launch of the PerQseal closure device for large-bore transcatheter procedures, including transcatheter aortic valve implantation (TAVI), thoracic endovascular aneurysm repair (TEVAR), and endovascular abdominal aneurysm repair (EVAR)—all of which require large bore femoral...
Thomas Zeller (Bad Krozingen, Germany) delves into data and design of the TOBA II (Tack Optimized Balloon Angioplasty II) trial with principal investigator William Gray (Philadelphia, USA) for Vascular News. Gray, who presented the one-year findings of TOBA II...
The IN.PACT SFA trial’s co-principal investigator John Laird, from the Adventist Heart and Vascular Institute in St. Helena, USA, has presented the five-year outcomes at the Vascular Interventional Advances annual conference (VIVA; 5–8 November, Las Vegas, USA). The results...
Six-month outcomes of a multicentre randomised controlled trial using the Lutonix 014 drug-coated balloon (BD; DCB) has demonstrated the DCB is safe to use in peripheral lower limbs, below the knee. The primary safety endpoint of freedom from major...
The first-in-human data from the DEEPER trial were presented at VIVA 2018 (5–8 November, Las Vegas, USA) by John Laird, Adventist Heart & Vascular Institute team at St Helena Hospital, USA.
The purpose of this trial is to evaluate the...
A study presented at VIVA 2018 (5–8 November, Las Vegas, USA) by Maureen Kohi, associate professor of Clinical Radiology, University of California, San Francisco, USA, has found similar patency outcomes in women and men after treatment with the Stellarex...
Data from the PREVEIL early feasibility study of the SurVeil drug-coated balloon (DCB, Surmodics) were shared in a late-breaking clinical trial presentation at the Vascular Interventional Advances conference (VIVA; 5–8 November, Las Vegas, USA). PREVEIL is a prospective, US,...
VentureMed Group has announced the initiation of a new study to determine if the FLEX Dynamic Scoring Catheter reduces dissections and the need for bailout stents in peripheral arteries. The study entitled “Arterial Dissections Following FLEX Scoring and Adjunctive...
The Tack Optimized Balloon Angioplasty II (TOBA II) clinical trial has successfully achieved both primary and secondary endpoints. One year results from the TOBA II study were presented in the late-breaking scientific session by William Gray, system chief, Division...
Results from an aggregated data analysis of the use of the Zilver PTX drug-eluting peripheral stent (DES; Cook Medical) in challenging patient populations concluded that risk factors such as Rutherford classification, sex, calcification and diabetes did not have a...
The five-year and final results from the pivotal IN.PACT SFA Trial and one-year all-subjects results from the Total IN.PACT Pooled Analysis of the IN.PACT Admiral drug-coated balloon in patients with peripheral arterial disease (PAD) in the superficial femoral (SFA)...
Clinical outcomes from the IMPERIAL Long Lesion Sub-study were presented at VIVA (5–8 November, Las Vegas, USA) demonstrating that the Eluvia drug-eluting vascular stent system (Boston Scientific) is safe and effective in treating patients with long, complex, calcified lesions...
The Surfacer Inside-Out Access Catheter System (Bluegrass Vascular) has demonstrated positive commercial use, consistently achieving central venous access in patients with upper body occlusions. A study presented in a poster session at the 2018 American Society of Nephrology (ASN)...
The incidence of ruptured abdominal aortic aneurysm (AAA) and associated mortality has decreased over the past 20 years in Sweden, indicating that altered smoking patterns have had an impact on overall AAA prevalence and aneurysm rupture and that endovascular aneurysm...
The results of a study seeking to compare access site complications in 4F and 6F devices for treatment of peripheral arterial disease (PAD) report that ambulatory treatment presents a valid option for endovascular procedures in lower limb arteries, with...
Humacyte has announced the initiation of a US phase II vascular trauma clinical trial of Humacyl, its investigational human acellular vessel, for vascular replacement or reconstruction in patients with life- or limb-threatening vascular trauma.
The phase II vascular trauma study...
“The best treatment for a 60-year-old man with an infrarenal abdominal aortic aneurysm that is above the threshold for intervention is arguably surgery based on the durability of this intervention. However, operating at this age (where open surgery is...
Endologix has announced that it has commenced a registered underwritten public offering of US$20 million of its shares of common stock. In addition, Endologix has granted the underwriter a 30-day option to purchase up to an additional US$3 million...
Terumo Aortic has announced the acquisition of Serom Medical Technology S.r.l. by Vascutek Ltd and the subsequent merger with Bolton Medical S.r.l. in Italy.
Serom Medical Technology S.r.l. has successfully represented Vascutek in Italy for more than 30 years and...
Rachel Bell (Guy’s and St Thomas’ Hospital, London, UK) describes a variety of specialist surgical options to treat infection after endovascular aneurysm repair (EVAR), a relatively rare occurrence that can have devastating complications. She stresses the importance of dealing...
Ronald L Dalman (Stanford, USA), vice president of the Society for Vascular Surgery, discusses how aligning an individual surgeon’s goals with those of the institution at which he/she is sited is important to counter the “epidemic” of burnout. Dalman...
The US Food and Drug Administration (FDA) has approved Valiant Navion thoracic stent graft system (Medtronic) for the minimally invasive repair of all lesions of the descending thoracic aorta, including thoracic aortic aneurysms, blunt thoracic aortic injuries, penetrating atherosclerotic...
Avinger, a developer of treatments for peripheral arterial disease (PAD), has announced Conformité Européenne (CE) Marking approval of Pantheris SV (Small Vessel), a product line extension of the Lumivascular atherectomy system. CE Marking allows for distribution of Pantheris SV...
Treating severely calcified arteries by endovascular means is still a challenge, although atherectomy, stents and lithotripsy are all techniques that are employed. Lithotripsy (also called Lithoplasty) seeks to microfragment the calcium seen in the vessels of peripheral arterial disease...
Andrew Klein (a consultant anaesthetist from Royal Papworth hospital in Cambridge, UK) tells Vascular News that research in cardiac surgery has shown that while the surgeon has a small, but important, effect on a patient’s outcome after surgery, it is the...
Chronic exposure to low dose radiation, such as that experienced by operators who routinely perform endovascular aneurysm repair (EVAR), has biological effects that can be mitigated with the use of lead shielding for the legs. However, many vascular and...
“The instructions for use (IFU) are not a clinical guideline, so trainees should really think about what the main considerations are when they are implanting devices in the aorta to create a durable repair,” said Tara Mastracci (Royal Free...
As her term as the first female president of the European Society for Vascular Surgery comes to end, Alison Halliday reflects on what she has achieved and the future of society. She also speaks to Vascular News about her...
Rivaroxaban, brand name Xarelto (Janssen), has received approval by the US Food and Drug Administration (FDA) to reduce the risk of major cardiovascular (CV) events, such as CV death, myocardial infarction and stroke, in people with chronic coronary or...
