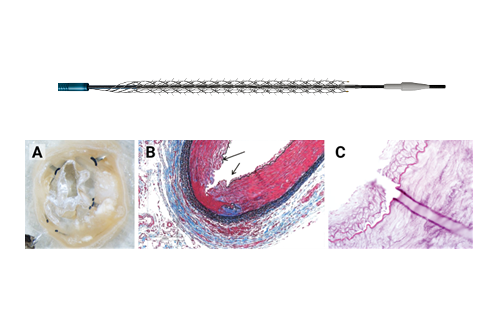
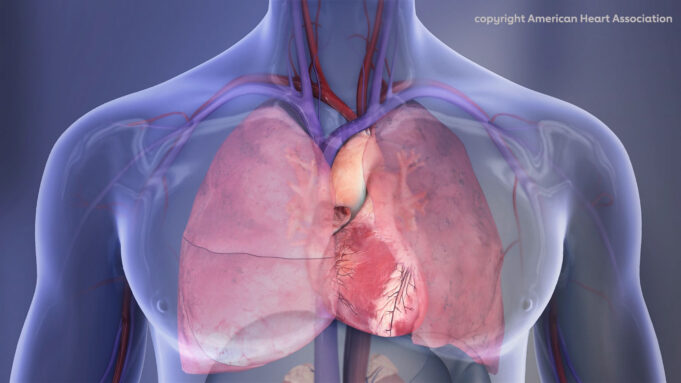
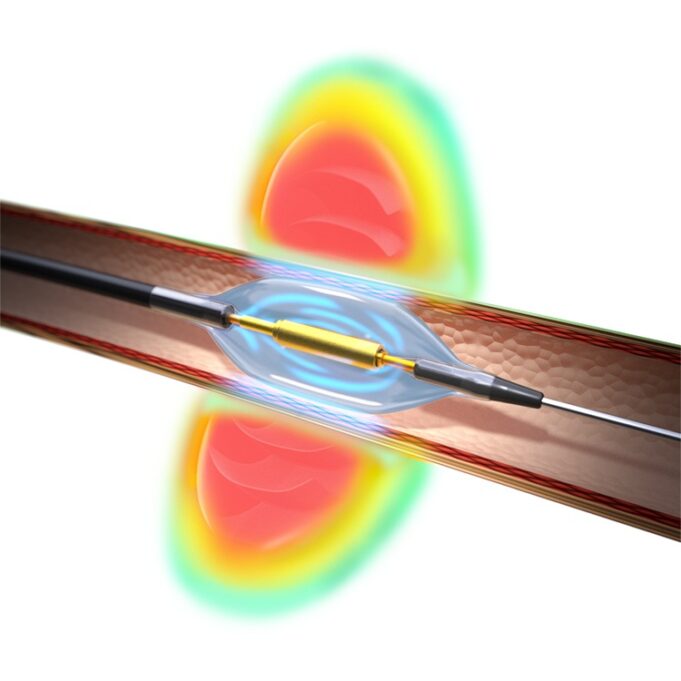
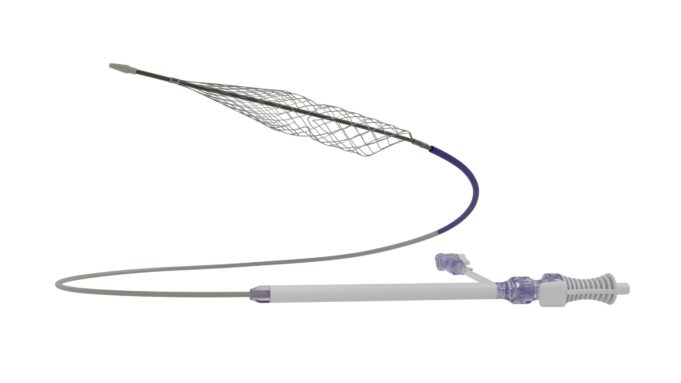
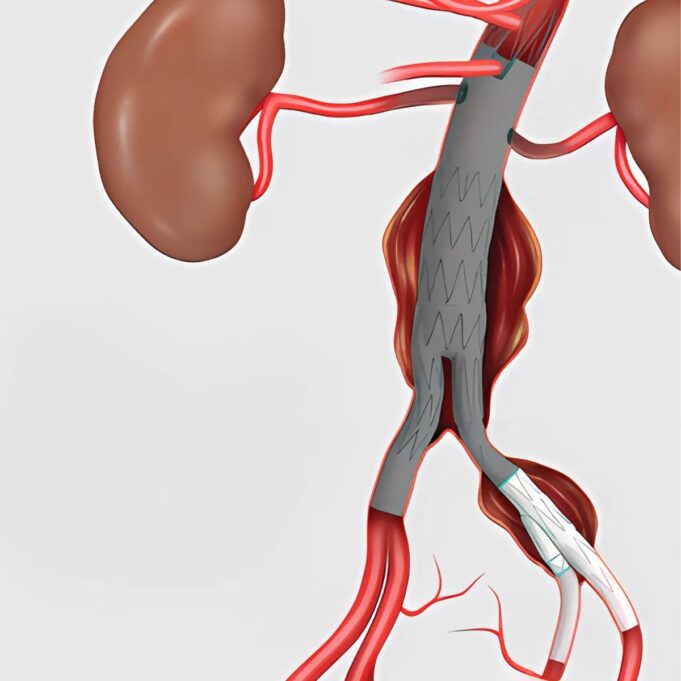
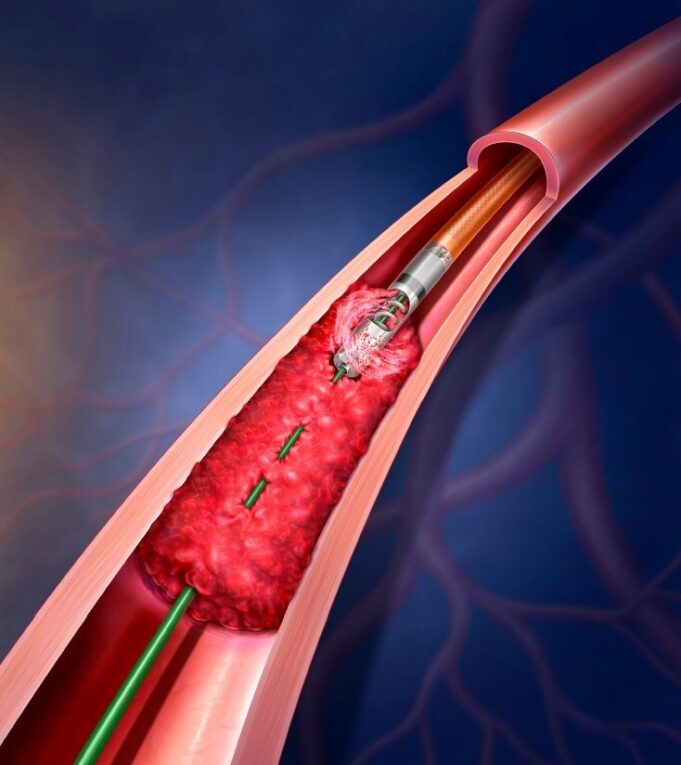
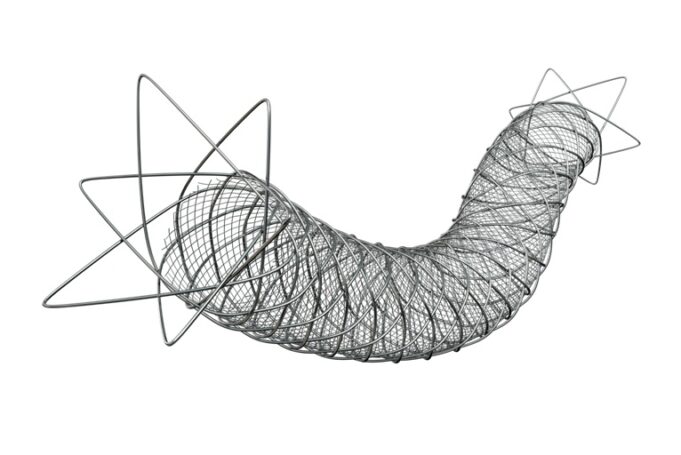
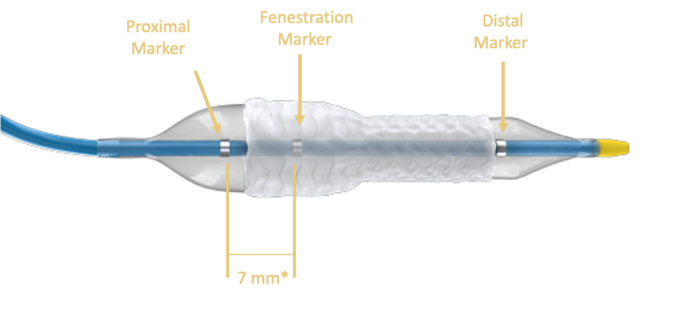
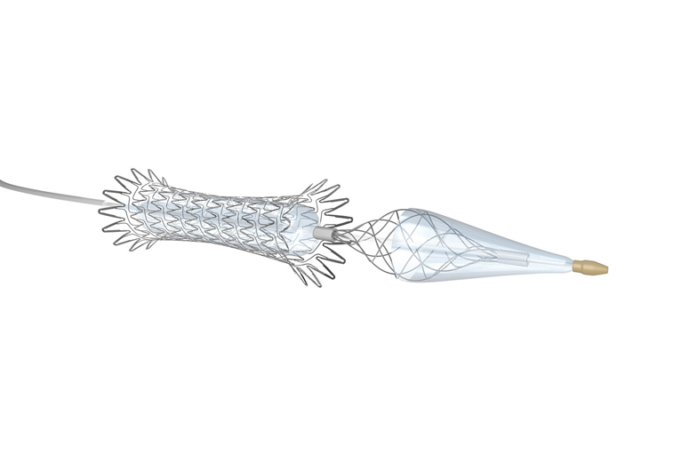
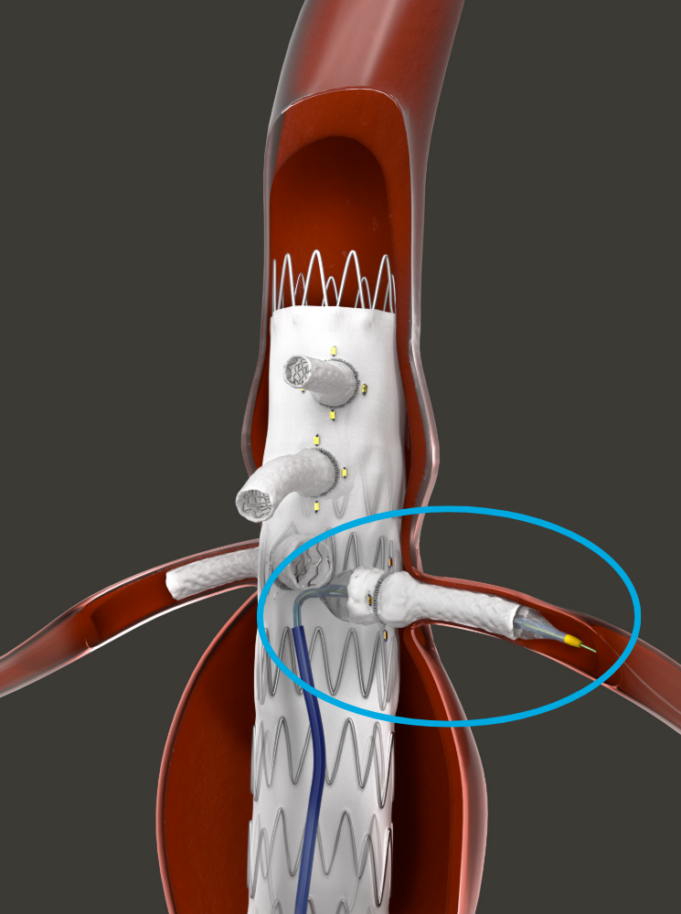
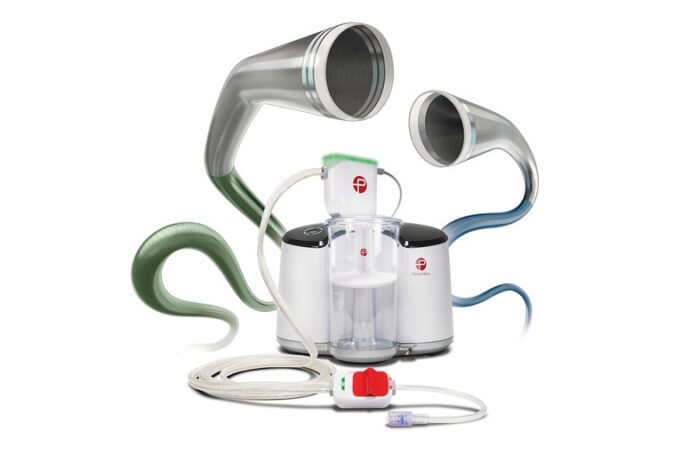
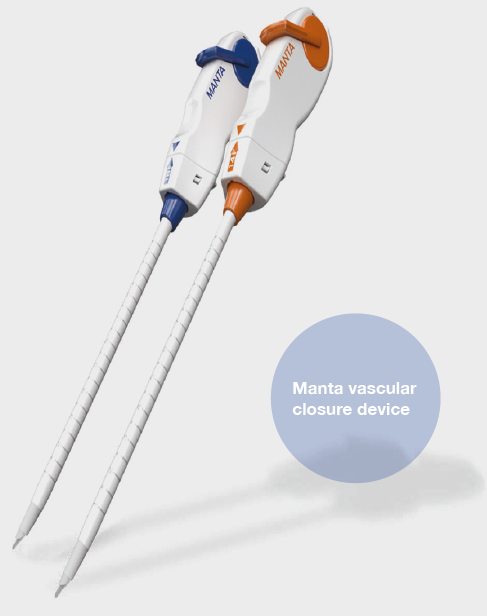
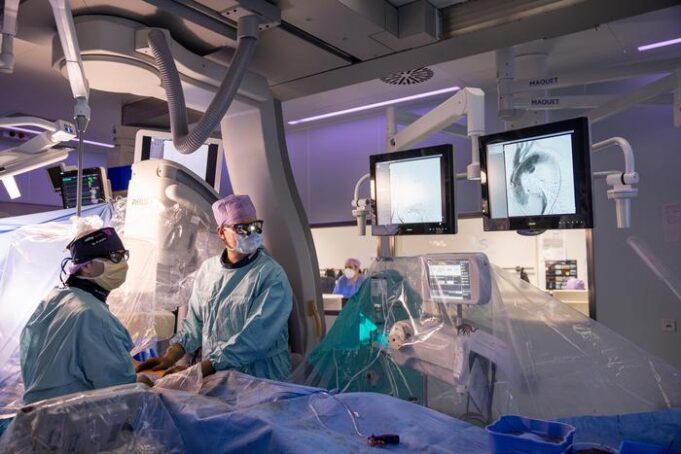
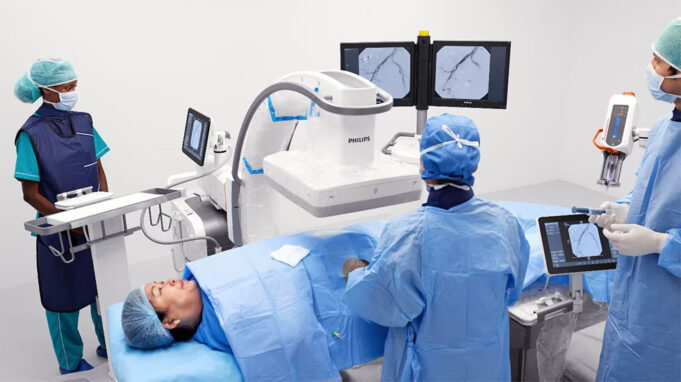
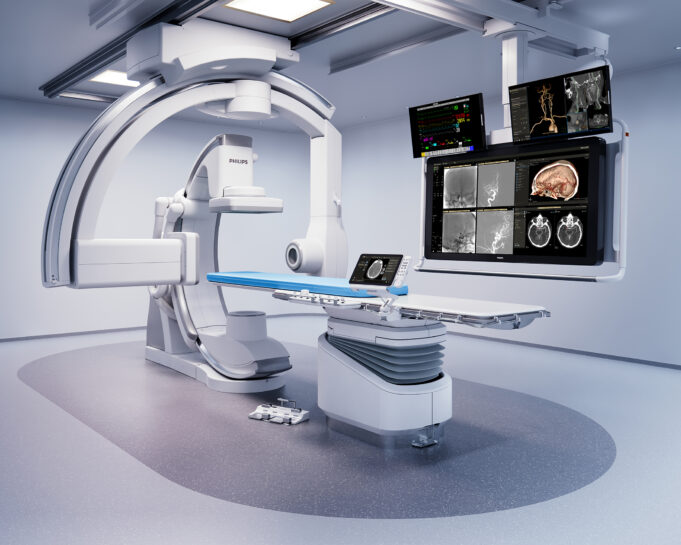
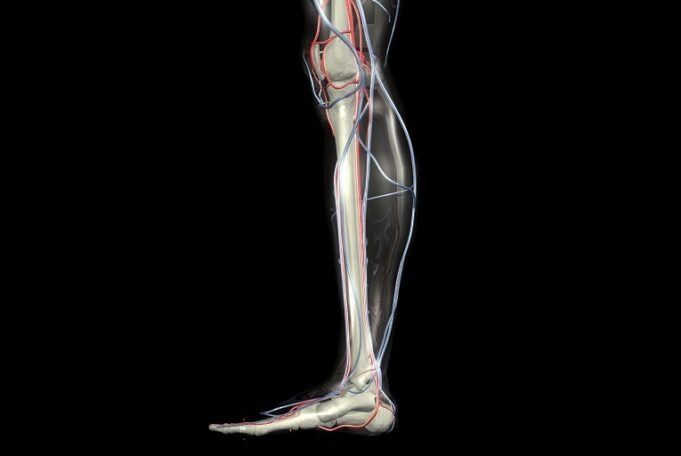
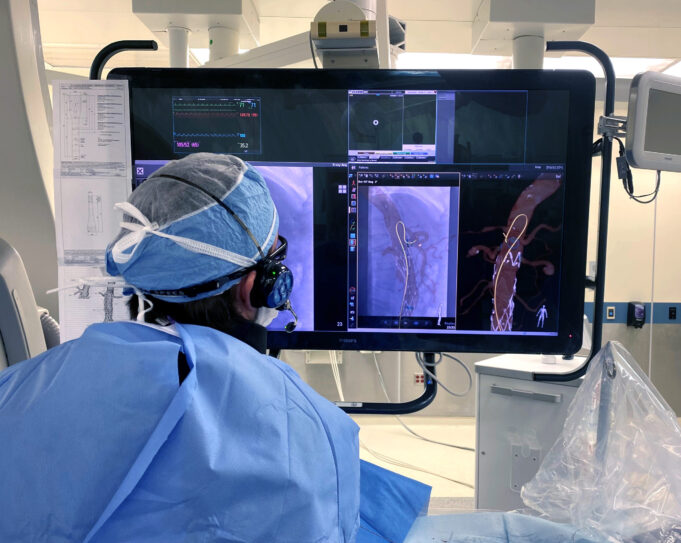
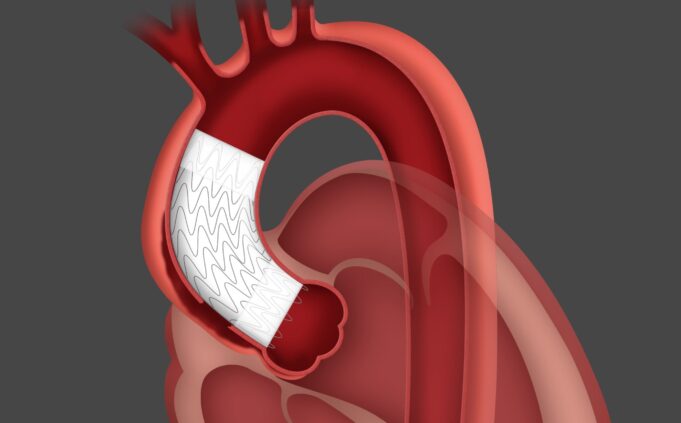
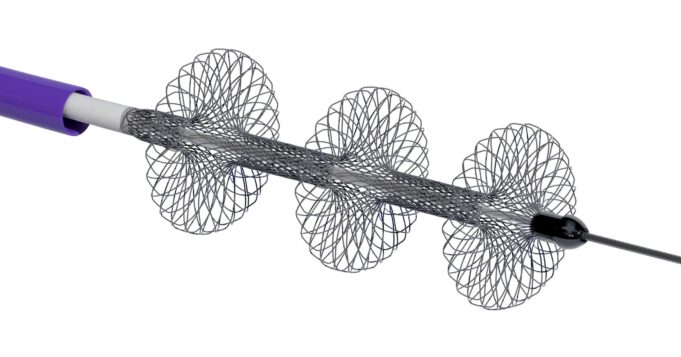
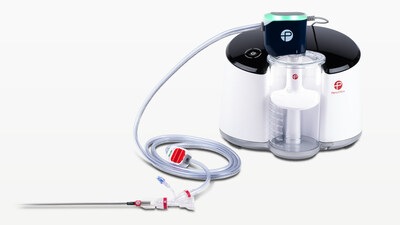
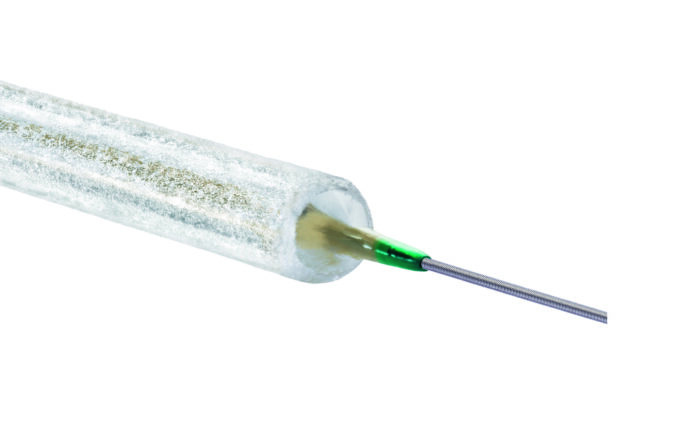
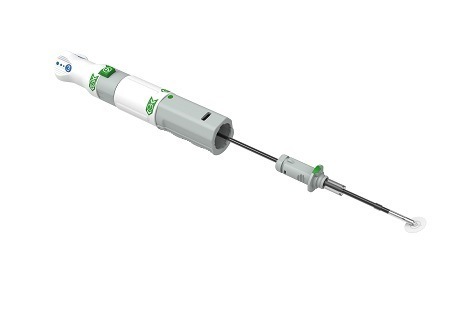
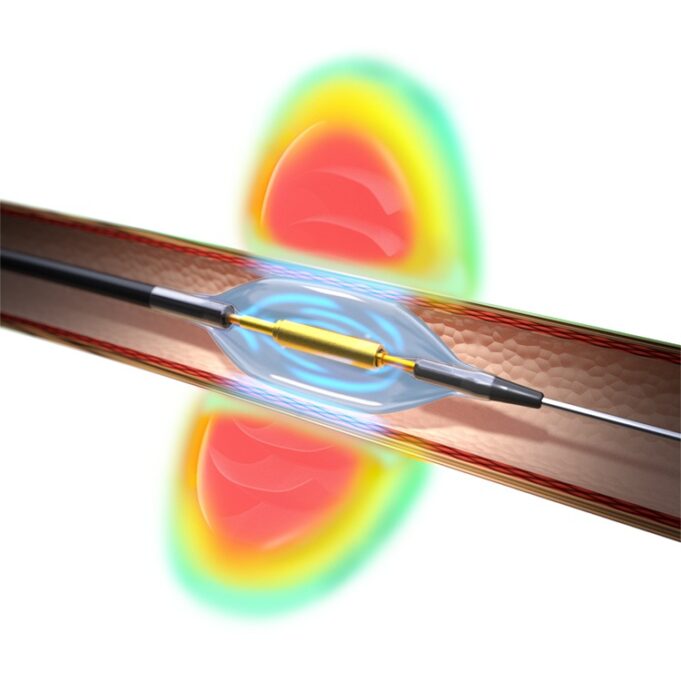
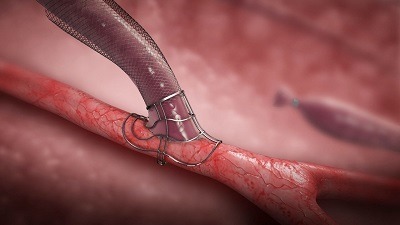
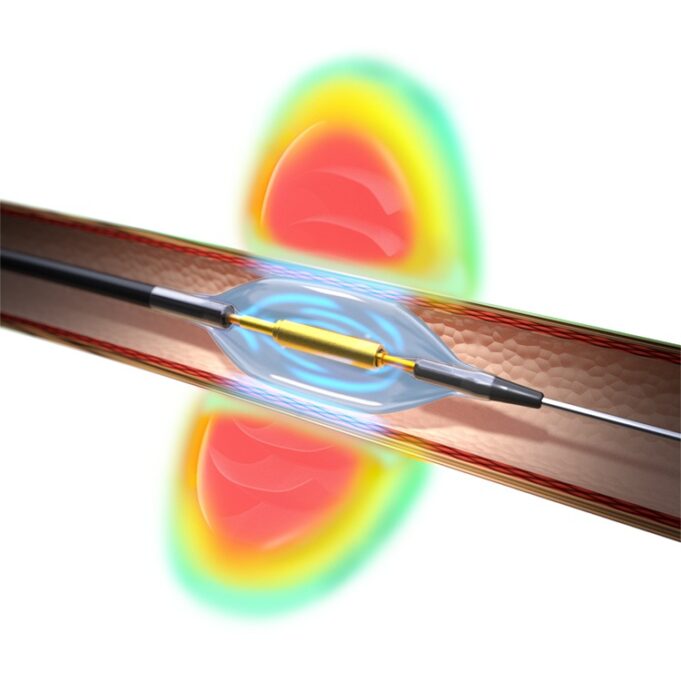
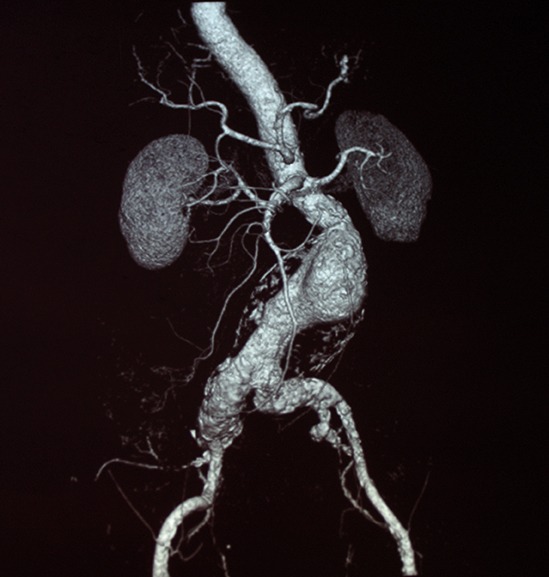
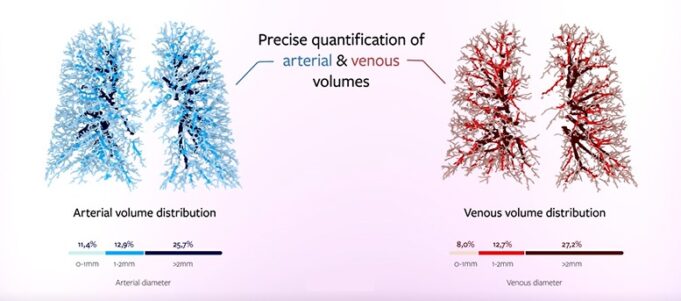
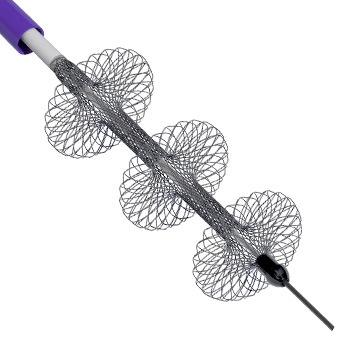
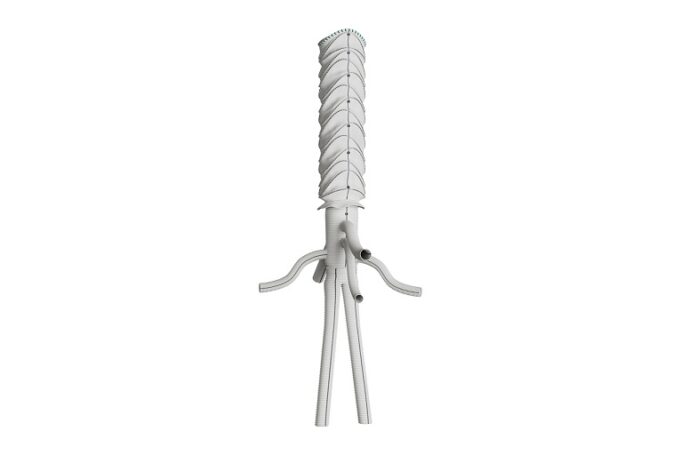
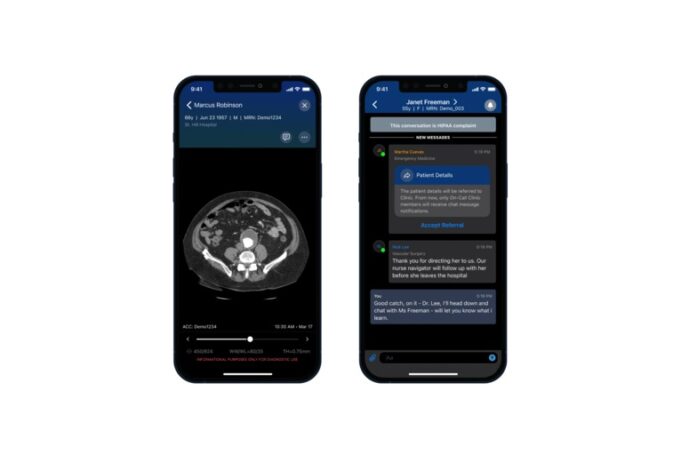
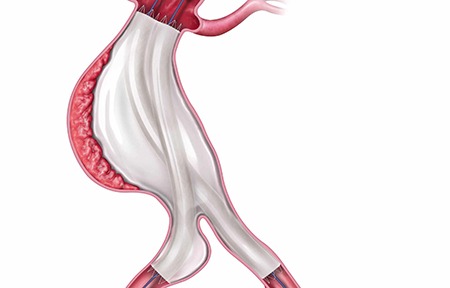
Shape Memory Medical today announced enrolment of the first patients in FLAGSHIP, the company’s prospective, single‑arm, open‑label first‑in‑human study evaluating its investigational false lumen embolisation (FLE) system for the treatment of aortic dissection.
FLAGSHIP is designed to evaluate safety and...
