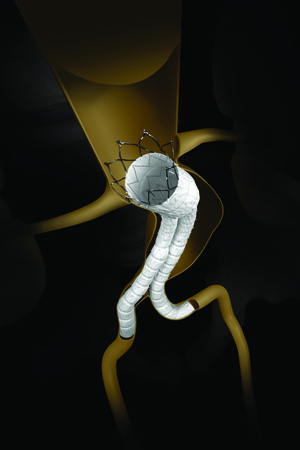
The US Food and Drug Administration (FDA) Circulatory System Devices Panel of the Medical Devices Advisory Committee has provided a favourable recommendation on the premarket approval application for INCRAFT AAA Stent Graft System (Cardinal Health). The panel voted 11 to 4 in favour of the benefits of the INCRAFT system.
According to the company, the INCRAFT system is an advanced endovascular aneurysm repair (EVAR) technology for the treatment of infrarenal abdominal aortic aneurysms (AAAs).
“We are pleased with the panel’s recommendation, which brings us one step closer to making the INCRAFT system available for thousands of high-risk patients in the USA,” says Shaden Marzouk, chief medical officer at Cardinal Health. “We appreciated the opportunity to present our data supporting the INCRAFT system and look forward to continuing discussions with the FDA to bring this technology to doctors and patients.”
While several EVAR devices are currently available in the USA, treatment options are limited for many AAA patients with small femoral or iliac arteries or with heavily calcified or tortuous vessels that could lead to complications during the introduction of EVAR devices.
Cardinal Health says that the INCRAFT system is an ultra-low profile and flexible stent-graft system designed to prevent rupture of infrarenal AAAs in a wide range of patient population.
The favourable vote of the advisory committee followed a review of clinical data from the pivotal INSPIRATION trial, a prospective, multicentre, single-arm study to evaluate the safety and effectiveness of the INCRAFT system in patients with AAA. The trial showed that the INCRAFT system met the primary safety and effectiveness endpoints, with a low rate of major adverse events at 30 days and a high rate of successful aneurysm treatment at one year. As presented at the Circulatory System Devices Panel meeting, the trial demonstrated high survival of nearly 80% and no aneurysm ruptures through four years of follow up.
The INCRAFT system, which received a CE mark in 2014, is commercially available in 39 countries. The INCRAFT system is an investigational device not available for sale in the USA.