Concept Medical Inc. has approached the FDA for an investigational device exemption (IDE) for their sirolimus-coated balloon (DCB). To support this process, they have raised US$60 million (for an undisclosed valuation) from cardiologist and serial entrepreneur Kiran Patel (Tampa, USA). An IDE...
Cagent Vascular, a developer of next-generation angioplasty balloons using proprietary serration technology, announces Brian Walsh to serve as chairman of the Board.
Brian Walsh has held several executive level positions in cardiovascular and ophthalmology specialties within the medical device...
This article is an advertorial by Medtronic.
In collaboration with leading clinicians, researchers and scientists worldwide, Medtronic Inc. offers the broadest range of innovative medical technology for the interventional and surgical treatment of cardiovascular disease. One of these innovations, the...
Eximo Medical has announced it has received 510(k) clearance from the US Food & Drug Administration (FDA) for its B-Laser atherectomy system for peripheral arterial disease (PAD). B-Laser is a transformative 355nm wavelength laser technology designed to address unmet...
Intact Vascular has announced that data from its Tack Optimized Balloon Angioplasty (TOBA) II pivotal study will be presented as a late-breaking clinical trial at the 15th annual VIVA conference in Las Vegas, USA, on 5–8 November.
The TOBA II...
Humacyte has announced the appointment of Douglas L Blankenship as chief financial officer, effective Monday, 8 October, 2018.
According to a company release, Blankenship is a highly accomplished financial professional with executive-level experience in financing, finance and accounting, technical operations,...
UK-based Veryan Medical has announced that the company has received premarket approval (PMA) for their BioMimics 3D vascular stent system from the US Food & Drug Administration (FDA). The device is approved for the treatment of symptomatic de novo...
Endologix, a developer and marketer of treatments for aortic disorders, has announced that the US Food and Drug Administration (FDA) has classified a voluntary recall action that Endologix took in July of this year, as a Class I recall....
Teleflex Incorporated has announced that it has acquired Essential Medical, a privately-held medical device company that has developed and commercialised the CE Marked Manta vascular closure device specifically designed for closure of large bore arteriotomies following procedures utilising devices...
Jotec, a fully owned subsidiary of CryoLife, has announced 30-day results of the PLIANT study, which was an observational, prospective, non-randomised, multicentre, international study conducted in 12 hospitals throughout Europe. This study enrolled 45 patients who were due to...
Five-year results from a Japanese post-market surveillance study aimed at evaluating the Zilver PTX drug-eluting stent (DES) in a real-world population show consistently positive outcomes. The findings were presented for the first time at the Cardiovascular and Interventional Radiological...
Twelve-month data prove that intravascular lithotripsy; a novel approach using pulsatile sonic pressure waves to modify intimal and medium calcium in stenotic peripheral arteries, is a safe and effective treatment option. This is the result of the Disrupt PAD...
Tobias Steinke (Dusseldorf, Germany) reported on the Lutonix Global arteriovenous (AV) Registry at CIRSE 2018 in Lisbon, Portugal. Lutonix Global AV Registry data add to real-world outcomes of using Lutonix DCB in AV access.
Straight after the presentation, Steinke tells Vascular News about...
Data from a study seeking to determine the impact of severe calcification and 12-month outcomes of femoropopliteal disease treatment using the Stellarex drug-coated balloon (DCB; Phillips) were presented by Fabrizio Fanelli, Careggi University Hospital, Florence, Italy, at CIRSE (Cardiovascular...
LeMaitre Vascular has acquired the vascular clot management business of Applied Medical Resources Corporation for US$14.2 million, of which US$11 million was paid at closing and US$3.2 million will be paid in two post-closing instalments. As part of the...
Shortages in the US vascular surgery workforce was the focus of this year’s Stanley Crawford Critical Issues Forum at the Society for Vascular Surgery’s (SVS) Vascular Annual Meeting (VAM; 20–23 June, Boston, USA). The session highlights a problem faced...
The findings of a new retrospective review carried out by Suh Min Kim (Dongguk University Ilsan Hospital, Goyang, Korea) and colleagues that aimed to establish an appropriate treatment strategy for arteriovenous graft (AVG) infection have been published in the...
In this issue:
Recruitment for vascular disease management struggles to keep up with expected demands on the workforce
Post-atherectomy IVUS better than angiography for dissection detection
Profile: Alison Halliday
New section: Vascular Access
In this issue:
Recruitment for vascular disease management struggles to keep up with expected demands on the workforce
Post-atherectomy IVUS better than angiography for dissection detection
Profile: Alison Halliday
New section: Vascular Access
In this issue:
– Arterial dissection after balloon angioplasty: an underestimated cause of excessive stenting and patency loss
– Leave nothing behind in the femoropopliteal segment: is vessel preparation followed by drug-coated balloon the ultimate tool?
– Is POBA dead? New concepts...
In this supplement:
– Application of iliac branch device for preserving the internal iliac artery in patients with aortoiliac aneurysmatic disease
– Treatment of aortoiliac aneurysms: Case report from Watford General Hospital
– Do the old open repair rules no longer apply?
Flow Forward Medical, a medical device company focused on improving outcomes for haemodialysis patients through the rapid creation of high-quality vascular access sites, has announced it has raised an additional US$1.2 million in Series A financing from a group...
Cook Medical have announced that a new 5mm diameter version of Zilver PTX was approved by the US Food and Drug Administration (FDA). It is the first 5mm drug-eluting stent in the USA with lengths available up to 140mm...
Medical technology company Vascular Simulations has released Replicator PRO, a realistic vascular replication system, at the 2018 Transcatheter Cardiovascular Therapeutics meeting (TCT; 21–25 September, San Diego, USA).
Aiming to be a better alternative to device reliance animal testing, Replicator...
Boston Scientific has announced that the US Food and Drug Administration (FDA) has approved its Premarket Approval (PMA) application to market the Eluvia Drug-Eluting Vascular Stent System, specifically developed for the treatment of peripheral arterial disease (PAD). The stent...
The largest prospective, independently-adjudicated study of drug coated balloons (DCBs) in a broad range of lesions in real-world patients confirms the safety and performance of the IN.PACT Admiral DCB (Medtronic) in the treatment of femoropopliteal artery disease. This is...
Andrew Holden (Auckland, News Zealand) interviews Stefan Mueller-Huelsbeck (Flensburg, Germany) to distil the key findings of the IMPERIAL trial one-year data that were presented for the very first time at CIRSE 2018 in Lisbon.
In the randomised controlled trial, Eluvia,...
The Eluvia drug-eluting vascular stent system (Boston Scientific) shows superior primary patency compared to the Zilver PTX drug-eluting stent (Cook Medical), concludes the 12-month follow-up of the IMPERIAL randomised controlled trial. These data were presented for the first time...
Cardiovascular Systems Inc. (CSI), a medical device company developing and commercialising interventional treatment systems for patients with peripheral and coronary artery disease, has announced that the first patient in Hong Kong has been treated with its Stealth 360 Peripheral...