In the SirPAD trial, angioplasty with sirolimus-coated balloons led to a lower incidence of major adverse limb events (MALE) at one year than angioplasty with uncoated balloons among patients undergoing endovascular treatment for infrainguinal artery disease.
Co-principal investigator Stefano Barco...
This advertorial, sponsored by Reflow Medical, is only available in selected countries and geographies.
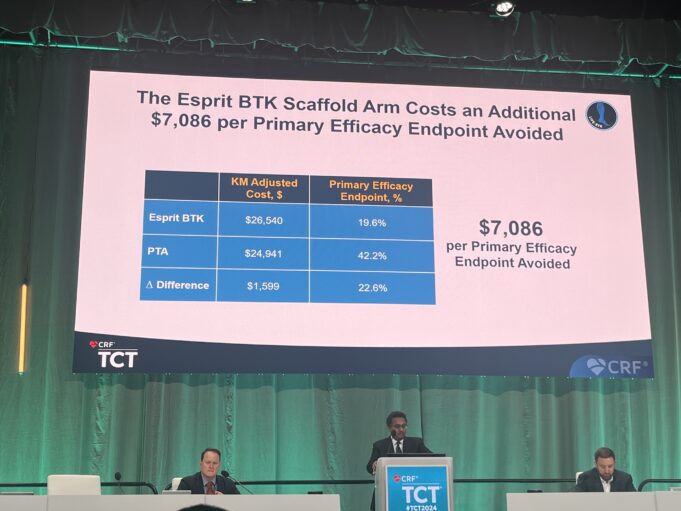
Addressing both recoil and impaired drug uptake remains central to improving durability in below-the-knee (BTK) chronic limb-threatening ischaemia (CLTI). This article explores how temporary scaffolding...
This advertorial, sponsored by Gore, is only available in selected countries and geographies.
Two Gore-sponsored registries are evaluating the performance of the company’s Viabahn VBX balloon-expandable endoprosthesis (VBX stent graft) in fenestrated and branched endovascular aneurysm repair (F/BEVAR). With...
Patients who underwent ultrasound-facilitated, catheter-directed fibrinolysis for acute pulmonary embolism (PE) had significantly better outcomes than those who received standard systemic anticoagulants, according to a study presented today at the American College of Cardiology’s Annual Scientific Session (ACC.26; 28–30...
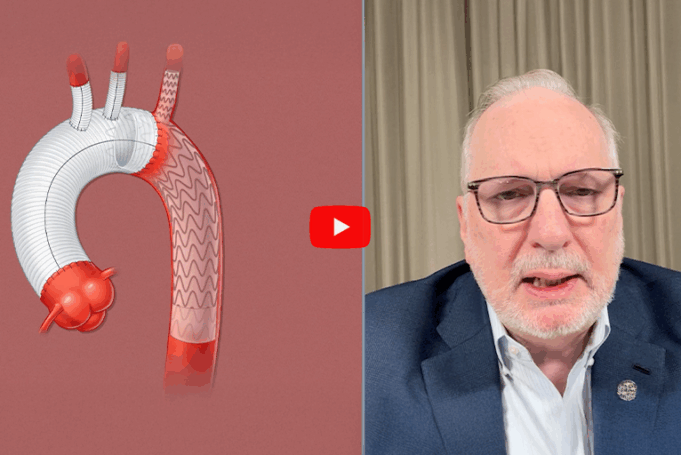
At the 2026 European Vascular Course (EVC; 8–10 March, Maastricht, Netherlands), Bijan Modarai (King’s College London and Guy’s and St Thomas’ NHS Foundation Trust, London, UK) addressed the nuances involved in management of subacute type B aortic dissection.
