Intact Vascular, a developer of medical devices for minimally invasive peripheral vascular procedures, has announced that it closed a Series C financing totaling US$20 million. This financing is designed to fund the company through premarket approval (PMA) of the Tack Endovascular System for the treatment of post-angioplasty dissections above the knee.
Major participants in the Series C financing included New Enterprise Associates (“NEA”), H.I.G. BioVentures and Quaker Partners. The financing is structured in two stages or tranches, with the second tranche closing later in 2018.
“We continue to be impressed with the extensive clinical program that Intact Vascular is advancing to illustrate the significant clinical benefits that a better solution for dissections can have on angioplasty for peripheral arterial disease and critical limb ischaemia,” said Justin Klein, partner at New Enterprise Associates and member of the Intact Vascular Board of Directors. “NEA is proud to partner with the Intact team, the world class group of TOBA trial series investigators, and our other investing partners to advance the Tack through its pivotal trials and launch as a global commercial growth opportunity.”
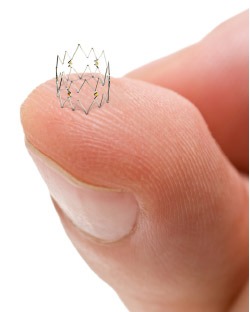
The Tack Endovascular System is a new solution for precision dissection repair following balloon angioplasty with a first-of-its-kind implant that is designed to help maintain vessel integrity and enhance blood flow to promote healing, improve outcomes and preserve limbs. Unrepaired dissections — which are frequent following balloon angioplasty — increase the probability of acute arterial occlusion and may continue narrowing the artery, which leads to lower long-term patency rates. The Tack Endovascular System leaves a minimal amount of metal in the artery, reduces mechanical stress on the arterial wall and preserves future treatment options.
“We are at an exciting inflection point in the development of the Tack Endovascular System. With the projected completion of primary endpoint analysis for our pivotal TOBA II trial, and the projected completion of enrollment in both TOBA II BTK and TOBA III, 2018 is a true watershed year for us,” said Bruce Shook, Intact Vascular’s president and CEO.
Intact Vascular is sponsoring three clinical trials to evaluate its Tack Endovascular System: TOBA II, TOBA III and TOBA II BTK. TOBA II is investigating the combination of the Tack device with both plain and BARD Lutonix drug-coated balloon (DCB) angioplasty in the arteries above the knee, and completed enrolment in March 2017. TOBA III is investigating the combination of the Tack system with the Medtronic IN.PACT Admiral DCB in the arteries above the knee (inclusive of long lesions), and is nearing full enrolment, and TOBA II BTK is investigating the combination of the Tack device with plain balloon angioplasty in the arteries below the knee and is actively enrolling patients.