Gore has announced the first European patient implant of the Gore Excluder Conformable AAA Endoprosthesis with Active Control System. This next-generation endovascular aneurysm repair (EVAR) device is indicated to treat the broadest range of abdominal aortic aneurysms (AAA) in...
Guidelines of the European Society for Vascular Surgery1 give an overview of the current best evidence for vascular access for dialysis. Although these guidelines are largely similar to their predecessors, Nicholas Inston argues they may not always lead to...
Patients with peripheral arterial disease (PAD) are found to have an increased risk for high opioid use. Nathan Itoga and colleagues from the University of Stanford (Stanford, USA), presented the findings of a recent study analysing data from the...
The first patient has been enrolled in the SAVAL pivotal trial to evaluate clinical outcomes of the SAVAL below-the-knee (BTK) drug-eluting stent system (Boston Scientific) in patients with critical limb ischaemia (CLI). The SAVAL BTK stent system is the...
BTG has announced it has acquired Novate Medical, a medical device company focused on the prevention of pulmonary embolism in patients at high risk of venous thromboembolic events.
Novate has developed Sentry, the first bioconvertible inferior vena cava filter, which...
Vascular Dynamics has revealed that enrolment of the first patients in an FDA-approved and best-in-class pivotal clinical trial has started. The CALM-2 study is designed to establish safety and efficacy of the novel endovascular baroreceptor amplification (EVBA) procedure using...
Avinger, a developer of treatments for peripheral artery disease (PAD), has announced the Company submitted a new 510(k) application to the US Food & Drug Administration (FDA) for the Pantheris SV (Small Vessel) Lumivascular Atherectomy System.
Pantheris SV is a...
Tilo Kölbel (University Heart Center Hamburg, Germany) talks about the findings of the STEP collaboration, and why endovascular procedure operators perhaps are "not the best people to examine neurological outcome" in their patients.
STEP is a multidisciplinary initiative which aims...
Real-world outcomes from the Transcarotid Artery Revascularisation (TCAR) Surveillance Project (TSP) were presented by Mahmoud Malas from John Hopkins Hospital (Baltimore, USA) at the Society for Vascular Surgery Vascular Annual Meeting (VAM; 20–23 June, Boston, USA). At baseline, patients...
The opening of an outpatient wound centre has been associated with a significant increase in peripheral vascular practice and a significant decrease in amputation rate. Venita Chandra and colleagues Alyssa M Flores, Matthew W Mell and Ronald L Dalman...
's's sAmarin Corporation, a biopharmaceutical company focused on the commercialisation and development of therapeutics to improve cardiovascular health, supported the recent presentation of a scientific poster at the European Society of Cardiology (ESC) Congress in Munich, Germany. This analysis...
Temple University’s systematic review and meta-analysis on eight independent intermittent claudication studies using pneumatic compression pumps has shown significant improvements in walking distances even after treatment was withdrawn. Paul van Bemmelen, of Temple University, Philadelphia, USA, conducted the meta-analysis....
Gore have announced the first implant in conjunction with the Gore ARISE study of the Gore Ascending Stent Graft, an investigational device and the only endovascular stent graft specifically designed to treat type A dissections of the ascending aorta....
Prevencio has announced data demonstrating its HART PAD test accurately diagnoses peripheral arterial disease (PAD) in diabetes mellitus patients, a patient population in which PAD prevalence has traditionally been difficult to assess. Researchers believe these important findings, presented at the...
Gore has announced FDA 510(k) clearance, approval from the Japanese Ministry of Health, Labour, and Welfare, and receipt of CE mark for the innovative Gore moulding and occlusion balloon, a compliant polyurethane balloon catheter designed in close collaboration with...
Xarelto, co-administered with aspirin, is indicated in the EU for the prevention of atherothrombotic events in adult patients with coronary artery disease (CAD) or symptomatic peripheral arterial disease (PAD) at high risk of ischaemic events.
The European Commission (EC) has...
AngioDynamics has provided a corporate update and acknowledged a recent, favourable US District Court ruling that impacts its vascular access business.
A press release states that in May of 2017, AngioDynamics filed a lawsuit against CR Bard (Bard) for violating...
Endologix has announced that it has appointed Jeffrey S Brown as the company’s chief operations officer, effective as of 13 August, 2018.
John Onopchenko, chief executive officer of Endologix, says, “I am very excited to have someone with Jeff’s credentials...
Medtronic plc has announced the full commercial launch of the IN.PACT Admiral drug-coated balloon (DCB) in Japan. The launch follows the completion of a post-market clinical trial, which enrolled 300 patients.
IN.PACT Admiral received approval last year from the Japanese...
Two-year outcomes from the LIBERTY 360° study have shown a marked improvement across all Rutherford classes, as well as high freedom from major amputation. The results were presented as a late-breaking presentation at the Amputation Prevention Symposium (AMP; 8–11...
Researchers at Okayama University report in The Journal of Vascular Access the use of a supporting device for accurately placing haemodialysis catheters in dialysis patients. The device was successfully used in a group of 10 patients and, according to...
NuCryo Vascular has signed a commercialisation agreement with Lokai Medical, a specialty distributor of coronary/peripheral and interventional devices, to distribute the PolarCath balloon dilatation system in the USA.
Balloon cryoplasty is one of the options for treating peripheral arterial disease (PAD)....
Cook Medical has introduced the 140mm-length Zilver PTX drug-eluting peripheral stent in both 6 and 7mm diameters in the USA. The longer length comes after an expanded indication approval by FDA to treat total lesion lengths up to 300mm...
Boston Scientific has announced that it has recently closed its acquisition of Claret Medical, which developed and commercialised the Sentinel cerebral embolic protection system—the only device cleared in the USA and Europe to protect patients against the risk of...
Modulated Imaging, a company developing optical imaging solutions powered by spatial frequency domain imaging (SFDI) for noninvasive assessment of tissue health, announced today that it has received FDA 510(k) clearance for its new medical device, the Clarifi imaging system.
Clarifi...
Recent studies have shown the advantages of 3D printing for simulation of vascular surgery includes significant reduction of procedural time, but the applications of producing life-sized 3D printed models can have important educational benefits beyond the operation planning stage.
Bernhard...
A study published recently in the Journal of Vascular Access has indicated that the use of arteriovenous thigh graft access is viable as an alternative for patients who have exhausted all other access options.
Denise Kim (Toronto General Hospital, University...
Sean Lyden of the Cleveland Clinic (Cleveland, USA) discusses Endologix' Ovation grafts and the use endovascular aneurysm repair (EVAR) in short neck and complex anatomies.
Reva Medical, a company developing bioresorbable polymer technologies for vascular applications, has announced that its Motiv bioresorbable scaffold is the first drug-eluting bioresorbable scaffold to receive CE mark approval for treatment of below-the-knee peripheral artery disease.