Focusing on...
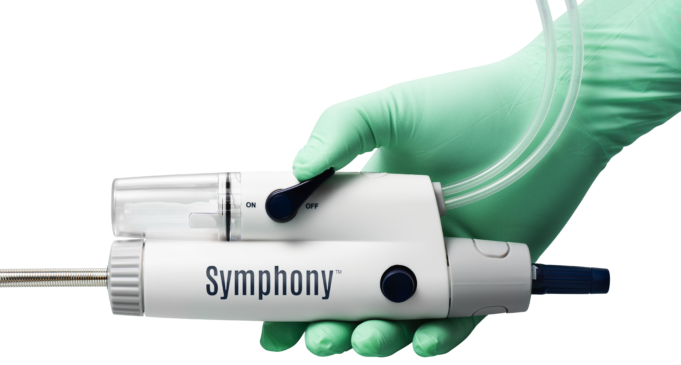
Imperative Care has announced the enrolment of the first patient in the CLEAR-IT study, a prospective, observational clinical study evaluating the performance of its Symphony and Prodigy thrombectomy systems.
“The CLEAR-IT study underscores Imperative Care’s commitment to advancing meaningful innovation grounded...
Nine medical societies are calling for updated safety standards in fluoroscopy laboratories, where clinicians face radiation exposure and orthopaedic injuries from heavy protective equipment.
A report published simultaneously today in JSCAI, Heart Rhythm, JVIR, and JVS–Vascular Insights details the health,...
Imperative Care has announced the closing of US$100 million in oversubscribed convertible note financing.
The round was co-led by new investors Elevage Medical Technologies and Perceptive Advisors in partnership with existing investor Catalio Capital Management. New investors Longaeva Partners and...
A white paper focused on the future of lower-limb vascular care and peripheral arterial disease (PAD) in the UK has been published by the All-Party Parliamentary Group on Vascular and Venous Disease (VVAPPG).
Developed in collaboration with the Association of...
The US Food and Drug Administration (FDA) has granted Breakthrough Device designation for the Fenestrated Treo abdominal stent-graft system, Terumo Aortic announced today.
The FDA’s Breakthrough Device designation programme is intended to fast-track the regulatory review process for certain medical...
iVascular has revealed the three-year follow-up results from the LUMIFOLLOW registry, a large real-world study evaluating the safety and efficacy of the Luminor drug-coated balloon (DCB) in the treatment of femoropopliteal artery disease. The data was presented by Eric...
iVascular has announced the one-year results from the BARISTA trial, the first clinical trial with Restorer bare metal stent evaluating its safety and efficacy in the treatment of iliac artery lesions. The study provides evidence supporting the performance of...
ROSSINI-Platform—said to be the largest-ever UK surgical trial—is set to commence in the coming months with the aim of assessing various methods for reducing post-intervention surgical site infection (SSI). The project will encompass six parallel randomised controlled trials (RCTs)...
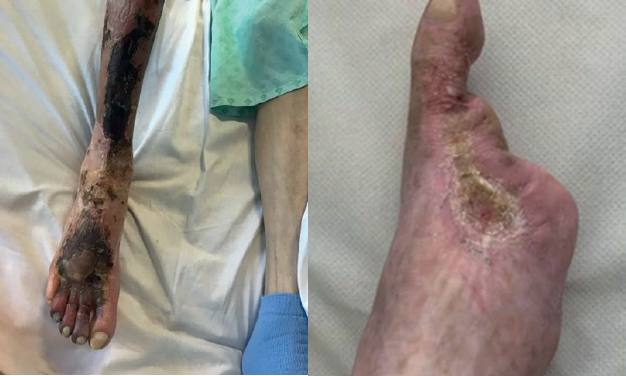
A large retrospective review of 3,126 patients with chronic wounds treated with multimodality intermittent topical oxygen therapy (ITOT) presented at the 2026 American Venous Forum (AVF; 28 February–4 March, Denver, USA) demonstrated superior and more durable wound healing outcomes...
For the first time in its history, the American Venous Forum (AVF) included a dedicated session focused on pulmonary embolism (PE) in its 2026 annual meeting (28 February–4 March, Denver, USA) programme. AVF executive committee member Patrick Muck (Cincinnati,...
VentureMed, the company behind the FLEX Vessel Prep system for arteriovenous (AV) access and peripheral arterial disease (PAD) interventions, is now doing business as Flex Vascular.
"The DBA (Doing Business As) name reflects the company's focused commitment to vascular innovation and...
Vena Medical recently announced it has received regulatory clearance from the US Food and Drug Administration (FDA) for the use of its flagship technology, the Vena MicroAngioscope system, in the peripheral vasculature.
According to the company, the Vena MicroAngioscope system...
A new severity-based classification system and strong endorsement for multidisciplinary pulmonary embolism response teams (PERTs) feature prominently in recently published multisociety guidelines on the evaluation and management of acute PE in adults.
The American College of Cardiology (ACC)/American Heart Association...
Nikolaos Patelis (Riyadh, Saudi Arabia) and colleagues have been investigating e-learning's role in training and educating the vascular community. Their most recent paper, published in Annals of Vascular Surgery, offers over five years of research on this topic. Here,...
BD today announced it has obtained CE marking for the Revello vascular covered stent, which the company describes as a next-generation endovascular solution for the treatment of atherosclerotic lesions in the common and external iliac arteries.
The Revello stent...
iVascular has announced the enrolment of the first patient in the fenCo clinical study, marking the initiation of a European clinical trial evaluating iCover in fenestrated endovascular aneurysm repair (FEVAR) for the treatment of complex abdominal aortic aneurysms. The...
The combination of pressurised topical oxygen therapy and non-contact cyclical compression addresses a trifecta of hypoxia, inflammation and lymphatic dysfunction in chronic wounds, showing promise as an adjunctive therapy to “accelerate healing, enhance clinical outcomes, reduce complications and achieve...
Humacyte recently announced the presentation of 36-month data assessing the durability of its Symvess acellular tissue-engineered vessel in extremity arterial trauma patients. The results were shared at the 2026 annual winter meeting for the Vascular and Endovascular Surgery Society...
Ben Li (Toronto, Canada) ponders the current state of artificial intelligence (AI) application in clinical practice, analysing the obstacles to scaled use.
AI has reached a level of technical maturity at which performance in many clinical and analytical tasks...
Preliminary three-month results from the first five human implantations of StentIt’s resorbable fibrillated scaffold (RFS) reveal a technical success rate of 100% and no device- or procedure-related serious adverse events at 30 days in patients with below-the-knee (BTK) chronic...
In a session at the 2026 Leipzig Interventional Course (LINC; 27–31 January, Leipzig, Germany) dedicated to pulmonary embolism (PE) management, Sahil Parikh (Columbia University Irving Medical Center, New York, USA) extolled the benefits of a PE response team (PERT)....
Despite original ambitions to become a journalist, the loss of a close relative to pulmonary embolism (PE) saw Marianne Brodmann (Graz, Austria) redirect her focus towards the medical world. Now the head of the Division of Angiology at the...
In a study comparing Philips’ Fiber Optic RealShape (FORS)-enabled guidewire with its enhanced successor LumiGuide, researchers found that the latter was associated with improved technical success and reduced procedure times.
Authors Paula Huertas, Carlos Timaran (UT Southwestern, Dallas, USA) and...
This advertorial, sponsored by Bentley, is only available in selected countries and geographies.
Recently conducted first-in-human procedures with a longer length of Bentley’s BeGraft Plus mark the latest in a series of moves from the company towards providing a...
Thomas J Fogarty, the world-renowned cardiovascular surgeon, innovator and inventor of the Fogarty catheter, died peacefully on 28 December 2025 at the age of 91.
Colleagues at Fogarty Innovation, a nonprofit educational incubator dedicated to advancing medical innovation, announced the news of their founder’s passing. “We are immensely grateful for all Tom did for patient care,...
Vascular Society of Great Britain and Ireland (VSGBI) president Keith Jones (Camberley, UK) shares his thoughts on the SWHSI-2 trial results.
The SWHSI-2 trial results looking at surgical wound healing by secondary intention (SWHSI) in the UK were first...
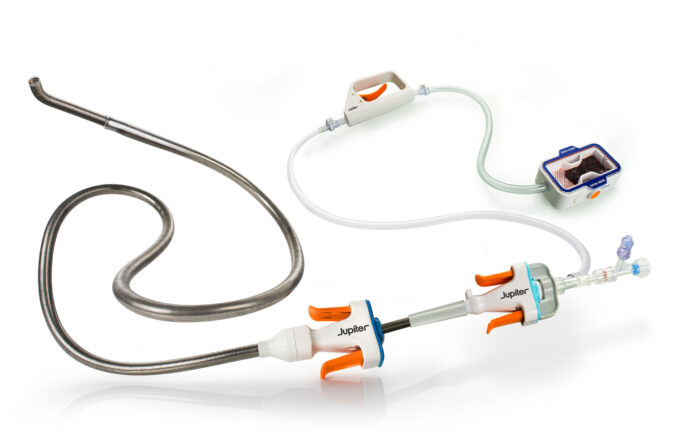
Jupiter Endovascular today announced completion of patient enrolment in the SPIRARE II pivotal trial evaluating the Vertex pulmonary embolectomy system in patients with acute, intermediate-risk pulmonary embolism (PE). The final subject was enrolled by Vikas Aggarwal (Henry Ford Hospital,...
Restoring blood flow to the brain by treating carotid artery stenosis does not appear to improve patients’ cognitive skills, according to preliminary late-breaking science presented today at the 2026 International Stroke Conference (ISC; 4–6 February, New Orleans, USA).
“Whether patients undergo...
Endospan today announced the one-year results of its TRIOMPHE investigational device exemption (IDE) study evaluating the Nexus aortic arch stent graft system. The data were presented at the Society of Thoracic Surgeons (STS) annual meeting (29 January–1 February, New...
GE HealthCare has announced that its Allia Moveo image-guidance platform has received US Food and Drug Administration (FDA) 510(k) clearance and CE mark.
The latest Allia platform was first unveiled at the Radiological Society of North America’s (RSNA) 2025 annual...
Four UK-based experts on the management of chronic limb-threatening ischaemia (CLTI) sat down at the 2025 Charing Cross (CX) Symposium to discuss the landscape of care, current challenges, and potential solutions to reduce both the prevalence of CLTI and...
Patrick Muck (Cincinnati, USA) examines some of the latest technologies available for radiation protection in the vascular space amid a “meteoric” rise in endovascular procedures.
Over the past decade, the adoption of endovascular techniques has accelerated dramatically, driven by technological...
Phraxis has announced the successful initiation of its post-approval study (PAS) for the EndoForce anastomotic connector, with the first procedure performed at Spartanburg Regional Healthcare System (Spartanburg, USA).
This milestone marks a significant transition from regulatory achievement to the establishment...
NOTE: This video is ONLY available to watch in selected countries and geographies Kate Steiner (East and North Hertfordshire NHS Trust, Stevenage, UK) and Matteo Tozzi (University of Insubria, Varese, Italy) discuss the growing body of evidence for treating...
Vascular surgeon Randy Moore (Calgary, Canada) comments on an emerging new technology using artificial intelligence (AI) as a tool to help predict aortic rupture on the sidelines of the 2025 VEITHsymposium (New York, USA; 18–22 November).
About three years ago, the number of papers in the vascular literature on artificial intelligence (AI) use in vascular surgery was minimal, observes Randy Moore (Calgary, Canada). More recently, that number has spiked to about 12,000, “which really paints...
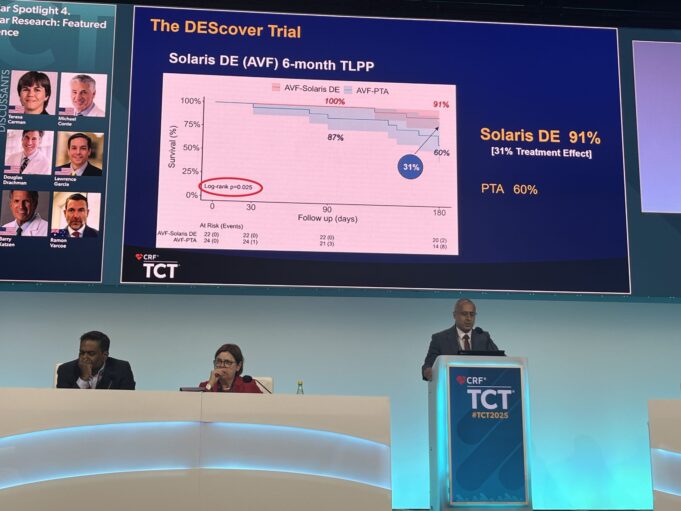
Effective, durable options to address faltering dialysis access circuits have remained elusive, leaving a significant unmet need in long-term access maintenance. Until now. Promising interim data have emerged from the prospective, multicentre DEScover trial evaluating the Solaris DE sirolimus-eluting,...
Penumbra has launched the Lightning Flash 3.0 computer-assisted vacuum thrombectomy (CAVT) system, a development that includes "significant upgrades" to the venous thromboembolism (VTE) platform, according to the company.
"Engineered for the rapid removal of pulmonary and venous thrombus, the latest...
In this issue:
CREST-2 analysis: Experts focus on medical management, trial design and plaque imaging following the presentation of landmark trial data
Opinion: Vascular Society of Great Britain and Ireland (VSGBI) president Keith Jones (Camberley, UK) shares his thoughts on...
In this issue:
CREST-2 analysis: Experts focus on medical management, trial design and plaque imaging following the presentation of landmark trial data
Opinion: Vascular Society of Great Britain and Ireland (VSGBI) president Keith Jones (Camberley, UK) shares his thoughts...
In an observational study of over 42,000 non-ruptured abdominal aortic aneurysm (AAA) repairs, researchers identified that dementia confers “clinically meaningful higher risks” across a spectrum of outcomes from mortality to discharge to higher levels of care.
Samir K Shah (University...
NOTE: This video is ONLY available to watch in selected countries and geographies
Matteo Tozzi (University of Insubria, Varese, Italy) comments on the current evidence supporting the use of drug-coated balloon (DCB) use in arteriovenous fistula (AVF) maintenance across...
InspireMD recently announced that results from the C-GUARDIANS pivotal trial have now been published in the Journal of the American College of Cardiology (JACC). The data demonstrate the safety and efficacy of carotid artery stenting (CAS) with the MicroNet-covered...
The development of endovascular aneurysm repair (EVAR) and stent grafts in the aortic space would never have happened without the cooperation between vascular surgeons and radiologists, says Michael Dake (Tucson, USA), an early pioneer. "This could not be done...
Boston Scientific and Penumbra today announced the companies have entered into a definitive agreement under which Boston Scientific will acquire Penumbra in a cash and stock transaction that values Penumbra at US$374 per share, reflecting an enterprise value of...
NOTE: This video is ONLY available to watch in selected countries and geographies
“I think the data that we have for the IN.PACT AV Access study show that there is a benefit for DCBs in terms of reducing...
An international panel of experts has released a series of consensus statements on the management and follow-up of acute type B intramural haematoma (IMH) and penetrating aortic ulcer (PAU), with findings recently published as an article in press in...
The European Society for Vascular Surgery (ESVS) has released updated clinical practice guidelines for the care of patients with descending thoracic and thoracoabdominal aortic pathologies. The document, which is available in the society’s dedicated journal, provides a “comprehensive and...
A US National Institutes of Health (NIH)-funded randomised controlled trial is due to commence this month to assess the efficacy of continuous digital monitoring in reducing diabetic foot ulcer recurrence among high-risk patients.
Led by Caitlin Hicks and colleagues at...
Solaris Endovascular has announced that the DEScover clinical trial has successfully completed enrolment.
DEScover is a prospective, multicentre clinical study evaluating the Solaris DE sirolimus-eluting, electrospun polytetrafluoroethylene (PTFE) covered stent for the treatment of vascular disease in dialysis access circuits.
The...
Haemonetics Corporation today announced the acquisition of Vivasure Medical, a Galway, Ireland-based company developing next-generation technology for percutaneous vessel closure.
Vivasure's PerQseal Elite system uses a proprietary bioabsorbable patch to seal large-bore (up to 26Fr) arteriotomies and venotomies from inside...
Terumo Aortic today announced enrolment of the first patient in the RapidLink pivotal investigational device exemption (IDE) study in the USA and Europe. The study is designed to evaluate the safety and effectiveness of the RapidLink device for the...
Black women with claudication had the highest rate of progression to chronic limb-threatening ischaemia (CLTI) after lower extremity revascularisation at 180 days, new data from a Medicare-linked Vascular Quality Initiative (VQI) procedural registry show.
The results were demonstrated in a...
“True patient-centred care demands more than interpreting scans and assessing flow." This is according to Launch Pad columnist Adam Talbot (Manchester, UK), who here argues that vascular surgeons must broaden their definition of limb function beyond the realm of...
From SWEDEPAD to CREST-2, Vascular News looks back at the stories that caught readers' attention in 2025.
1. SWEDEPAD re-opens paclitaxel safety discussion, finds drug-coated devices do not improve outcomes
Drug-coated balloons and stents were not associated with reduced risk of amputation or...
