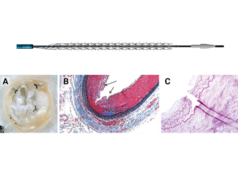
The first-in-human data from the DEEPER trial were presented at VIVA 2018 (5–8 November, Las Vegas, USA) by John Laird, Adventist Heart & Vascular Institute team at St Helena Hospital, USA.
The purpose of this trial is to evaluate the safety and efficacy of the Temporary Spur stent system (Reflow Medical), in conjunction with a drug-coated balloon (DCB) in patients with below-the-knee disease. The primary efficacy endpoint of the trial is binary arterial flow of treated lesion sites by continuous wave Doppler through six months. The dual primary safety endpoints are freedom from device- and procedure-related death through 30 days post-procedure and freedom from target limb major amputation and clinically driven target lesion revascularisation through 12 months post-procedure.
This trial is a prospective single centre non-randomised single arm study. Trial enrolment began in October 2017 and was completed in May 2018, with 21 patients having completed trial and procedure enrolment and undergoing follow-up. Twelve patients have completed follow-up through six months, and all 21 have completed follow-up out to at least three months.
Laird reported that all 21 enrolled patients have met the safety endpoints of the trial to this date, with no clinically driven target lesion revascularisation, no major adverse limb events, and freedom from device- and procedure-related death through 30 days post-procedure.
Out of the group that has completed six-month follow-up, 11 patients (92%) were found to have patent flow by waveform Doppler through six months, the primary efficacy endpoint, confirmed by ultrasound/angiogram for Spur/DCB-treated areas. Adjudication of the Doppler ultrasound results and angiographic follow-up results has been conducted with a core lab. The majority of patients in this trial have seen an improvement in clinical outcomes, with an average decrease in Rutherford class score by two classes (max –4, min 0) at six months. Of patients with wounds, 88% saw complete wound healing at six months and all saw improvement.