Late last year,...
B. Braun has announced the recent French reimbursement approval for its peripheral drug-coated balloon (DCB) SeQuent Please OTW effective as of July 2018.
As published in the Official Journal of the French Republic, B. Braun's over-the-wire DCB is now reimbursed...
Laminate Medical Technologies (Laminate), a privately-held start-up developing the VasQ implanted blood vessel external support device for patients requiring arteriovenous (AV) fistula as vascular access for haemodialysis, have announced the first forearm fistula cases in Germany.
The first surgeries were performed...
Laminate Medical Technologies (Laminate), a privately-held start-up developing the VasQ implanted blood vessel external support device for patients requiring arteriovenous (AV) fistula as vascular access for haemodialysis, have announced the first forearm fistula cases in Germany.
The first surgeries were performed...
Cardiovascular Systems, Inc. (CSI), a medical device company developing and commercialising innovative interventional treatment systems for patients with peripheral and coronary artery disease, today announced that it has signed an agreement with Aerolase Corp. to co-develop a new laser...
Laminate Medical Technologies (Laminate), a privately-held start-up developing the VasQ implanted blood vessel external support device for patients requiring arteriovenous (AV) fistula as vascular access for haemodialysis, have announced the first forearm fistula cases in Germany.
The first surgeries were performed...
Five-year follow-up data from STABLE I, a global feasibility study of the Zenith Dissection Endovascular stent (Cook Medical), were presented by Joseph Lombardi (Cooper University Health Care, Camden, USA) at the Society for Vascular Surgery’s Vascular Annual Meeting (SVS...
Ra Medical Systems has filed a registration statement on Form S-1 with the US Securities and Exchange Commission relating to a proposed initial public offering of shares of its common stock. The number of shares to be offered and...
Boston Scientific has signed an agreement to acquire Claret Medical, which developed and commercialised the Sentinel cerebral embolic protection system. The device is used to protect the brain during certain interventional procedures, predominately in patients undergoing transcatheter aortic valve...
In this issue:
NICE draft guideline casts shadow over EVAR for unruptured aneurysms
Europe braces for harder times in medical device innovation while US FDA eases regulations
Profile: Tilo Kölbel
In this issue:
NICE draft guideline casts shadow over EVAR for unruptured aneurysms
Europe braces for harder times in medical device innovation while US FDA eases regulations
Profile: Tilo Kölbel
Cardiovascular Systems, Inc. (CSI) has signed an exclusive international distribution agreement with OrbusNeich to sell its coronary and peripheral orbital atherectomy systems (OAS) outside the USA and Japan. OrbusNeich manufactures and sells an extensive portfolio of coronary and peripheral...
Cagent Vascular has announced the completion of US$11.87 million in Series B funding. The round was led by two strategic investors including one that participated in the Series A financing. Additional participants included Balestier Investments, Ben Franklin Technology Partners1,...
The iDissection Classification trial results published in the Journal of Invasive Cardiology have shown that with the use of intravascular ultrasound (IVUS) investigators were able to identify more dissections following percutaneous transluminal angioplasty than with angiography.
Post-percutaneous transluminal angioplasty (PTA)...
EchoNous Vein (EchoNous), an ultrasound-based tool designed specifically for nurses to improve peripheral IV catheter placements, has received US Food and Drug Administration (FDA) approval.
A company release explains that EchoNous Vein provides immediate, clear images at depths from 1–5cm...
Otsuka Holdings, Otsuka Medical Devices, and ReCor Medical have signed a merger agreement pursuant to which Otsuka Holdings will acquire the remaining shares in ReCor from its current holders. The transaction is subject to approval by ReCor’s shareholders.
Otsuka Holdings’...
Merit Medical Systems has been named the exclusive global distributor of the Q50 Plus Stent Graft Balloon Catheter (QXMédical). The Q50 Plus is used in abdominal and thoracic endovascular aneurysm repair (EVAR) procedures to repair abdominal aortic aneurysms (AAA)...
BD (Becton, Dickinson and Company) announced it has completed the acquisition of TVA Medical, a company that develops minimally invasive vascular access solutions for patients with chronic kidney disease requiring haemodialysis including the everlinQ endoAVF System.
The addition of TVA...
BD (Becton, Dickinson and Company) announced it has completed the acquisition of TVA Medical, a company that develops minimally invasive vascular access solutions for patients with chronic kidney disease requiring haemodialysis including the everlinQ endoAVF System.
The addition of TVA...
Born into a family of medical professionals, Tilo Kölbel was destined to become one himself. He tells Vascular News about his journey to becoming a vascular surgeon, the proudest moments in his career and the research that he is...
A new surgical navigation system, Intra-Operative Positioning System (IOPS) that is being developed by Centerline Biomedical along with Cleveland Clinic in the USA could help to do away with concerns around operator exposure to radiation during endovascular procedures.
The concept...
The Society for Vascular Surgery (SVS) has introduced a new award, the International Lifetime Achievement Award, which recognises “outstanding contribution to vascular surgery” and global acclaim. Enrico Ascher (NYU Langone Health, New York, USA), who describes it as “the...
The first patient has been implanted with the Gore TAG Conformable Thoracic Stent Graft with Active Control System after being included on the Australian Register of Therapeutic Goods last month in Australia.
The first implants were performed by Professor Ian...
The RADIANCE-II study of the Paradise Ultrasound Denervation System (ReCor Medical) for the treatment of hypertension has been approved by the US Food & Drug Administration (FDA).
Building upon the recent positive results of the RADIANCE-HTN SOLO study, RADIANCE-II will...
Results from the DETOUR I trial evaluating the DETOUR System (PQ Bypass) for percutaneous bypass showed promising 12-month durability for patients with extremely long blockages in the superficial femoral artery (SFA). The data were presented as a late-breaking clinical...
An update to last year’s FDA letter, expressing concern over risk of type III endoleaks in relation to endovascular graft systems, now states that the increased risk is specific to one device which was discontinued in 2014.
Endologix’ AFX Strata,...
One-year results from the LUCY (Evaluation of females who are underrepresented candidates for abdominal aortic aneurysm repair) registry were reported at the Society for Vascular Surgery Annual Meeting (SVS; 20–23 June, Boston, USA). The LUCY study is the first...
The 2000th E-liac stent graft (JOTEC) in the Netherlands was implanted successfully on the 20 June, JOTEC, a fully owned subsidiary of CryoLife has announced. The endovascular implantation was performed at Zuyderland Hospital in Heerlen, Netherlands, by the vascular...
The annual census of UK consultants and higher speciality trainees—Focus on Physicians 2017–18—indicates that more than half of all consultants and two thirds of trainees reported frequent gaps in trainees’ rotas, with one in five respondents saying these are...
Ellipsys Vascular Access System (Avenu Medical) has been granted De Novo marketing authorisation from the Food and Drug Administration (FDA). Ellipsys is a minimally-invasive catheter-based system designed for end-stage renal disease (ESRD) patients requiring haemodialysis.