Gore in Japan has announced the first clinical use of the Gore Excluder thoracoabdominal branch endoprosthesis (TAMBE) in the East Asian country. The device, which enables endovascular treatment of thoracoabdominal aortic (TAAAs) and pararenal aneurysms while preserving blood flow...
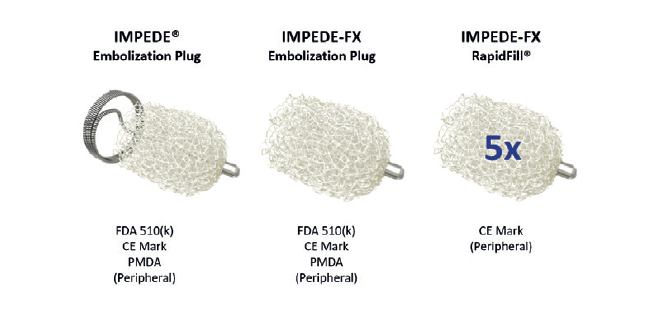
Shape Memory Medical announced that its Impede embolisation plug product family has received certification as a Class III device under the European Union (EU) Medical Device Regulation (MDR) 2017/745. The Impede devices, previously CE-marked under the Medical Device Directive...
Cook Medical has issued a letter to affected customers recommending certain Zenith Alpha 2 thoracic endovascular graft proximal (ZTA2-P/PT) components be removed from where they are used or sold, a medical device recall notice issued by the US Food...
Alik Farber (Boston, USA) speaks to Vascular News about his life and career in vascular surgery. The surgeon-in-chief at Boston Medical Center and James Utley professor and chair of surgery at Boston University Chobanian and Avedisian School of Medicine...
“In the early days of endografts, we didn’t really know what we were doing,” muses Christopher Zarins (Stanford, USA), a pioneer of the endovascular revolution in vascular surgery, recalling his early experiences with stent grafts and endovascular aneurysm repair...
Gore recently announced the first implantation of the investigational Gore ascending stent graft in the ARISE III trial for the treatment of an acute type A dissection.
The implant was performed at Emory University (Atlanta, USA) by Brad Leshnower, site...
The interim results of a first-in-human trial of a sirolimus-eluting covered stent graft—Solaris DE (Solaris Endovascular)—for the treatment of dialysis access dysfunction demonstrate the promise of the device in treating vascular access outflow stenosis, an investigator in the trial...
Medtronic has announced the first commercial use of its Liberant thrombectomy system, which is indicated for the removal of fresh, soft emboli or thrombi from the vessels of the peripheral arterial and venous systems. Medtronic notes that Liberant expands...
Bendit Technologies announced today that the US Food and Drug Administration (FDA) has granted 510(k) clearance for the Bendit17 microcatheter, which the company describes as the smallest steerable microcatheter on the market.
Bendit shares in a press release that the...
Penumbra recently announced that it has secured CE-mark approval for the Lightning Bolt 12 and Lightning Bolt 6X with TraX, which the company notes represent the next generation of its computer-assisted vacuum thrombectomy (CAVT) devices.
Penumbra states that these...
NOTE: This video is ONLY available to watch in selected countries and geographies
The SUCCESS PTA Study examining a real-world population of peripheral arterial disease (PAD) patients has produced “very convincing” data on the benefits of the SELUTION SLR™ Drug-Eluting...
Philips has announced the expanded commercial availability of LumiGuide 3D device guidance—the much-heralded first real-time artificial intelligence (AI)-enabled light-based navigation solution for image-guided therapy—across Europe and the USA.
LumiGuide integrates with Philips' Azurion platform, which is designed to drive procedural...
Angiolutions has announced that the first patient has been successfully treated with its investigational A3-Shield system in the PRINCIPIIS FIH trial, a first-in-human (FIH) study evaluating the device’s safety and effectiveness.
The trial is being conducted at several centres in...
Life Seal Vascular has announced the first clinical implants of their investigational Cygnum aneurysm sac management device (ASMD), with two patients enrolled in the ASCEND first-in-human (FIH) study in New Zealand.
A press release notes that the Cygnum device lines...
The PAD Collaborative, a group of 17 organisations convened by the American Heart Association (AHA) to address the growing burden of peripheral arterial disease (PAD) in the USA, has released a new and expanded version of its interactive map.
The...
Akura Medical has secured a US$53 million first close in series C financing, with the funds to be used to support development activities for the Katana thrombectomy system and the NavIQ quantification software, alongside completion of enrolment in the...
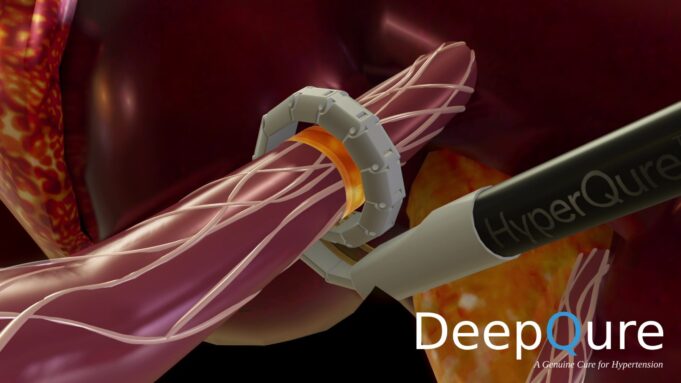
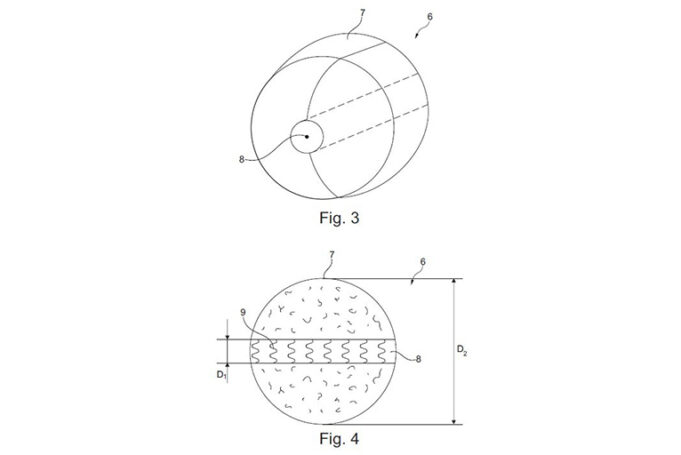
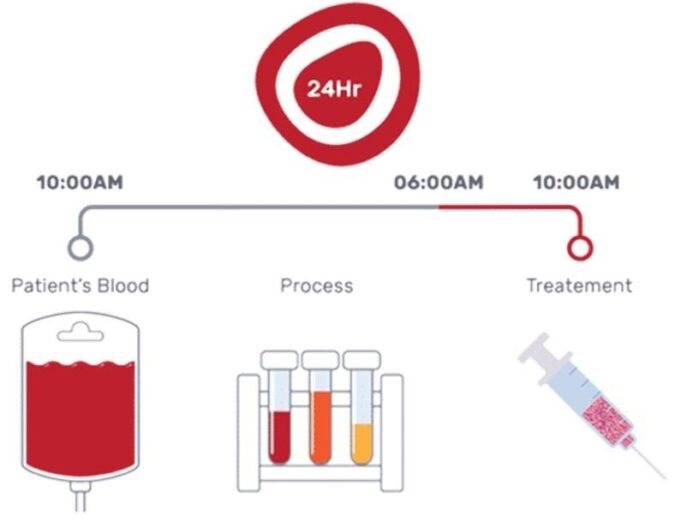
This advertorial is sponsored by Shape Memory Medical.
The field of embolisation has seen an increasing number of devices enter the market, raising an important question: how much does the choice of embolic device influence outcomes? The Impede embolisation plug...
Pathfinder Medical recently announced the appointment of interventional radiologist Lorenzo Patrone (Azienda USL Toscana Centro, Florence, Italy) as its chief medical officer (CMO).
Patrone has been collaborating with Pathfinder since 2019 as lead clinical consultant. A press release states that...
The international CREST-2 study has found that, for people with severe carotid artery narrowing who have not experienced recent stroke symptoms, a minimally invasive carotid artery stenting (CAS) procedure—combined with intensive medical therapy—significantly lowered stroke risk compared with medical...
A systematic review and meta-analysis of more than 300 original investigations on atherectomy in peripheral arterial disease (PAD) highlights “overall favourable” clinical outcomes. This and other findings from what researchers say is the first investigation to provide a “comprehensive”...
University of Leicester experts have been awarded nearly £2 million to set up a global vascular research centre, based at the National Institute for Health and Care Research (NIHR) Leicester Biomedical Research Centre, a press release reports.
The funding, from...
No major vascular complications were seen in the ELITE trial using the latest-generation PerQseal Elite (Vivasure Medical), a patch-based, resorbable, sutureless vascular device for large-bore arterial closure. Results of the trial were presented during a late-breaking trials session at...
Terumo Aortic today announced enrolment of the first patient in the Fenestrated Treo pivotal investigational device exemption (IDE) study in the USA. This study is designed to evaluate the endovascular repair of juxtarenal and suprarenal aortic aneurysms using the...
A focused session at the 2025 Vascular Interventional Advances (VIVA) conference (2–5 November, Las Vegas, USA) homed in on the latest findings from the SWEDEPAD registry-based randomised controlled trials (RCTs), with panellists discussing the results with reference to the...
Vesalio recently announced two new US Food and Drug Administration (FDA) 510(k) clearances for its aspiration devices, designed for peripheral and neurovascular applications.
By integrating aspiration technology with its flagship mechanical retrieval platform, powered by proprietary Drop Zone and microfiltration...
Tara Mastracci (Barts Health NHS Trust, London, UK) advocated a comprehensive, three-armed approach to the future of personalised aortic dissection management at the 2025 Interdisciplinary Aortic Dissection Symposium (IADS; 11 September; London, UK).
Opening her talk, Mastracci defined personalised care...
RapidAI today announced the US Food and Drug Administration (FDA) clearance of Aortic Management, part of Rapid Aortic, which the company describes as "a comprehensive deep clinical AI solution designed to transform the acute assessment and longitudinal management...
A new US-wide poll announced today by Gore and Egg Medical sheds light on the growing concern for radiation exposure in operating rooms and Cath labs across the country.
The findings revealed that 100% of interventional cardiologists, 96% of interventional...
This advertorial is sponsored by Shockwave Medical.
Three vascular surgeons discuss how and when they deploy the transformative Shockwave Javelin first-of-its-kind Forward Intravascular Lithotripsy (IVL) Platform in cases of heavily calcified peripheral vascular occlusive disease.
Viewpoints on when and how to...
Vexev has announced the results of the CANSCAN trial evaluating the use of semi-autonomous ultrasound scanning utilising the VxWave ultrasound imaging system for arteriovenous fistula (AVF) mapping in dialysis clinics.
According to the company, the trial provides evidence that the...
ViTAA Medical today announced that the US Food and Drug Administration (FDA) has granted 510(k) clearance for AiORTA Plan, which the company describes as a "fully automated, hyper-precise aortic surgery planning solution".
The tool aims to automate key preoperative measurements...
Elucid has announced the launch of its PlaqueIQ image analysis software for the quantification and classification of plaque morphology in the carotid arteries. According to the company, as the “first and only” computed tomography (CT)-based plaque analysis software indicated for...
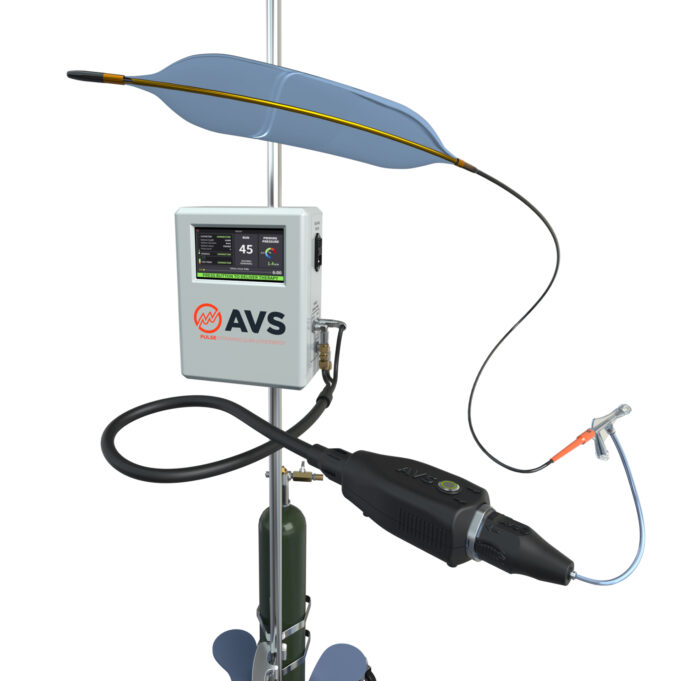
Christopher Metzger (Ballad Health CVA Heart Institute, Kingsport, USA) reported at the 2025 Vascular Interventional Advances (VIVA) conference (2–5 November, Las Vegas, USA) that the Pulse intravascular lithotripsy (IVL) system (Amplitude Vascular Systems) successfully treated severely calcified femoropopliteal disease...
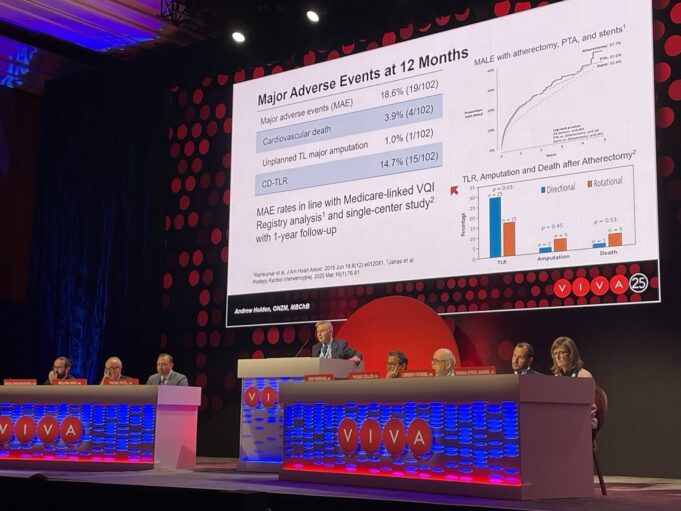
Results at 12 months from the FORWARD PAD investigational device exemption (IDE) and feasibility studies demonstrate low rates of major amputation and cardiovascular death in patients treated with the Javelin peripheral intravascular lithotripsy (IVL) catheter (J&J MedTech/Shockwave Medical). Andrew...
One-year outcomes in the ROADSTER 3 post-approval study to evaluate the safety and effectiveness of transcarotid artery revascularisation (TCAR) using the Enroute stent and neuroprotection system (Boston Scientific) in standard-surgical-risk patients revealed a 1.5% intention-to-treat rate of composite major...
Thirty-day results from the PERFORMANCE III study of integrated embolic protection (IEP) in transcarotid artery revascularisation (TCAR) demonstrate "very low" 30-day event rates. Sean Lyden (Cleveland Clinic, Cleveland, USA) shared this finding during a late-breaking trials session at the...
DISRUPT BTK II results out to 12 months demonstrate the strong safety profile and consistent effectiveness of the Shockwave peripheral intravascular lithotripsy (IVL) system (J&J MedTech/Shockwave Medical) for the treatment of calcified lesions below the knee (BTK) in patients...
Full results from the ENGULF pivotal study of the Hēlo thrombectomy system (Endovascular Engineering) in acute intermediate risk pulmonary embolism (PE) patients demonstrated a 0.95% major adverse event rate (MAE) at 48 hours and 25.1% reduction in right-to-left ventricular...
Three-year data from the LIFE-BTK randomised controlled trial, presented today at the 2025 Vascular Interventional Advances (VIVA) conference (2–5 November, Las Vegas, USA), demonstrate the prolonged efficacy and safety of the Esprit BTK drug-eluting resorbable scaffold (DRS; Abbott) in chronic...
Mechanical thrombectomy by means of computer-assisted vacuum thrombectomy (CAVT) using the 16Fr Lightning Flash system (Penumbra) along with anticoagulation for acute-to-intermediate pulmonary embolism (PE) demonstrated functional assessment improvements, earlier physiological recovery and greater reduction in pulmonary artery obstruction when...
“The question is less about should we offer AI education for vascular surgeons—I think most would agree that this will become necessary—and more about how and when it should be implemented, who should fund it, and who should...
InspireMD, developer of the CGuard Prime carotid stent system for the prevention of stroke, today announced the appointment of Peter Soukas (The Miriam Hospital and Brown University, Providence, USA) as chief medical officer.
“We are thrilled to have Dr Soukas...
Joseph Bavaria (Jefferson Health, Philadelphia, USA) speaks to Vascular News about a technique he says offers a “complete and total thoracic solution” for acute dissections, chronic dissections, and aneurysms. The technique in question: open zone 2 arch repair followed...
Former Society for Vascular Surgery (SVS) President Anton N. Sidawy, MD, FACS, DFSVS, became the 106th president of the American College of Surgeons (ACS) at the ACS Clinical Congress 2025 in Chicago earlier this month (Oct. 4–7).
A professor of...
In this issue:
SWEDEPAD reopens paclitaxel safety discussion, finds drug-coated devices do not improve outcomes
Opinion: “All patients need to be offered the chance to take part in a randomised controlled trial,” says Athanasios Saratzis (Leicester, UK)
Closer to the...
In this issue:
SWEDEPAD reopens paclitaxel safety discussion, finds drug-coated devices do not improve outcomes
Opinion: “All patients need to be offered the chance to take part in a randomised controlled trial," says Athanasios Saratzis (Leicester, UK)
Closer to...
Sharing 10-year findings from the ACST-2 trial at the 39th European Society for Vascular Surgery (ESVS) annual meeting (23–26 September, Istanbul, Türkiye), Alison Halliday (University of Oxford, Oxford, UK) highlighted that, if successful, both carotid artery stenting (CAS) and...
Surmodics has announced that results from a sex-specific analysis of 160 real-world acute, subacute, and chronic thrombotic limb ischaemia patients from its PROWL registry study were presented by Peter Monteleone (Dell Medical School at the University of Texas at...
The Centers for Medicare & Medicaid Services (CMS) has finalised its final National Coverage Determination decision, meaning both radiofrequency and ultrasound renal denervation for uncontrolled hypertension will now be covered under Medicare.
Medtronic and Recor Medical, both of which produce...
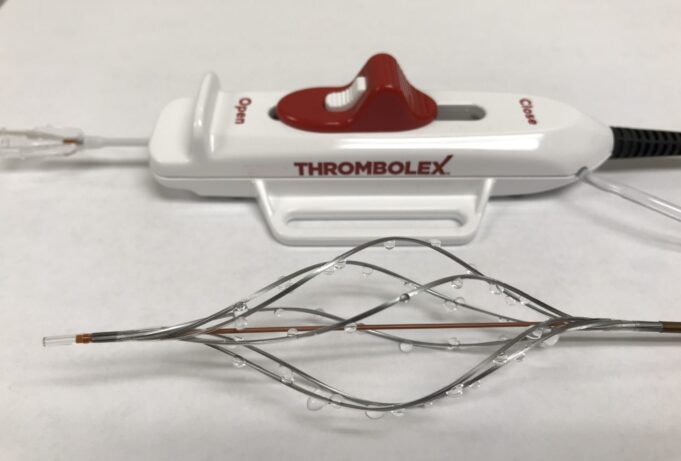
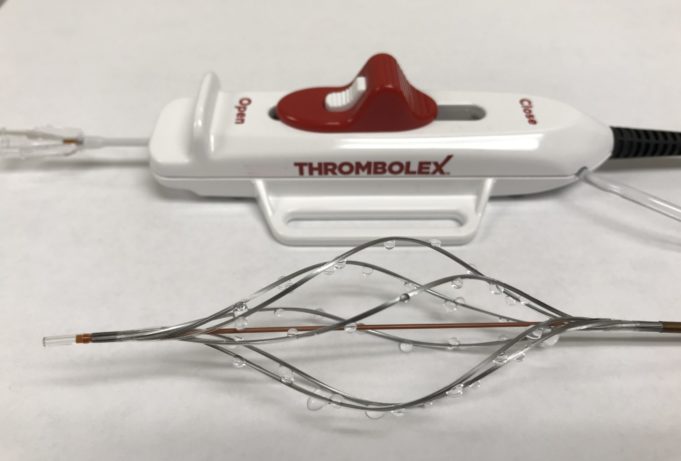
Prespecified interim analysis data from the first 50 patients in RAPID-PE have demonstrated "excellent safety" and "remarkably efficient lab times" in patients receiving on-the-table pharmacomechanical lysis without any post-procedure infusion with the Bashir endovascular catheter (Thrombolex) for the treatment...
The Freeman Hospital in Newcastle has become the first in the UK to implant the Esprit BTK everolimus-eluting resorbable scaffold system (Abbott), a press release reports.
The Esprit BTK system, which was approved for use in Europe in August, is...
Ahmed Eleshra (Mansoura, Egypt) writes on behalf of the Egyptian Vascular and Endovascular Society (EVES) board about the upcoming EVES Summit 2025 (29 October–1 November, Cairo, Egypt).
From 29 October to 1 November 2025, Cairo will once again take its place at the...
The use of mechanical thrombectomy, specifically computer-assisted vacuum thrombectomy (CAVT) using the 16Fr Lightning Flash system (Penumbra), with anticoagulation achieves superior reduction in right heart strain compared to anticoagulation therapy alone in patients with acute intermediate-high-risk pulmonary embolism (PE)....
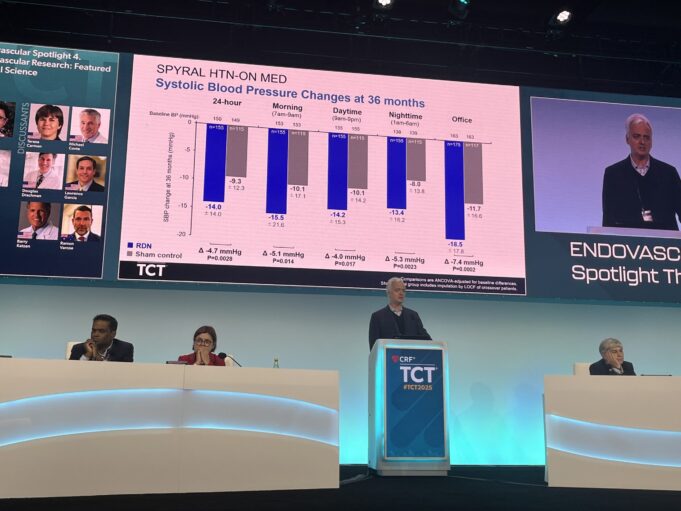
Medtronic has announced new, long-term results from its final report of the SPYRAL HTN-ON MED randomised clinical trial, showing that patients treated with the Symplicity Spyral renal denervation (RDN) procedure experienced significantly greater reductions in blood pressure compared to...
“How did an ineffective and costly intervention become routine care in the NHS ?” Ian Chetter (Hull York Medical School, University of Hull, Hull, UK) posed at the 39th European Society for Vascular Surgery (ESVS) annual meeting (23–26 September,...
Shockwave intravascular lithotripsy (IVL) “expands endovascular strategies for iliac occlusive disease and can facilitate a ‘leave nothing behind’ approach,” Stefano Fazzini (Tor Vergata University, Rome, Italy) and colleagues write in an open-access Journal of Vascular Surgery (JVS) paper highlighting...
In his latest Launch Pad column, Adam Talbot (Manchester, UK) writes about moral injury in vascular surgery, suggesting steps that could be taken to prevent it.
Vascular surgery often places surgeons in high-pressure situations where the right course of action...
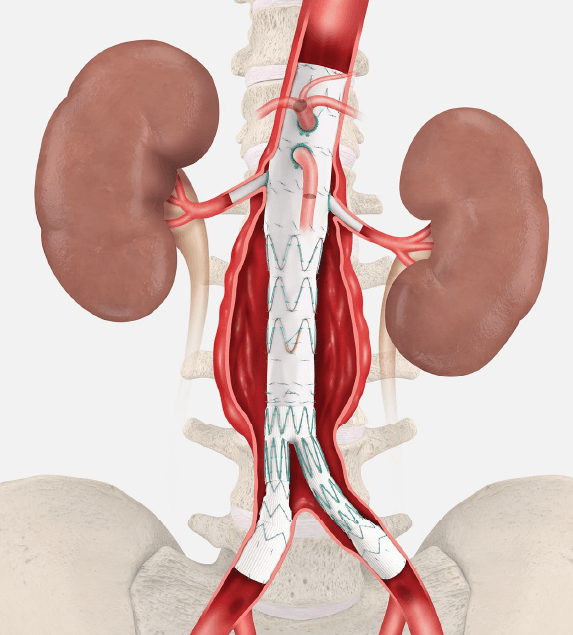
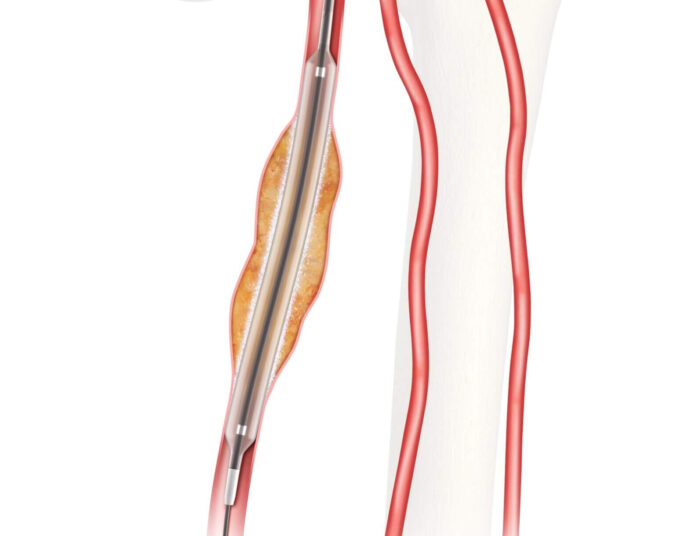
Shape Memory Medical has reached 50 per cent enrolment in its ongoing AAA-SHAPE pivotal trial, the company announced this week.
AAA-SHAPE is a prospective, multicentre, randomised, open-label trial to determine safety and effectiveness of the Impede-FX RapidFill device to...
Results from a multicentre randomised controlled trial (RCT) indicate that pre-emptive inferior mesenteric artery (IMA) embolisation during endovascular aneurysm repair (EVAR) does not significantly reduce aneurysm sac volume or rates of type II endoleak.
The CLARIFY IMA study—which the authors...
Nurea's PRAEVAorta 2 artificial intelligence (AI)-powered software has received US Food and Drug Administration (FDA) 510(k) clearance, a company press release reveals.
According to Nurea, PRAEVAorta 2 provides vascular physicians with advanced automated measurement tools for aortic diameters on contrasted...
Data from the AMDS PERSEVERE and PROTECT trials—both assessing the use of the Ascyrus medical dissection stent (AMDS, Artivion)—were presented in late-breaking science presentations at the 39th European Association for Cardio-Thoracic Surgery (EACTS) annual meeting (8–11 October, Copenhagen, Denmark).
The...
Medtronic recently announced it has received US Food and Drug Administration (FDA) labelling approval for the Endurant stent graft system, by adding ruptured abdominal aortic aneurysm (rAAA) clinical evidence and removing the rAAA treatment warning. A press release notes...
Getinge’s Advanta V12 covered stent system has received CE mark approval under the European Union Medical Device Regulation (EU MDR) for use as a bridging stent in fenestrated endovascular aneurysm repair (FEVAR), branched endovascular aneurysm repair (BEVAR), and iliac...
Researchers have shown that quiescent-inflow single-shot (QISS) magnetic resonance imaging (MRI) is able to identify more below-the-knee vessel segments than digital subtraction angiography (DSA) in patients with chronic limb-threatening ischaemia (CLTI). Taking first prize for best abstract, Alexander Crichton...
BD has announced the enrolment of the first patient in the XTRACT registry, a prospective, multicentre, single-arm, postmarket registry study designed to evaluate the real-world performance of the Rotarex catheter system in the treatment of patients with peripheral arterial...
Medtronic has announced the full distribution of the Neuroguard integrated embolic protection (IEP) system for carotid artery disease following its limited market distribution, the company announced. This follows the announcement earlier this year of an exclusive U.S. distribution agreement...
The Brazilian Health Regulatory Agency (ANVISA) recently approved Cardionovum's Aperto over-the-wire (OTW) drug-coated balloon (DCB) for commercial use in Brazil, a press release has revealed.
Cardionovum shares that this approval enables Brazilian patients undergoing dialysis to benefit from a therapy...
Optimal trial design for vascular access studies went under the microscope at the Vascular Access Society of Britain and Ireland (VASBI) 2025 annual scientific meeting (25–26 September, Bournemouth, UK), where speakers highlighted some of the challenges facing researchers in...
Cardionovum has announced that the APERTO 600 study, evaluating the efficacy of the Aperto over-the-wire (OTW) drug-coated balloon (DCB) in vascular access occlusion management, has been published in the Journal of Vascular Access.
APERTO 600 is a single-centre, retrospective cohort...
Microbot Medical, manufacturer and distributer of the Liberty endovascular robotic system, today announced that the Japanese Patent Office has granted the company its first patent, which covers the core Liberty system technology—a compact robotic device for driving and manipulating...
Terumo Aortic and Bentley today announced their partnership in a clinical study in the USA. The objective is to obtain US Food and Drug Administration (FDA) investigational device exemption (IDE) approval for Terumo Aortic’s fenestrated Treo device, in combination...
University Hospitals Plymouth (UHP) NHS Trust has become the first centre in the UK to treat patients with new laser catheter technology for vascular disease.
The interventional radiology (IR) and vascular surgery team at UHP are now able to treat...
FastWave Medical today announced the appointment of its principal investigators and steering committee for the upcoming investigational device exemption (IDE) pivotal trial of Artero, the company's peripheral electric intravascular lithotripsy (E-IVL) system.
Sahil Parikh, director of endovascular services at Columbia...
Health Canada has authorised the Esprit BTK everolimus-eluting resorbable scaffold system for the treatment of below-the-knee (BTK) chronic limb-threatening ischaemia (CLTI), Abbott revealed today in a press release.
Until this recent Health Canada approval for the Esprit BTK system, Abbott...
The European Vascular Course (EVC) recently announced the appointment of a new executive board. The event—held annually in Maastricht, The Netherlands—will now be led by a three-person team of vascular surgeons succeeding Michael Jacobs, who founded the event in...
Xeltis has announced clinical data from the aXess EU pivotal trial, the prospective study investigating the patency, safety, and performance of its restorative vascular access conduit— aXess—in adult patients with end-stage renal disease requiring vascular access to start or...
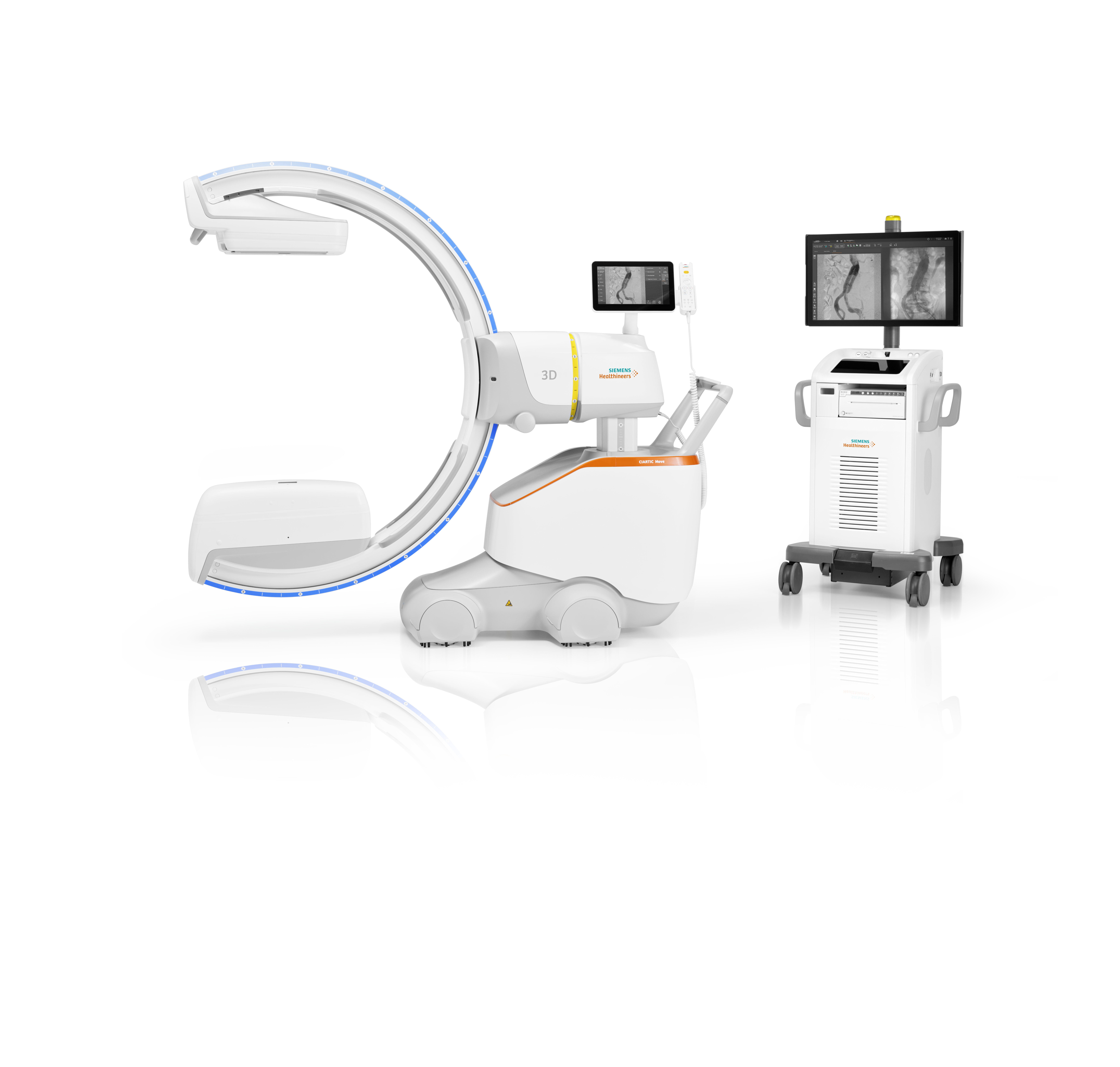
At the 2025 Charing Cross (CX) Symposium (23-25 April, London, UK), Michele Antonello (Padua, Italy) and Nikolaos Tsilimparis (Munich, Germany) sat down to discuss a key advancement in surgical imaging: CIARTIC Move. The innovative robotic C-arm boasts self-driving capabilities,...