“Good vascular access makes a...
iose
A new study, published in Circulation: Cardiovascular Interventions, shows that one fifth of patients undergoing transcatheter aortic valve implantation (TAVI) have evidence of carotid artery disease. It also indicates that while the presence of carotid disease may be a...
Results from the SAFE MANTA IDE clinical trial—the first pivotal trial for a dedicated large bore closure device (Manta, Essential Medical)—show haemostasis was achieved in less than one minute in 86% of the primary analysis cohort, and in less...
everlinQ endoAVF System (TVA Medical) is now approved by the US Food and Drug Administration for de novo marketing. This innovative, minimally invasive technology uses magnetic catheters and radiofrequency energy to create an arteriovenous fistula (AVF) for haemodialysis access...
The UK’s National Institute for Health and Care Excellence (NICE) has issued draft guidance on abdominal aortic aneurysm (AAA) diagnosis and management. The most notable recommendation within the guideline is related to repairing unruptured aneurysms where the guideline states...
Major adverse events in patients undergoing transcarotid artery revascularisation (TCAR) were similar to those receiving a carotid endarterectomy, a study has found, despite the higher medical risk in the former group. Marc Schermerhorn (Beth Israel Deaconess Medical Center, Boston,...
Terumo has announced that it acquired CE mark for the Kanshas drug-coated balloon catheter used in the treatment of lower extremity peripheral arterial disease.
With a September launch planned in Europe, Terumo is expected to be the first Japanese...
Veryan Medical has announced completion of enrolment of the 500th patient in the MIMICS-3D pan-European registry, a prospective, multicentre, observational registry to evaluate the BioMimics 3D Self-Expanding Stent System in the treatment of femoropopliteal artery disease.
The Registry is evaluating...
Humacyte, an innovator in biotechnology and regenerative medicine, has announced that Jeffrey Lawson has been appointed president and chief executive officer. The former chairman and chief executive officer, Carrie S Cox, has assumed the role of executive chairman.
Lawson, an...
George Chrysant gives an overview of combined revascularisation interventions for coronary and carotid disease, arguing that "carotid artery stenting and coronary artery bypass grafting as a combination appears to be significantly safer and associated with better outcomes."
Atherosclerosis affects multiple...
Medtronic has announced that it has received US FDA approval for 200mm and 250mm lengths of the IN.PACT Admiral drug-coated balloon (DCB) to treat long superficial femoral artery lesions in patients with peripheral artery disease.
"As superficial femoral artery disease...
The US Food and Drug Administration (FDA) Circulatory System Devices Panel of the Medical Devices Advisory Committee has provided a favourable recommendation on the premarket approval application for INCRAFT AAA Stent Graft System (Cardinal Health). The panel voted 11...
Fresenius Medical Care, the world's largest provider of dialysis products and services, and Humacyte, Inc., a medical research, discovery and development company, have announced a strategic, global partnership and a US$150 million equity investment. This agreement has the potential to...
In this supplement:
Assessing the needs of peripheral arterial disease and critical limb ischaemia
The role of Philips solutions in solving needs: Highlighting tools and devices
Perfusion angiography with the 2D Perfusion software
Patient cases
Read or download the educational...
In this article, Bijan Modarai outlines recent research on the effects of radiation exposure in high-volume interventionalists, why a future with alternative modalities to X-ray guided procedures is an exciting prospect, and what safety measures could make a difference...
A new study suggests that the familial risk of abdominal aortic aneurysm is much higher than previously thought and this excess risk is highest both in female relatives and in relatives (of any sex) of female patients. According to...
Countries in the European Union have long been the first to receive new innovations in medical technology, as the EU’s Medical Device Directive (MDD) provided quicker routes to implementation of new devices than its equivalent in the USA, the...
MicroStent (Micro Medical Solutions), a vascular stent designed to reduce below-the-knee amputations caused by critical limb ischaemia (CLI) resulting from peripheral arterial disease (PAD) has been implanted for the first time in the USA. The company received approval from...
Sanford Health and Freudenberg Medical, a global developer and manufacturer for the medical device industry, have entered into a joint research and development agreement to create a device to treat blood clots and blockages within blood vessels.
According to a...
A recent healthcare analysis shows that a novel, catheter-based endovascular system for creating haemodialysis access offers quality-of-life benefits and cost savings compared to the traditional surgical approach in end-stage renal disease patients. The retrospective analysis comparing the everlinQ endoAVF System...
Piotr Musiałek, from the Department of Cardiac and Vascular Diseases, John Paul II Hospital, Kraków, Poland, presented the expanded 24-month follow-up results from the PARADIGM-Extend Clinical Study utilising CGuard EPS (InspireMD) at EuroPCR 2018 (22–25 May, Paris, France).
PARADIGM-Extend is...
Avinger has announced that the company received 510(k) clearance from the US Food & Drug Administration (FDA) for its next generation Pantheris Lumivascular atherectomy system, the first-ever image-guided atherectomy device for the treatment of peripheral arterial disease (PAD). Lumivascular...
Medtronic has announced first-ever-data from the SPYRAL HTN-ON MED Study. Initial study results found statistically significant and clinically important blood pressure reductions in hypertensive patients prescribed anti-hypertension medications treated with the Symplicity Spyral renal denervation (RDN) system with no...
Institut Jantung Negara (IJN; Kuala Lumpur, Malaysia) has become the first hospital in Asia to perform peripheral vascular intervention (PVI) using VascuFlex Multi-LOC (B Braun), a multiple stent delivery system designed for the treatment of peripheral arterial disease (PAD).
The procedure...
Med Alliance SA, a company developing and commercialising the first sirolimus micro-reservoir drug-coated balloon (SELUTION DCB) to treat patients suffering from peripheral artery disease (PAD), coronary artery disease (CAD), arteriovenous fistulas (AVF) and grafts (AVG) for end-stage renal disease,...
Marc Schermerhorn (Boston, USA) discusses the three-year results of an analysis on the long-term survival of Nellix EVAS system (Endologix) IDE trial patients vs. a cohort of EVAR patients. The study found that EVAS is associated with higher long-term...
Surmodics, a provider of medical device and in vitro diagnostic technologies to the healthcare industry, announced it has reached an agreement with Embolitech to acquire an innovative thrombectomy platform technology and related intellectual property with broad potential peripheral vascular...
Endospan, a company developing off-the-shelf endovascular repair of Aortic Arch Disease including aneurysms and dissections, has announced that it has elected Jeff Elkins to its board of directors. Elkins has been president and CEO of Veniti since July 2015.
Previously, Elkins was COO and...