Vascular News editorial board member Ross Milner (UChicago, Chicago, USA), the Louis Block professor of surgery and chief of vascular surgery at the University of Chicago, became the new president of the Midwestern Vascular Surgical Society (MVSS) in Cincinnati,...
Researchers have reported comparable five-year outcomes of the Gore Excluder iliac branch endoprosthesis (IBE; Gore) in both the investigational device exemption (IDE) and GREAT registry studies of the device.
Meghan Barber (University of Chicago, Chicago, USA) shared this main finding...
Sentante recently shared that the US Food and Drug Administration (FDA) has granted Breakthrough Device designation for the company's robotic stroke system.
A press release details that the Sentante stroke system allows for the remote delivery and manipulation of endovascular...
Terumo Aortic today announced what it describes as a "significant policy milestone" with the Centres for Medicare & Medicaid Services (CMS) establishing a new Medicare Severity Diagnosis-Related Group (MS-DRG), MS-DRG 209, for complex aortic arch procedures.
A company press release...
The CORE-MD—Coordinating Research and Evidence for high-risk Medical Devices—consortium has published new consensus recommendations in The Lancet Regional Health Europe, setting out scientifically robust methodologies for clinical investigations of high-risk medical devices.
The recommendations respond directly to a request from...
It was by chance rather than design that Dawn Coleman (Durham, USA) entered a career in vascular surgery, she tells Vascular News. Her unorthodox route into the profession, however, has not hampered Coleman’s rise to several senior positions. Now...
This advertorial is sponsored by Siemens Healthineers.
Speaking to Michele Antonello (University of Padua, Padua, Italy) at the 2025 Charing Cross (CX) Symposium (23–25 April, London, UK), Nikolaos Tsilimparis (Ludwig Maximilian University Hospital, Munich, Germany) highlighted the benefits of a...
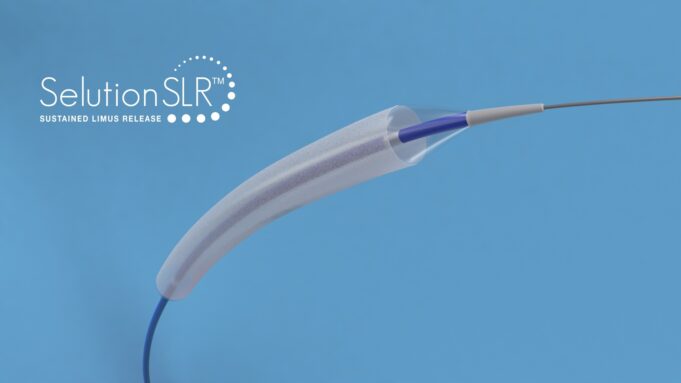
A first report from the SAVE prospective, multicentre, single-blinded randomised controlled trial of the Selution sustained limus release (SLR; Cordis) drug-eluting balloon (DEB) in patients with failing arteriovenous fistula (AVF) undergoing haemodialysis has demonstrated “significantly higher efficacy” compared with...
Positive 12-month outcomes for the Selution Sustained Limus Release (SLR; Cordis) drug-eluting balloon (DEB) were observed, with consistent haemodynamic, functional and clinical improvements irrespective of lesion location or Rutherford classification in both above-the-knee (ATK) and below-the-knee (BTK) arteries.
Michael Lichtenberg...
The Society for Vascular Surgery (SVS) recently published a comprehensive update to its clinical practice guidelines (CPGs) for the management of intermittent claudication (IC), urging clinicians to prioritise conservative treatment strategies and patient-centred care.
The new recommendations, which appeared in...
This article forms part of an educational supplement sponsored by Shockwave Medical. Explore the full Shockwave E8 and Javelin series here.
The Shockwave Javelin peripheral intravascular lithotripsy (IVL) catheter “bridges a critical gap” in the management of chronic limb-threatening ischaemia (CLTI) in patients with complex...
This article forms part of an educational supplement sponsored by Shockwave Medical. Explore the full Shockwave E8 and Javelin series here.
The Shockwave E8 catheter “significantly broadens the therapeutic landscape” for patients with diffuse below-the-knee (BTK) and below-the-ankle (BTA) disease. This is according to Marta...
This article forms part of an educational supplement sponsored by Shockwave Medical. Explore the full Shockwave E8 and Javelin series here.
Bruno Migliara (Peschiera del Garda, Italy) views the Shockwave E8 as an essential part of his treatment algorithm for complex calcified lesions in patients...
This advertorial, sponsored by Bentley, is only available in selected countries and geographies.
The story of iliac stenting in patients with TASC C and D lesions is one of evolution. From traditional self-expanding, bare metal stents to modern covered...
It was anounced today that Rebecca Middleton, chief executive officer (CEO) of the charity HBA Support, has founded and launched the Vascular Voice Network. The aim of the UK-based initiative, a press release reports, is to "break down silos...
Following a lively, three-pronged debate on the ‘gold standard’ in carotid revascularisation at this year’s Charing Cross (CX) Symposium (23–25 April, London, UK), a trio of vascular experts sat down in the CX Studio to discuss their favoured procedural...
Microbot Medical today announced that the US Food and Drug Administration (FDA) has granted 510(k) clearance for the Liberty system, the first FDA cleared single use, remotely operated robotic system for peripheral endovascular procedures, states the company in a recent press...
Shape Memory Medical today announced the completion of patient enrolment in the EMBO postmarket surveillance (EMBO-PMS) registry, the company’s prospective, multicentre registry of the Impede and Impede-FX embolisation plugs, and Impede-FX RapidFill device when used for peripheral vascular embolisation.
A...
The application of artificial intelligence (AI) is becoming an increasingly prominent topic on the agendas of key vascular meetings and the contents pages of vascular surgery journals. At this juncture, Vascular News looks at some of the recently shared...
Against the backdrop of an ageing population and increasing demand for complex endovascular aortic solutions, Santi Trimarchi (Fondazione IRCCS Cà Granda Ospedale Maggiore Policlinico, Milan, Italy) and a global team of experts recently authored a consensus statement on ascending...
Medtronic has announced it received approval in Japan from the Pharmaceuticals and Medical Devices Agency (PMDA) for its Symplicity Spyral renal denervation system for the treatment of resistant hypertension. The company will now initiate the process to obtain approval...
This educational supplement is sponsored by Shockwave Medical.
In this supplement:
Maria Antonella Ruffino (Lugano, Switzerland) explains how the Shockwave E8 and Shockwave Javelin peripheral intravascular lithotripsy (IVL) technologies are reshaping outcomes for patients with chronic limb-threatening ischaemia (CLTI).
Bruno Migliara...
The US Food and Drug Administration (FDA) recently announced that it has begun daily publication of adverse event data from the FDA Adverse Event Reporting System (FAERS).
The agency says that "this represents a significant step forward in modernising the...
This advertorial, sponsored by Gore, is only available in selected countries and geographies.
The Gore Viabahn VBX balloon-expandable endoprosthesis (VBX stent graft) has achieved an expanded indication as a bridging stent for fenestrated and branched endovascular aneurysm repair (F/BEVAR)....
In this issue:
Vascular surgery meets artificial intelligence (AI): Vascular News looks at some of the latest research and opinion on the topic
First official European Society for Vascular Surgery (ESVS) consensus statement consolidates expert opinion on "truly novel" ascending thoracic endovascular...
In this issue:
Vascular surgery meets artificial intelligence (AI): Vascular News looks at some of the latest research and opinion on the topic
First official European Society for Vascular Surgery (ESVS) consensus statement consolidates expert opinion on "truly novel"...
Drug-coated balloons and stents were not associated with reduced risk of amputation or improved quality of life compared with uncoated devices in the SWEDEPAD 1 and 2 trials. In addition, higher five-year mortality with drug-coated devices in patients with...
Cook Medical has enrolled the final patient in the global clinical study of its Zenith Fenestrated+ endovascular graft (ZFEN+), signifying completion of patient recruitment in the pivotal study.
The investigational ZFEN+ clinical study is being conducted under an Investigational Device Exemption...
Boston Scientific has recalled its Carotid Wallstent Monorail endoprosthesis owing to a “manufacturing defect” that has led to devices having an inner lumen that is smaller than specifications, causing resistance when withdrawing the stent delivery system.
A US Food and...
In a candid, cross-continental exchange, vascular surgeons Robert Shahverdyan (Hamburg, Germany) and Ari Kramer (Spartanburg, USA) share their real-world experiences using the FLEX Vessel Prep™ (VP) system (VentureMed) in arteriovenous (AV) access interventions.
From their initial impressions of using the...
Recor Medical has announced that the Paradise ultrasound renal denervation system has received approval for manufacturing and marketing in Japan for the treatment of resistant hypertension. This marks the first approval in the country for a medical device indicated...
This advertorial, sponsored by Abbott, is only available to readers in the USA.
“We now have something on label and FDA approved that we can use as a first-line therapy for tibial vessels,” Richard Pin (Southcoast Health, New...
At this year’s Charing Cross (CX) Symposium (23–25 April, London, UK), Manj Gohel (Cambridge University Hospitals, Cambridge, UK) spoke with Stephen Black (Guy’s and St Thomas’ Hospital, London, UK) about key takeaways from the venous programme.
Reflecting on the meeting,...
Abbott today announced it has received CE mark approval in Europe for the Esprit BTK everolimus-eluting resorbable scaffold system (Esprit BTK system), a technology designed for the treatment of below-the-knee (BTK) peripheral arterial disease (PAD).
Abbott notes that the Esprit...
The Shockwave Javelin peripheral intravascular lithotripsy (IVL) catheter “bridges a critical gap” in the management of chronic limb-threatening ischaemia (CLTI) in patients with complex multilevel disease. This is according to Ashish Patel and Narayanan Thulasidasan (London, UK), who here...
The Shockwave E8 catheter “significantly broadens the therapeutic landscape” for patients with diffuse below-the-knee (BTK) and below-the-ankle (BTA) disease. This is according to Marta Lobato (Barakaldo, Spain), who here presents a case in which the Shockwave E8 played a...
Bruno Migliara (Peschiera del Garda, Italy) views the Shockwave E8 as an essential part of his treatment algorithm for complex calcium in patients with below-the-knee (BTK) chronic limb-threatening ischaemia (CLTI), here outlining a case demonstrating his clinical experience with...
At this year’s Charing Cross (CX) Symposium (23–25 April, London, UK), two leading figures in the endovascular aneurysm repair (EVAR) field—Andrew Holden (Auckland City Hospital, Auckland, New Zealand) and Dai Yamanouchi (University of Wisconsin, Madison, USA)—sat down to discuss...
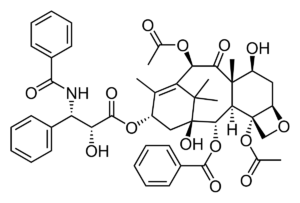
The US Food and Drug Administration (FDA) has granted approval for Cook Medical to initiate an investigational device exemption (IDE) study on the Advance Evero 18 everolimus-coated percutaneous transluminal angioplasty (PTA) balloon catheter, the company reported today.
The clinical...
Merit Medical Systems announced today the successful enrolment of the first patient in the WRAP North America registry.
The WRAP North America registry is designed to enrol up to 250 patients from the USA and Canada on haemodialysis who experience...
Strokes after transfemoral carotid artery stenting (TFCAS) were “the most disabling and lethal” when compared with transcarotid artery revascularisation (TCAR) and carotid endarterectomy (CEA) in a multicentre, retrospective analysis of Vascular Quality Initiative (VQI) data using modified Rankin Scale...
New guidelines from the American College of Cardiology (ACC) and the American Heart Association (AHA) for the prevention and management of high blood pressure have, for the first time, added renal denervation as a treatment option for patients with...
A post-hoc comparison of the BEST-CLI and PREVENT III multicentre prospective randomised controlled trials (RCTs) aimed at evaluating outcomes of vein bypass in chronic limb-threatening ischaemia (CLTI) patients over the past two decades found that they have “significantly improved”...
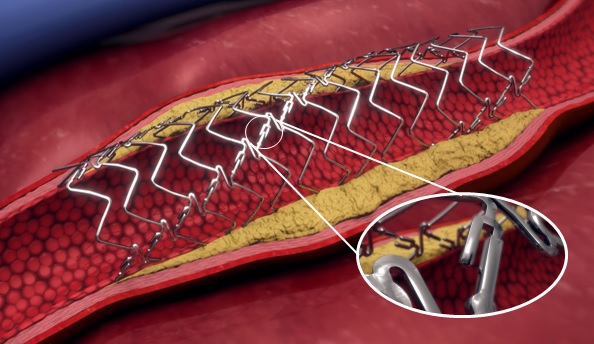
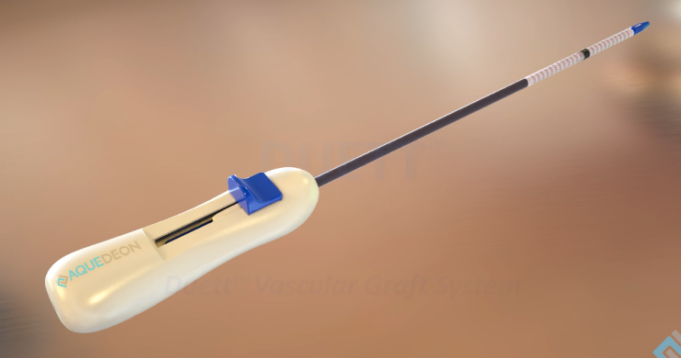
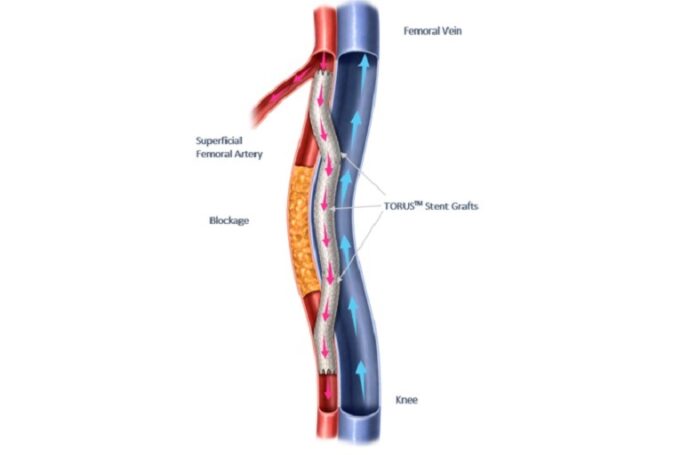
The US Food and Drug Administration (FDA) has approved the expansion of Aquedeon Medical's investigational device exemption (IDE) clinical trial evaluating the Duett vascular graft system, a company press release reveals. Duett is a novel device designed to facilitate open...
The implementation of a massive open online course (MOOC) has been shown to increase radiation safety behaviour in the hybrid operating room and decrease fluoroscopy time. This and other findings—including uncertainty around the course’s ability to reduce radiation dose—were...
InspireMD has announced that it has raised approximately US$58 million in combined gross proceeds through an equity private placement (PIPE) financing and the exercise of warrants that were triggered by the company’s recent premarket application approval (PMA) of the...
Ten-year data highlight the long-term efficacy and durability of the Endurant stent graft (Medtronic) in abdominal aortic aneurysm (AAA) patients who survived beyond five years post-enrolment in the ENGAGE registry. However, researchers also draw attention to the incidence of...
Vexev and US Renal Care have announced the successful completion of enrolment in the CANSCAN clinical trial.
The first-of-its-kind, multicentre feasibility study enrolled 120 patients with chronic kidney disease to evaluate the use of semi-autonomous ultrasound scanning utilising the VxWave...
Inari Medical—now part of Stryker—has today announced the launch of its next-generation InThrill thrombectomy system.
The company has stated in a recent press release that this is the first and only purpose-built small vessel and arteriovenous (AV) access thrombectomy system...
AngioDynamics today announced enrolment of the first patient in AMBITION BTK, a randomised study of the Auryon atherectomy system in patients with below-the-knee chronic limb-threatening ischaemia (CLTI).
A press release details that AMBITION BTK is a prospective, multicentre, randomised controlled...
The UK Medicines and Healthcare products Regulatory Agency (MHRA) has today announced "important" new steps to secure access for patients to the latest medical technologies available in Europe and other advanced countries.
As well as improving patient access to technologies,...
Getinge recently announced that it has received US Food and Drug Administration (FDA) premarket approval (PMA) for the iCast covered stent system. The device is now available as a bridging stent for the treatment of patients with aneurysmal disease.
Getinge...
Medtronic today announced that the first patient has been enrolled in the Peripheral Onyx liquid embolic (PELE) clinical trial, which will evaluate the safety and effectiveness of the Onyx liquid embolic system (LES) for embolisation of arterial haemorrhage in...