The first 24-month data from the LUTONIX AV investigational device exemption trial were presented at Charing Cross yesterday. These interim data show a sustained effectiveness benefit for the Lutonix drug-coated balloon (BD) and indicate that it leads to an...
oowth
At the Charing Cross Symposium (CX; 24–27 April, London, UK) delegates heard compelling research-in-progress from a multidisciplinary team of surgeons, statisticians and health economists that the initial aortic diameter plus annual secondary sac diameter, as measured by ultrasound close...
Laerdal Medical has announced the launch of their new simulation training technology, SimMan Vascular. The company has partnered with endovascular simulator developer Mentice to produce the SimMan Vascular system, which was introduced at the Simulation User Network conference (SUN;...
The BATTLE trial comparing a drug-eluting stent (Zilver PTX, Cook Medical) vs. a bare metal stent (Misago, Terumo) for the treatment of intermediate femoropopliteal lesions has failed to the show superiority of the paclitaxel-coated stent at one-year follow-up. The...
In an exciting session at the Charing Cross Symposium (CX; 24-27 April, London, UK), the audience witnessed the demonstration of a novel treatment strategy with intravascular lithotripsy for calcified arterial stenosis. It will now become a CX tradition to...
Stroke is a major concern following TEVAR (thoracic endovascular aortic repair) and Charing Cross delegates heard the results from a collaborative study that pooled practice data from a number of high-volume centres in order to benefit patients undergoing the...
The first two patients have been successfully enrolled in the RelayBranch Early Feasibility Study. This trial will assess the safety and efficacy of the RelayBranch Thoracic Stent-Graft system (Terumo Aortic) in patients with thoracic aortic pathologies requiring treatment proximal...
Ulf Teichgräber, Jena, Germany, discusses the EffPAC trial 12-month results which were presented for the first time at CX 2018
Terumo Aortic have announced the European limited market release of the RelayPro thoracic stent graft system at the 2018 Charing Cross Symposium (CX; 24–27 April, London, UK).
RelayPro is a low profile, next generation device designed to expand the...
Medtronic has announced that it has received US Food and Drug Administration (FDA) approval for the IN.PACT Admiral drug-coated balloon (DCB) to treat long superficial femoral artery (SFA) lesions up to 360mm in patients with peripheral artery disease (PAD).
Approval was...
Vascutek and Bolton Medical, subsidiaries of Terumo Corporation of Japan, have combined into Terumo Aortic. Combining the aortic companies into a single business, Terumo looks to grow its presence in the global aortic and vascular implants market.
The new company...
LimFlow, a developer of peripheral endovascular technology focused on the treatment of critical limb ischemia (CLI), has announced it has secured €27 million (US$33.5 million) in an oversubscribed Series C financing.
Proceeds from the Series C financing will fund the...
Colin Bicknell (Imperial College, London, UK) speaks to Vascular News about his experience of using EndoAnchors in thoracic endovascular aortic repair (TEVAR), sharing his tips and techniques.
Intact Vascular, a developer of medical devices for minimally invasive peripheral vascular procedures, has announced that it closed a Series C financing totaling US$20 million. This financing is designed to fund the company through premarket approval (PMA) of the Tack...
Interrad Medical has announced the selection of their SecurAcath Subcutaneous Catheter Securement Device for the Innovation and Technology Payment programme by the UK National Health Service (NHS) England.
The Innovation and Technology Payment (ITP) programme is designed to remove significant barriers to...
Abbott has gained FDA approval for its Perclose Proglide suture-mediated closure system. The device is designed to close access sites created by large bore catheters, stopping the blood flow.
The ProGlide is used after the insertion of large bore catheters...
This article is an advertorial by Veryan Medical.
Authors: PA Gaines, T Sullivan, G Ansel, C Caro
Introduction
Medical advances in the treatment of femoropopliteal artery disease over the last decade reflect the on-going fight to maintain patency; and that battle is...
Cook Medical has announced that physicians in the US and Canada are once again able to order the company's Beacon Tip Torcon NB Advantage 5F catheter, used in angiographic procedures. In 2015 and 2016, Cook recalled all Beacon Tip...
SoundBite Medical Solutions has announced it has received CE marking for the SoundBite Crossing System, a novel first-in-class wire-based technology that is intended for the treatment or peripheral chronic total occlusions (CTOs) in a simpler and clinically efficient method.
“We...
The handheld medical imaging system HyperView (HyperMed Imaging) has received CE mark, allowing for distribution in Europe. The system is designed to detect perfusion of superficial tissue in support of wound care.
The HyperView System is a fast, handheld, battery...
Drug-coated balloon (DCB) angioplasty devices have been categorised by the US Centers for Medicare and Medicaid Services (CMS) into the same billing code as plain balloon angioplasty, following the expiration of the technology’s transitional pass-through and new technology add-on...
Maria Antonella Ruffino spoke to Vascular News at LINC 2018 about the differences between mesh-covered stents and conventional devices in treatment of carotid artery disease, as well as the differences between currently available mesh-covered stents.
The TOBA BTK (Tack Optimized Balloon Angioplasty Below the Knee) clinical trial results have recently been published in Catheterization and Cardiovascular Intervention. The multicentre pilot study focused on collecting data supporting the safety and performance of the Tack Endovascular...
The first results have been announced from ENCORE, a pooled, global analysis of several prospective clinical trials and registries studying polymer endovascular aneurysm repair (Polymer EVAR) using Ovation Abdominal Stent Graft Systems (Endologix).
ENCORE is a pooled, retrospective analysis of...
The 3,000th E-xtra Design Engineering vascular implant (Jotec/CryoLife) was recently inserted successfully. The endovascular implantation was performed at Cologne University’s academic teaching hospital, Porz am Rhein hospital, Cologne, Germany, by senior consultant Thomas May and senior physician Dr Guido...
Results and data on vascular embolic device GPX (Fluidx Medical Technology), a new proprietary in situ setting embolic agent that combines the benefits of coils, gel-beads, and other embolics, were presented at the 2018 Society of Interventional Radiology conference (17-22 March, Los...
The Horizon stent graft system, developed to treat abdominal aortic aneurysms, has received CE mark. The stent graft system is produced by Endospan, a company specialising in off-the-shelf endovascular aortic repair (EVAR).
“Horizon is a unique platform that can be used...
The first patients have been enrolled in a prospective, multicentre, non-randomised study intended to evaluate the ability of the Reflow Wingman Catheter (Reflow Medical) to cross chronic total occlusions (CTOs) in peripheral lesions.
Initial patient enrolments were performed by Nilesh...
Data presented at American College of Cardiology 2018 Scientific Sessions (ACC; 10–12 March, Orlando, USA) indicate that a novel multi-protein, artificial intelligence (AI)-driven test accurately diagnoses aortic stenosis—a condition with a 50% mortality-rate when left untreated. The HART AS...
Highlights:
CX celebrates 40 Years of Looking Forward
CX peripheral arterial controversies
The future of EVAR and abdominal aortic controversies
CX Aortic Workshop
CX Venous Controversies
CX Innovation Showcase programme highlights
Summary of Vascular News profiles 2017/18
https://vascularnews.com/wp-content/uploads/sites/7/2018/03/VN-CX18_lowres_US.pdf
Highlights:
CX celebrates 40 Years of Looking Forward
CX peripheral arterial controversies
The future of EVAR and abdominal aortic controversies
CX Aortic Workshop
CX Venous Controversies
CX Innovation Showcase programme highlights
Summary of Vascular News profiles 2017/18
https://vascularnews.com/wp-content/uploads/sites/7/2018/03/VN-CX18_low-res_EU.pdf
The second patent for peripheral artery disease-focused device company XableCath has been granted in the USA. The new patent specifically describes a catheter that controls tissue contact and has a rigid ring implement that opens a blood vessel occlusion by...
Medical technology company Bentley, a developer and manufacturer of devices for the minimally invasive treatment of vascular diseases, has announced that it will officially open its new headquarters on 21 April 2018.
The move to the state-of-the-art facilities, also...
The 24-month data from the Viabahn BX Flex balloon-expandable stent (Gore) investigational device exemption (IDE) trial are “promising” with sustained clinical and patency benefits and a favourable safety profile consistent with the previously published nine-month outcomes.
These results were presented...
Preliminary findings from an initial study with 25 patients have shown that measuring the changes in oxygen concentration at baseline, during, and after endovascular revascularisation, can predict wound healing and functional improvement in critical limb ischaemia patients.
Marianne Brodmann, Medical...
Andrew Holden spoke to Vascular News at LINC 2018 (Leipzig Interventional Course; 30 January–2 February, Leipzig, Germany) about the new data from the IN.PACT AV access trial and its significance, as well as Holden's experience with the drug-coated balloon...
Contego Medical has announced the beginning of enrolment in the PERFORMANCE I trial, a clinical trial designed to evaluate the safety and feasibility of the Neuroguard IEP carotid stent and post-dilation balloon system, with integrated embolic protection. The Neuroguard IEP...
Cardiovascular Systems Inc (CSI) presented 18-month outcomes from its LIBERTY 360 study at the Cardiovascular Research Technologies (CRT; 3-6 March, Washington, DC, USA). The study evaluates the acute and long-term clinical and economic outcomes of endovascular device interventions, including...
The first patient has been treated in the EVAS2 IDE confirmatory clinical study of the investigational Nellix EndoVascular Aneurysm Sealing (EVAS) System (Endologix) by Sajjad M Hussain, chief of the Department of Vascular Surgery at St Vincent Health and...
Terumo has entered into a definitive asset purchase agreement with Medeon Biodesign for its XPro large bore vascular closure system. The transaction consists of an up-front payment of US$20 million and milestone payments.
According to a press release, Medeon will continue to provide...
New two-year results from the IN.PACT SFA Japan trial were presented at the Leipzig Interventional Course (LINC; 30 January–2 February, Leipzig, Germany) by Osamu Iida, Kansai Rosai Hospital, Hyogo, Japan. The results were consistent with two-year findings from the...
The BioMimics 3D (Veryan Medical) helical swirling flow stent system provides an alternative to antiproliferative drugs, according to one-year data from the MIMICS-2 trial. The results were presented at Leipzig Interventional Course (LINC; 30 January–2 February 2018, Leipzig, Germany)...
Abbott and Surmodics have announced an agreement whereby Abbott will have exclusive worldwide commercialisation rights for Surmodics' SurVeil drug-coated balloon (DCB) to treat the superficial femoral artery, which is currently being evaluated in a US pivotal clinical trial. Separately, Abbott...
Access Vascular has announced that the company received 510(k) clearance from the US Food and Drug Administration (FDA) for its lead product, the HydroPICC, a peripherally inserted central catheter (PICC), intended for long-term use. In pre-clinical testing, the HydroPICC...
CBSET, a not-for-profit preclinical research institute dedicated to translational research, education, and advancement of medical technologies, has announced that its scientists have published data and analyses “Defining drug and target protein distributions after stent-based drug release: Durable versus deployable...
Vascular News interviews Panos Kitrou, Patras University Hospital, Rio, Greece, on the interim data from the LUTONIX DCB AV registry trial. The data were presented at the Leipzig Interventional Course 2018 (LINC; 30 January-2 February, Leipzig, Germany).
https://www.youtube.com/watch?v=g1xN3knVdw4
Endologix, a California-based developer and marketer of innovative treatments for aortic disorders, has announced that John McDermott is stepping down as the company’s Chief Executive Officer (CEO). McDermott will remain in his current role until a successor is found.
"His...
PQ Bypass has announced the enrolment of the first patients in the pivotal DETOUR II trial in the USA. The trial is a prospective, single-arm clinical trial designed to evaluate the DETOUR System for percutaneous femoropopliteal bypass.
The study will...
Corindus has announced receiving 510(k) clearance from the US Food and Drug Administration (FDA) for use of the CorPath GRX system in peripheral vascular interventions. The system brings the added benefits of robotic precision and radiation protection to minimally invasive...
The Food and Drug Administration (FDA) has today issued the final rule on “human subject protection; acceptance of data from clinical investigations for medical devices”.
The rule updates the FDA’s standards for accepting clinical data from clinical investigations conducted...
Michael Edmonds is professor of Diabetic Foot Medicine, King’s College London, and consultant physician, King’s College Hospital, London, UK. A pioneer in many areas of the diabetic foot, Edmonds speaks about his interest in diabetes and its treatment, the...
Data from the LOCOMOTIVE registry first-in-man trial investigating the VascuFlex Multi-Loc, B Braun’s multiple stent delivery system, were presented by Klaus Amendt (Diakonissenkrankenhaus, Mannheim, Germany) at the Leipzig Interventional Course (LINC; 30 January–2 February 2018, Leipzig, Germany). The data...
After two years of follow-up, the ILLUMENATE Global study has confirmed the durability of the Stellarex drug-coated balloon (Spectranetics/Philips) with primary patency rates comparable to those reported in randomised controlled trials.
The data were presented by principal investigator Thomas Zeller...
Cardiovascular Systems, a medical device company developing and commercialising interventional treatment systems for peripheral and coronary artery disease, announced today that the first patients were treated using its FDA-cleared extended length Diamondback 360 peripheral orbital atherectomy device (OAD) to treat peripheral artery...
This educational supplement is for distribution within Europe only.
In this supplement:
The rationale of directional atherectomy and DCB angioplasty for the treatment of “no-stent zones”
CASE #1: Popliteal and tibial artery occlusions
CASE #2: Severe and calcified CFA disease
CASE #3: Long...
Tokyo-based Terumo Corporation has announced an investment of £33 million in one of its subsidiary companies, Vascutek Limited, located near Glasgow, UK.
According to a company release, the investment has been approved for the major expansion of Vascutek’s headquarters and...