A five-year prospective cohort study—using longitudinal registry data from two population-based screening trials—has found that high-dose statin use was associated with decreased abdominal aortic aneurysm (AAA) growth rates and lowered risk of undergoing repair, rupture, and death. Researchers therefore...
Medtronic today announced it has entered into an exclusive US distribution agreement with Japan-based Future Medical Design Co (FMD) to sell specialty and workhorse peripheral guidewires. This agreement includes the first 400cm, 0.018" peripheral guidewire available in the USA,...
FastWave Medical has secured its eighth US patent for its electric intravascular lithotripsy (E-IVL) platform, Artero, reflecting the company's continued innovation in developing next-generation solutions for peripheral arterial disease (PAD) treatment, the company reports in a press release.
"IVL has...
Cook Medical has announced that its Zenith iliac branch device (ZBIS) is now commercially available in the USA with US Food and Drug Administration (FDA) approval as an endovascular treatment option for aortoiliac or iliac aneurysmal disease.
The company has...
The US Centres for Medicare & Medicaid Services (CMS) has issued its proposed National Coverage Determination (NCD) on renal denervation, recommending coverage for the therapy for the treatment of uncontrolled hypertension.
A 30-day public comment period is open for healthcare...
Geneticure has announced that the US Patent and Trademark Office has granted a patent for a genetic-based weighted algorithm model for predicting response to renal denervation.
This patent protects Geneticure’s proprietary method of using a patient’s genetic profile to predict...
“Spinal fluid drainage should be considered when the risk of spinal cord injury from the procedure is greater than the risk of severe complications from spinal fluid drainage.” This was one of the concluding findings of a study presented...
Why do rural adults and racial and ethnic minorities with vascular disease get major leg amputations more often? A new study published today in Epidemiology uses artificial intelligence (AI) to solve the mystery, finding an unaccounted-for factor that researchers think...
Gore today announced CE-mark approval of an expanded indication for the Gore Viabahn VBX balloon-expandable endoprosthesis (VBX stent graft) when used as a bridging stent with branched and fenestrated aortic endografts in the treatment of aortic aneurysms involving the...
Merit Medical recently announced the appointment of Martha Aronson as the company's new president and chief executive officer (CEO), effective 3 October 2025. Fred P Lampropoulos will remain as chairman of the board, president and CEO of Merit through...
Humacyte announced today that Symvess has been awarded Electronic Catalog (ECAT) listing approval from the US Defense Logistics Agency.
ECAT is an internet system that provides the Department of Defense (DOD) and other federal agencies with access to manufacturers’...
“We were really excited to know that the SVS is supporting our research,” Sujin Lee (Massachusetts General Hospital, Boston, USA) told Vascular News on receiving the SVS Foundation James S T Yao Resident Research Award for a paper examining...
Cook Medical has initiated a Class I recall of its Beacon Tip 5Fr angiographic catheter.
This follows reports of tip separation that could result in serious injury or death. The recall, identified as the most serious type by the...
Vivasure Medical has announced the submission of a premarket approval application to the US Food and Drug Administration (FDA) for its PerQseal Elite vascular closure system for arterial procedures.
In a recent press release the company has stated that...
Teleflex today announced that is has completed the previously announced acquisition of substantially all of the Vascular Intervention business of Biotronik.
The company notes in a press release that the acquisition adds a broad portfolio of therapeutic products to...
Society for Vascular Surgery (SVS) President-Elect Linda Harris (University at Buffalo, Buffalo, USA) extolled the values of “co-elevation” in her presidential address at the 7th annual Women’s Vascular Summit (2–3 May, Chicago, USA), invoking lessons on teamwork from the...
StentIt has announced the successful implantation of its Resorbable Fibrillated Scaffold (RFS). As part of the VITAL-IT 1 study, patients with chronic limb-threatening ischaemia (CLTI) below the knee have successfully been treated using the RFS implant. In this clinical...
Embolization recently announced it has recevied 510(k) clearance from the US Food and Drug Administration (FDA) for its Nitinol Enhanced Device (NED)—a vascular embolisation device intended for arterial and venous embolisation in peripheral vasculature.
Using proprietary shape-memory biocompatible polymers, Embolization's coil...
InspireMD has been granted premarket approval (PMA) from the US Food and Drug Administration (FDA) for its CGuard Prime carotid stent system in the USA—news that follows the company’s recent announcement of CE-mark approval under the European Medical Device...
Endovascular Engineering (E2) has completed patient enrolment in the pivotal cohort of its ENGULF trial, involving the Hēlo pulmonary embolism (PE) thrombectomy system.
The investigational device exemption study was carried out at 19 interventional cardiology, radiology, and vascular surgery centres...
Gore has announced that the Gore Tag thoracic branch endoprosthesis (TBE) is now approved by the US Food and Drug Administration (FDA) for use in Zones 0 and 1, expanding its indication for the endovascular repair of lesions in...
The findings of a new analysis from the STRIDE trial, presented at the American Diabetes Association (ADA) 2025 scientific session (20–23 June, Chicago, USA) and published in Diabetes Care, demonstrate the potential vascular benefits of semaglutide in patients with...
New regulations have come into effect in the UK which place a greater emphasis on medical device manufacturers to monitor the safety and performance of their products.
The new Post-Market Surveillance (PMS) regulations came into effect from Monday (16 June)...
Inquis Medical has announced that its Aventus thrombectomy system has received 510(k) clearance from the US Food and Drug Administration (FDA) for an expanded indication to treat pulmonary embolism (PE).
The Aventus System is a next-generation mechanical thrombectomy platform developed...
Penumbra has announced the completion of enrolment in the STORM-PE clinical trial.
The pivotal, prospective, multicentre randomised controlled trial enrolled 100 patients to evaluate computer assisted vacuum thrombectomy (CAVT) using Penumbra's Lightning Flash plus anticoagulation versus anticoagulation alone for the...
Can the world of vascular surgery return to the core therapeutic components of open surgery, unite them with the skillset brought by the endovascular revolution, and combine with a robotic surgery delivery system to create a third revolution in...
Jacques Busquet (Val d’Or Hospital, Paris, France) speaks to Vascular News about his life and career. A proponent of endovascular techniques—having co-founded the International Society of Endovascular Specialists (ISEVS) alongside Edward Diethrich—Busquet highlights the need to train the next...
Nectero Medical recently announced that the American Medical Association’s CPT editorial panel has approved two category III CPT codes applicable to the Nectero endovascular aneurysm stabilisation treatment (EAST) system procedure via a percutaneous or open approach. The company notes...
The four-branch Gore Excluder thoracoabdominal branch endoprosthesis (TAMBE) device demonstrated low mortality at one year with renal artery occlusion being the predominant device-related event associated with small renal arteries and pararenal aneurysms. So concluded Mark A Farber (University of...
“We're living in a new world order” when it comes to “no-option” chronic limb-threatening ischaemia (CLTI) patients, says Anahita Dua (Massachusetts General Hospital, Boston, USA). In recent years, into this arena has stepped transcatheter arterialisation of the deep veins...
An evaluation of trends in the utilisation of carotid artery stenting (CAS) versus carotid endarterectomy (CEA) has found there to be “no major change” to tip the balance following the 2023 US Centers for Medicare and Medicaid Services (CMS)...
Staged total endovascular aortic repair (TEAR) utilising arch branched and thoracoabdominal fenestrated and branched endografts is effective, but identified predictors of morbidity and mortality—including stroke—highlight the importance of individualised risk assessment to optimise outcomes. These are some of the...
Recent THRIVE study data show that Penumbra’s computer-assisted vacuum thrombectomy (CAVT) technology not only has the potential to improve outcomes for lower extremity acute limb ischaemia (LE-ALI) patients, but may also reduce healthcare resource use, thus potentially lowering overall...
Sherene Shalhub (Oregon Health and Science University, Portland, USA) set out a new biologically informed framework that “captures the diverse spectrum of arterial fragility and repair outcomes across inherited and acquired aortic disease” during today’s Roy Greenberg Distinguished Lecture...
Reflow Medical recently announced that the US Food and Drug Administration (FDA) has granted de novo clearance for the company’s Spur peripheral retrievable stent system, which is designed for the treatment of de novo or restenotic lesions following predilatation...
In this issue:
CX 2025: Cost-effectiveness outcomes defy expectations in latest BEST-CLI data drop
Reality check: Study brings “significant” gap between randomised and real-world vascular patient populations into sharp focus
In profile: Jacques Busquet (Paris, France)
Interview: Gustavo Oderich...
In this issue:
CX 2025: Cost-effectiveness outcomes defy expectations in latest BEST-CLI data drop
Reality check: Study brings “significant” gap between randomised and real-world vascular patient populations into sharp focus
In profile: Jacques Busquet (Paris, France)
Interview: Gustavo Oderich...
BD has announced plans to initiate a patient data registry for the Rotarex atherectomy system to measure real-world outcomes for patients with peripheral arterial disease (PAD).
Known as XTRACT, this prospective, multicentre, single-arm, post-market registry study will assess the clinical...
Terumo Interventional Systems has announced the early commercial availability of its US Food and Drug Administration (FDA)-approved Roadsaver carotid stent system. Indicated for use with Terumo’s Nanoparasol embolic protection system, Roadsaver is intended to treat carotid artery stenosis in...
Vascular News is pleased to announce Adam Talbot (Manchester, UK) as its new Launch Pad columnist, taking over from Sarah Sillito. In his first column, Talbot writes about “reframing success” in vascular surgery and embracing the increasingly palliative aspect...
Orwa Falah (Edinburgh Royal Infirmary, Edinburgh, UK), chair of the Edinburgh Aortic Summit (EAS), speaks to Vascular News ahead of this year’s inaugural meeting.
Falah explains that EAS 2025, set to be held 12–13 June in Edinburgh, UK, aims to...
Following in the footsteps of some of the pioneers of cardiovascular surgery, Gustavo Oderich recently stepped into position as the Michael E DeBakey distinguished chair in surgery, professor of surgery, chief of vascular and endovascular surgery and director for...
A retrospective, nationwide cohort study has revealed carotid endarterectomy (CEA) performed better than carotid artery stenting (CAS) in France over the course of a decade, while also highlighting the "overuse" of both procedures in the same period. Based on...
The randomised data foundation on which clinicians base decision-making for vascular surgery patients is derived from populations that do not represent the patients they see and treat on a day-to-day basis, a new research paper has found.
The review, recently...
A comment from Andrew Holden (Auckland City Hospital, Auckland, New Zealand) conveyed surprise at new cost-effectiveness data from the BEST-CLI trial that were shared at the 2025 Charing Cross (CX) Symposium (23–25 April, London, UK). The findings were revealed...
Endospan recently announced the presentation of 30-day results from the statistical dissection primary arm of the TRIOMPHE investigational device exemption (IDE) clinical study. The data were presented during the late-breaking trial session at the American Association for Thoracic Surgery...
On Tuesday 6 May, UK Member of Parliament (MP) Jim Shannon hosted the annual UK Parliamentary drop-in event for the All Party Parliamentary Group for Vascular and Venous Disease (VVAPPG).
The event brought together charities, patients, representative bodies, clinicians, industry partners, and Parliamentarians to...
iVascular has announced that its Luminor drug-coated balloon (DCB) is now available in Japan.
“This approval from the MHLW is evidence of the quality, safety, and efficacy of Luminor DCB,” said Lluis Duocastella, chief executive officer at iVascular. “We...
R3 Vascular recently announced that the first patient in its ELITE-BTK pivotal trial has been treated by Brian DeRubertis (New York-Presbyterian and Weill Cornell Medicine, New York, USA). The trial evaluates R3 Vascular’s next-generation drug-eluting bioresorbable scaffold, Magnitude, for...
In the largest systematic review and meta-analysis of atherectomy data to date, Thomas Zeller (Bad Krozingen, Germany) reported that the technique is effective in both calcified and non-calcified lesions but observed a trend toward lower target-lesion revascularisation (TLR) in...
The established ‘gold standard’ approach to carotid revascularisation remained enshrined as the go-to operation as far as the CX audience gathered in Theatre 3 for a carotid and acute stroke challenges great debate earlier today was concerned.
An overwhelming 76%...
Results from the Central blood pressure and variability evaluation in abdominal aortic aneurysm (AAA), or CAVE-ON, study, which sought to measure the impact of blood pressure control on AAA growth rates, found no significant association between growth and central...
The next spring forward in aortic stent grafting will be rooted in a greater understanding of the biomechanical forces acting upon devices alongside continued advances and specialisation in imaging, according to some of the founding fathers of endovascular aneurysm...
Results of the ACCESS 2 study, evaluating arteriovenous fistula (AVF) outcomes following use of the Sirogen (Vascular Therapies) sirolimus-eluting collagen implant, have shown that the device failed to meet non-inferiority for clinical fistula maturation compared to control at six...
In Wednesday's peripheral arterial programme, which hosted several podium-first presentations, lower limb drug-eluting technologies took centre stage—signalling a potential shift in the treatment paradigm for chronic limb-threatening ischaemia (CLTI). Investigators shared compelling new data from studies ranging from first-in-human...
Vivasure Medical has announced European CE mark approval of the PerQseal Elite vascular closure system, a sutureless and fully bioresorbable large-bore vessel closure device. The company plans to launch the product in select European markets this summer.
Leveraging Vivasure’s PerQseal...
The 40-year history of endovascular aneurysm repair (EVAR) of the aorta wends a circuitous route, from behind the Iron Curtain in Soviet Ukraine, to Buenos Aires, Argentina, points east and west in the United States, and far beyond.
It's...
New data and hot topic debates form the centrepiece of today’s (Wednesday’s) vascular access and renal interventions sessions at CX 2025, running throughout the full day in Theatre 3. Presentations will address critical challenges in current practice, including determining...
The Rouleaux Club—the UK’s national vascular trainee society—in association with the CX Symposium 2025 and BIBA Medical, has announced a new competition intended to educate the public on deep vein thrombosis (DVT) and pulmonary embolism (PE).
Entitled “The Swollen Leg”,...
More than 50 podium-first presentations headline the 47th Charing Cross (CX) International Symposium—opening today (Wednesday 23 April) at the ExCeL London—with the aim of addressing some of the most urgent challenges currently facing the vascular world. Led by co-chairs...
Venova Medical has announced the completion of patient enrolment in the company's VENOS-2 early feasibility study of the Velocity percutaneous arteriovenous fistula (pAVF) system for haemodialysis access.
The multicentre study is being performed under an Investigational Device Exemption (IDE) granted...
Issue highlights:
CX 2025 preview: Global vascular community is London bound for Challenges update
A year in profiles: Highlights from interviews with Bijan Modarai, Kevin Mani, Ramon Varcoe and Vincent Rowe
Does Medtronic’s decision to discontinue offering Ellipsys signal the end of endoAVF?
Multidisciplinary,...
Issue highlights:
CX 2025 preview: Global vascular community is London bound for Challenges update
A year in profiles: Highlights from interviews with Bijan Modarai, Kevin Mani, Ramon Varcoe and Vincent Rowe
Does Medtronic's decision to discontinue offering Ellipsys signal...
Terumo Neuro has announced that its carotid stent system has received premarket approval (PMA) from the US Food and Drug Administration (FDA).
This milestone marks the first dual-layer micromesh carotid stent approved in the USA, offering physicians a clinically proven option...
A multi-institutional analysis has demonstrated fenestrated endovascular aneurysm repair (FEVAR) could be a “safer and more durable option” than chimney EVAR (ChEVAR) in elective juxtarenal abdominal aortic aneurysm (AAA) cases with suitable anatomy, offering potentially “greater generalisability to real-world...
A recent partial regulatory device approval made fenestrated and branched endovascular aneurysm repair (F/BEVAR) a key topic at this year’s Japanese Society for Cardiovascular Surgery (JSCVS) annual meeting (20–22 February, Shimonoseki and Moji City, Japan), writes Hiroshi Mitsuoka (Shizuoka, Japan).
The modernisation...
Cagent Vascular has announced its first patient enrolment in the Serranator versus plain balloon angioplasty optical coherence tomography (OCT) study.
This prospective, randomised (2:1 treatment to control) dual-centre study will enrol up to 60 patients. The study will utilise intravascular...
Microbot Medical has shared that it presented for the first time the data from its ACCESS-PVI pivotal trial at the Society of Interventional Radiology (SIR) annual scientific meeting (29 March–2 April, Nashville, USA).
The study was performed at three...
Speaking to Vascular News at the 28th European Vascular Course (EVC; 9–11 March, Maastricht, The Netherlands), Kevin Mani (Uppsala, Sweden) addresses the issue of aortic infection, which he says represents a “humongous challenge for the patient, especially when it...
Surmodics has announced the commercial release of the Pounce XL thrombectomy system, the latest in a suite of Pounce thrombectomy systems that provide endovascular removal of acute or chronic clot from peripheral arteries.
Intended for removal of thrombi and emboli...
Gore recently announced the expansion of the Gore Tag conformable thoracic stent graft with Active Control system product line, following US Food and Drug Administration (FDA) approval of four new large-diameter tapered designs: 34x28mm, 37x31mm, 40x34mm and 45x37mm.
News of...
Barend Mees (Maastricht, The Netherlands), arterial programme director for the European Vascular Course (EVC), outlines several highlights from this year’s meeting (9–11 March, Maastricht, The Netherlands).
Mees notes that the 2025 programme touched on some lesser considered topics such as...