Cagent Vascular, a developer of technology for vessel dilatation in cardiovascular interventions, has announced positive results from the first-in-human PRELUDE study of the company's percutaneous transluminal angioplasty (PTA) balloon catheter.
Principal invesitagor Andrew Holden (Auckland, New Zealand) presented 30-day and...
Early data from the first randomised controlled trial to compare two drug-coated balloons (DCBs) suggest that the primary patency obtained with the Ranger DCB (Boston Scientific) is “comparable” to that achieved with the IN.PACT DCB (Medtronic) in the treatment...
Highlights:
COMPASS PAD signals new direction in anticoagulation of peripheral arterial disease patients
Gutterless chimney endograft could be the “pot of gold at the end of the rainbow”
Ross Naylor: ESVS carotid guidelines
Profile: Michael Edmonds
https://vascularnews.com/wp-content/uploads/sites/7/2018/02/77-Vascular-News_US.pdf
Highlights:
COMPASS PAD signals new direction in anticoagulation of peripheral arterial disease patients
Gutterless chimney endograft could be the “pot of gold at the end of the rainbow”
Ross Naylor: ESVS carotid guidelines
Profile: Michael Edmonds
https://vascularnews.com/wp-content/uploads/sites/7/2018/02/77-Vascular-News_EU.pdf
Endologix, a developer and marketer of treatments for aortic disorders, has announced the completion of enrollment in the ELEVATE (Expanding Patient Applicability with Polymer Sealing Ovation Alto Stent Graft) IDE clinical study. The objective of the 75-patient study is to...
An international meta-analysis of clinical experience in patients who received an endovascular arteriovenous fistula (endoAVF) for haemodialysis access was presented at Leipzig Interventional Course (LINC; 30 January–2 February, Leipzig, Germany) 2018.
The meta-analysis, which analysed data from 236 patients...
Primary endpoint data from the first-in-man study of SELUTION, Med Alliance’s novel sirolimus-coated balloon, were presented at the Leipzig Interventional Course (LINC; 29 January–2 February, Leipzig, Germany) by principal investigator Thomas Zeller, Universitaets-Herzzentrum, Bad Krozingen, Germany.
Median late lumen loss...
LimFlow has announced completion of enrolment of the original 10-patient cohort in the US feasibility study of the LimFlow percutaneous deep vein arterialisation system.
The company also announced that the US FDA has accepted the company’s proposal to expand...
Medtronic added to its body of clinical evidence supporting the IN.PACT Admiral drug-coated balloon with new presentations that demonstrated durable and consistent clinical outcomes in peripheral artery disease.
The new data presented at the Leipzig Interventional Course (LINC) in...
ItBentley has launched BeGraft Plus, a covered balloon expandable stent indicated for restoring and improving the patency and treatment of aneurysms, acute perforations and ruptures of the iliac and renal arteries in the European markets.
Eric Verhoeven (Nuremberg, Germany) officially...
Thomas Zeller (Bad Krozingen, Germany) presented the 12-month results from Veryan Medical’s MIMICS-2, a prospective, single arm, multicentre clinical study to evaluate the safety and effectiveness of the BioMimics 3D helical, swirling flow stent system in the treatment of patients with...
The post-market, non-interventional, multicentre, non-randomised, single-arm study will enroll approximately 150 patients across 25 sites in Europe and Russia, and will evaluate the safety and performance of a ChEVAR procedure using the Endurant II/IIs stent graft system (Medtronic) in...
Medtronic has announced the launch of the ENCHANT (Endurant ChEVAR New Indication Trial) study. The post-market, non-interventional, multi-centre, non-randomised, single-arm study will enrol approximately 150 patients across 25 sites in Europe and Russia, and will evaluate the safety and...
The final results of the ENDURE study, investigating the efficacy of the Chocolate Touch drug-coated balloon (QT Vascular), show “promising evidence of the drug effect by way of low late lumen loss and high patency at 12 months,” according...
Bluegrass Vascular Technologies has announced the enrolment of its first patients in the SAVE-US (Surfacer System to Facilitate Access in Venous Occlusions–United States) pivotal trial.
The SAVE-US trial is a pre-market investigational device exempt (IDE) study evaluating the safety and efficacy of the Surfacer Inside-Out Access...
Enrolment is complete in the Lutonix below-the-knee trial with BD planning to submit a pre-market approval application to the US Food and Drug Administration (FDA) in late 2018.
The Lutonix below-the-knee trial is a prospective, multicentre, randomised, controlled trial intended...
The first patients have been enrolled in the US FDA pivotal study of VasQ (Laminate Medical Technologies), an implanted blood vessel external support device for patients requiring arteriovenous fistula as vascular access for haemodialysis.
The first surgeries were performed by...
Cardiovascular Systems (CSI) has announced two new partnerships broadening the company’s product portfolio. According to a company release, CSI is now the exclusive US distributor of OrbusNeich balloon products. Additionally, the company has signed an original equipment manufacturer agreement...
A new study suggests thoracic endovascular aortic repair (TEVAR) may be appropriate in uncomplicated Type B aortic dissection (TBAD) based on several anatomic criteria determined by CT imaging.
Historically, the standard of care for uncomplicated patients with TBAD has been...
The registry will combine noninvasive, vascular ultrasound testing data with vascular treatment and outcomes data, making it possible to analyse the relationships between diagnosis and care provided to patients with vascular disease.
The Society for Vascular Ultrasound (SVU), the Society...
There has been significant progress in limb salvage in patients with peripheral artery disease (PAD) and critical limb ischaemia (CLI) over the past two decades. These advancements have been promoted by the increased knowledge and understanding of the disease...
XableCath, a clinical stage medical device company, announced its XableCath blunt tip support catheter has received clearance from the US Food and Drug Administration (FDA). The blunt tip catheter facilitates true lumen passage of lesions, both above and below...
BD, a leading global medical technology company, has announced it has completed the acquisition of C R Bard, creating a new health care industry leader with approximately US$16 billion in annualised revenue.
In a press release, BD state that the combined...
Endovascular navigation startup Centerline Biomedical has successfully completed a fifth preclinical study at Cleveland Clinic facilities evaluating its surgical navigation system, the Intra-Operative Positioning System (IOPS). The study was the company’s most noteworthy study to date; demonstrating ability to...
Global healthcare company Penumbra has entered into a joint venture with virtual reality (VR) company Sixense Enterprises, for the purpose of exploring healthcare applications of the technology.
MVI Health represents a unique partnership that brings together Sixense Enterprises, a team with deep...
Avinger, a developer of treatments for peripheral artery disease (PAD), has announced the first use of their next generation Pantheris lumivascular atherectomy system. The device was used by vascular surgeon Arne Schwindt to treat patients at St Franziskus Hospital in Münster, Germany.
Schwindt,...
Growing up in Kenya, Stéphan Haulon’s early experience of the medical profession came in the form of the “flying doctors”, who piloted small planes to remote areas to care for patients. Combined with his post-medical school experience providing humanitarian...