Charting the development of the Charing Cross (CX) venous and lymphatic programme from a sidebar event to becoming an “integral part” of the meeting’s agenda, executive board member Stephen Black (Guy’s and St Thomas’ NHS Foundation Trust and King’s...
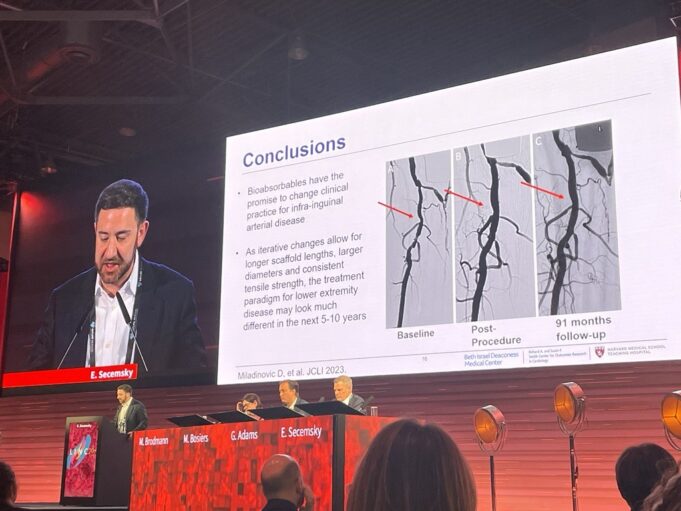
“These crucial results will help to shape next steps in research, including ongoing randomised controlled trials comparing bioresorbable scaffolds to angioplasty, and eventually, to drugeluting stents.” These are the words of Michel Bosiers (University Hospital Bern, Bern, Switzerland), sharing...
Semaglutide significantly improved maximal walking distance in people with symptomatic peripheral arterial disease (PAD) and type 2 diabetes in a trial designed to evaluate the use of a GLP-1 agonist to manage PAD.
In addition to improvements in walking ability...
Cook Medical has announced that its Zilver PTX drug-eluting stent (DES) has lower rates of in-stent occlusions among patients with restenosis at three years than the Eluvia DES (Boston Scientific), according to real-world data from the REALDES study. The company notes that the data, published by Tsuyoshi...
Evident Vascular has announced the successful closing of its Series B financing with new investors Shangbay Capital and two undisclosed multinational strategics joining founding investor Vensana Capital.
A press release details that the funding will accelerate development of the...
Merit Medical Systems has announced that the six-month results from the randomised arm of the Wrapsody arteriovenous access efficacy—WAVE—trial are scheduled for publication in the April issue of Kidney International, ahead of the presentation of 12-month results from the...
The importance of radiation safety training for vascular surgery trainees was highlighted by a recent study that detailed a learning curve effect between first- and second-year fellows performing fluoroscopically guided interventions.
Using optically stimulated dosimeters fitted outside and under lead...
Vascular News editorial board members Ian Loftus (London, UK) and Ross Milner (Chicago, USA) are joined by Dittmar Böckler (Heidelberg, Germany), Colin Bicknell (London, UK) and Tilo Kölbel (Hamburg, Germany) to share their expert insights on a range of topics, including:
Thresholds and indications for intervention
Current clinical trials...
Ellen Dillavou (WakeMed Heart Center, Raleigh, USA) asks whether the high bar for evidence being sought to bring new de novo devices to market is stifling innovation in the field of vascular access.
In the last decade we have seen...
Imperative Care today announced US Food and Drug Administration (FDA) 510(k) clearance of the 82cm version of its Symphony 16Fr catheter, the company’s latest innovation designed to elevate care for patients with venous thrombosis.
“Thrombectomy procedures have emerged as one...
Lorenzo Patrone (London, UK), course director of the Find Your Algorithm (FYA) congress, speaks to Vascular News about the highlights of this year’s meeting (27 February–1 March, London, UK).
In particular, Patrone points to a lecture on endovascular aneurysm repair...
Vascular News editorial board members Nicholas Inston (Birmingham, UK; CX executive board member) and Bart Dolmatch (Palo Alto, USA) are joined by Kate Steiner (Stevenage, UK; CX executive board member) to share their expert insights on a range of...
Tune in on Monday, 24 March, at 15:15 GMT / 16:15 CET / 11:15 ET / 10:15 CST for a Vascular News live broadcast on topical issues related to the management of aortic disease.
Vascular News editorial board members Ian Loftus (London, UK) and Ross...
Janet Powell, the clinical trial investigator who played a pivotal role in several landmark abdominal aortic aneurysm (AAA) studies, has died at the age of 79. At the time of her death, Powell was professor of vascular biology...
Life Seal Vascular has announced the appointment of Matt Thompson as chief executive officer (CEO).
A company press release has stated that Thompson’s career as a vascular surgeon and medical device executive positions Life Seal Vascular to accelerate the...
Xeltis has today announced the appointment of clinical nephrology experts An De Vriese and Haimanot (Monnie) Wasse to its medical advisory board.
De Vriese is currently head of the division of nephrology and infectious disease in AZ Sint-Jan Brugge, in...
Speaking to Vascular News at the 28th European Vascular Course (EVC 2025; 9–11 March, Maastricht, The Netherlands), Maarten Snoeijs (Maastricht, The Netherlands) gives an update on the ongoing OASIS randomised controlled trial.
OASIS is set to determine the best strategy...
Vascular News editorial board members Robert Morgan (London, UK) and Eric Secemsky (Boston, USA) are joined by Marianne Brodmann (Graz, Austria), Naseer Ahmad (Manchester, UK), Thomas Zeller (Bad Krozingen, Germany) and Brian DeRubertis (New York, USA) to share their...
Reflow Medical has announced the opening of its European subsidiary in Landsberg am Lech, Germany. This strategic expansion strengthens the company’s international presence and enhances its ability to serve markets outside the USA, a press release notes.
Reflow Medical Europe...
Inari Medical, now part of Stryker, recently announced the launch of its Artix thrombectomy system. Purpose-built for the distinct needs of the peripheral arterial system, Artix is a combined aspiration plus mechanical thrombectomy solution that delivers procedural control and...
Philips has recently announced that it will no longer sell its Tack endovascular system in the USA following a Class I recall issued by the US Food and Drug Administration (FDA).
A Class I recall is the FDA’s most serious...
Today, Shockwave Medical announced the US launch of its Shockwave Javelin peripheral intravascular lithotripsy (IVL) catheter, a platform designed to modify calcium and cross narrowed vessels in patients with peripheral artery disease (PAD).
PAD is the narrowing or blockage...
Stereotaxis has announced a US Food and Drug Administration (FDA) regulatory submission for the first robotically navigated catheter designed to expand usage of robotic magnetic navigation into the broader endovascular field.
Emagin 5F is the first in a family of...
DeepQure has announced progress of clinical trials of its extravascular renal denervation device, HyperQure.
The company is actively conducting clinical trials in both South Korea and the USA, which it says demonstrate “promising” results. In South Korea, seven patients have...
Boston Scientific has entered into a definitive agreement to acquire SoniVie Ltd, a privately held medical device company that has developed the TIVUS intravascular ultrasound system. An investigational technology, the TIVUS system is designed to denervate nerves surrounding blood...
Cook Medical’s Zenith Alpha 2 (ZTA2) thoracic endovascular graft is now available throughout the United States. A company press release states ZTA2 is the first aortic device on the market that has evolved to accommodate CO2 flushing and has an...
Teleflex today announced it has entered into a definitive agreement to acquire Biotronik’s Vascular Intervention business.
The acquisition reflects Teleflex’s commitment to investing in the estimated US$10 billion interventional cardiology and peripheral vascular market. A company press release states that...
Robert Shahverdyan (Hamburg, Germany) writes for Vascular News on the technologies shaping the future of the field of percutaneous arteriovenous fistula (AVF) creation for patients requiring haemodialysis, following his recent address at the 2025 Leipzig Interventional Course (LINC 2025;...
Humacyte today announced the commercial launch of Symvess (acellular tissue engineered vessel-tyod) for use in adults as a vascular conduit for extremity arterial injury when urgent revascularisation is needed to avoid imminent limb loss, and when autologous vein graft...
Shape Memory Medical today announced the first European enrolment in the AAA-SHAPE pivotal trial, the company’s prospective, multicentre, randomised, open-label trial to determine the safety and effectiveness of the Impede-FX RapidFill device to improve abdominal aortic aneurysm (AAA) sac...
In a Charing Cross (CX) Live discussion, filmed at the 2024 CX Symposium, Andrew Holden (Auckland, New Zealand) and Eric Secemsky (Boston, USA) pinpoint several salutary lessons that can be taken from the now resolved paclitaxel controversy that was...
Stryker announced today that it has completed the acquisition of Inari Medical. A press release notes that the addition of Inari brings an established peripheral vascular position to Stryker in the fast-growing venous thromboembolism (VTE) segment.
“The acquisition of Inari...
Neeraj Bhasin (Huddersfield, UK), chair of the Circulation Foundation (CF), highlights recent efforts from the charity to raise awareness and funds for research into the prevention and treatment of vascular disease.
The CF was formed in 1992 and is the...
Solaris Endovascular has announced an enrolment milestone in its DEScover clinical trial, assessing a sirolimus-eluting covered stent graft—Solaris DE—for the treatment of patients facing challenges with dialysis access dysfunction and peripheral arterial disease (PAD).
In a press release, Solaris Endovascular...
Critical challenges in open and endovascular treatment of aortic disease will be brought into focus when world leaders in cardiac, aortic, and vascular therapies return to London this spring for the 47th annual Charing Cross (CX) Symposium 2025 (23–25...
InspireMD and North American Science Associates (NAMSA) today announced that, pursuant to a previously announced strategic outsourcing partnership, the companies are working together to conduct the CGUARDIANS II pivotal study of InspireMD’s CGuard Prime 80cm carotid stent system for use...
Nada Elzefzaf writes about the inaugural Women in Vascular Forum that took place at the recent Egyptian Vascular and Endovascular Society (EVES) 2024 Summit (30 October–2 November, Cairo, Egypt). The session, Elzefzaf shares, “marked a pivotal moment in the...
A new guideline from the American Society of Echocardiography (ASE) aims to provide more detail for clinicians performing ultrasound-guided vascular cannulation.
Guidelines for Performing Ultrasound-Guided Vascular Cannulation: Recommendations of the American Society of Echocardiography is published in the February 2025...
A US$5-million gift from Lorraine and Bill Dodero is set to establish the eponymously named Lorraine and Bill Dodero Limb Preservation Center at University Hospitals (UH) Harrington Heart & Vascular Institute in Cleveland, USA. Led by interventional cardiologist Mehdi...
Despite a “dramatic” decrease in the use of prophylactic cerebrospinal fluid drains (CSFDs) for the prevention of spinal cord ischaemia (SCI) in patients undergoing fenestrated and branched endovascular aortic repair (F/BEVAR), the rate of SCI has tracked a similarly...
A new initiative from Abbott hopes to increase awareness of peripheral arterial disease (PAD) in the UK with the aim of preventing unnecessary amputations. Following the launch of the campaign, Samih Al Mawass, divisional vice president of Abbott's vascular...
Thirty-day results from the ROADSTER 3 study have demonstrated that transcarotid artery revascularisation (TCAR) is a safe and effective approach in patients who are deemed to be ‘standard risk’ for experiencing adverse events related to carotid endarterectomy (CEA). These...
Gustavo Oderich (Houston, USA) speaks to Vascular News about non-radiation-based imaging, highlighting various technologies that he says have the potential to change the game.
“We definitely need to away from radiation,” the newly appointed professor of surgery at Baylor...
Having initially been geared towards a career in engineering, Vincent Rowe was ultimately drawn to medicine’s dual offering of intellectual stimulation and interpersonal engagement. He overcame a fear of the sight of blood to pursue what has so far...
While “vessel preparation” is mandatory for optimal peripheral arterial disease (PAD) treatment outcomes, especially in the context of endovascular intervention, a clear definition of the term, is still lacking. So says Fabrizio Fanelli (Careggi University Hospital, Florence, Italy) at...
Michele Piazza (Padua, Italy) advises how to manage the challenging anatomical scenario of a narrow aorta during fenestrated and branched endovascular aneurysm repair (F/BEVAR), highlighting accurate, patient-specific planning and use of the correct bridging stent technology as the keys...
Surmodics has announced the successful early clinical use of its Pounce XL thrombectomy system. The device received US Food and Drug Administration (FDA) 510(k) clearance in September 2024, and is currently in limited market release (LMR), with full commercial...
Cagent Vascular has announced the launch of the Serranator SL-PRO percutaneous transluminal angioplasty (PTA) serration balloon catheter for chronic limb-threatening ischaemia (CLTI) and pedal disease.
A press release notes that Serranator SL-PRO utilises Cagent’s proven serration technology on a redesigned...
iVascular has shared that Mariano Palena (Policlinico Abano Terme, Abano Terme, Italy) presented the results of an observational and prospective registry on the use of the iVolution Pro self-expandable stent in the flexion zone at this year's Leipzig Interventional...
Koen Deloose (AZ Sint Blasius Hospital, Dendermonde, Belgium) presented new sub-cohort analyses from the TINTIN trial at this year's Leipzig Interventional Course (LINC 2025; 28–30 January, Leipzig, Germany), offering deeper insights into the long-term outcomes for different types of...
Pending the results of several trials and “iterative changes” to device design, bioabsorbable scaffolds are set to change the treatment paradigm for lower extremity peripheral arterial disease (PAD) within the next decade. So concluded Eric Secemsky (Beth Israel Deaconess...
AngioDynamics today announced the initiation of AMBITION BTK, a randomised study of the Auryon atherectomy system in the treatment of below-the-knee (BTK) chronic limb-threatening ischaemia (CLTI).
AMBITION BTK is a multicentre, randomised controlled trial (RCT) designed to evaluate the clinical...
After the official launch of the BeFlow earlier this week, Bentley has announced that it successfully launched two additional products at the Leipzig Interventional Course (LINC 2025; 28–30 January, Leipzig, Germany): the BeGraft stent graft system and the BeFlared...
Cagent Vascular has announced the start of the POINT FORCE registry, a postmarket clinical follow-up study of the Serranator percutaneous transluminal angioplasty (PTA) serration balloon catheter.
This prospective, multicentre, single-arm study will enrol a minimum of 500 patients at up...
Amplitude Vascular Systems (AVS) recently announced that it has completed a Series B round of financing of US$36 million. The funding will support the US peripheral commercial launch as well as US coronary and carotid investigational device exemption (IDE)...
New data from a large, real-world study support the use of drug-eluting devices to reduce amputations, readmissions, and healthcare costs in the treatment of peripheral arterial disease (PAD), Marianne Brodmann (Medical University of Graz, Graz, Austria) informed attendees at...
Bentley today announced the market launch of its BeFlow iliac covered stent system at the Leipzig Interventional Course (LINC 2025; 28–30 January, Leipzig, Germany).
A press release notes that the BeFlow has been designed for the treatment of iliac...
Gore recently announced the first commercial use of its Excluder conformable abdominal aortic aneurysm (AAA) endoprosthesis with Active Control system in Canada.
The news coincides with Health Canada approval of the device for the treatment of AAA patients with aortic...
Vincent Jongkind (Amsterdam, The Netherlands) discusses the current guidelines on left subclavian artery (LSA) preservation, whether coverage of the LSA can result in an increase of neurological complications, and what advantages can be gained from having an off-the-shelf endovascular...
Artivion has announced data from the AMDS PERSEVERE clinical trial presented in a late-breaking science presentation at the Society of Thoracic Surgeons (STS) 2025 annual meeting (25–27 January, Los Angeles, USA). The data include clinical outcomes across the full...
Following a session on sexual misconduct in surgery delivered at the 2024 Vascular Society of Great Britain and Ireland (VSGBI) annual scientific meeting (27–29 November, Brighton, UK), Ian Chetter (Hull, UK), Andrew Garnham (Wolverhampton, UK) and Mei Nortley (Oxford,...
Front Line Medical Technologies has announced the UK launch of COBRA-OS, which the company claims is the world’s smallest aortic occlusion device, designed to save lives by rapidly controlling critical bleeding. The COBRA-OS has received approval for sale in...
Concept Medical has announced the successful completion of patient enrolment in the SirPAD trial, with over 1,250 patients now enrolled.
A press release notes that the SirPAD (Major adverse limb events in patients with femoropopliteal and below-the-knee peripheral arterial disease...
The US Food and Drug Administration (FDA) has issued a statement outlining measures to enhance protections against medical device shortages.
According to Michelle Tarver, director of the FDA’s Center for Devices and Radiological Health (CDRH), supply chain disruptions—often caused...
In this issue:
One-year SAVVE trial results are ‘most encouraging data ever produced for a bioprosthetic vein valve’
New data support role of F/BEVAR for treatment of thoracoabdominal aortic aneurysms
In profile: Vincent Rowe (Los Angeles, USA)
Laura Capoccia...
In this issue:
One-year SAVVE trial results are 'most encouraging data ever produced for a bioprosthetic vein valve'
New data support role of F/BEVAR for treatment of thoracoabdominal aortic aneurysms
In profile: Vincent Rowe (Los Angeles, USA)
Laura Capoccia...
This advertorial is sponsored by Terumo Aortic.
On 3 October 2024, a significant milestone was achieved as Terumo Aortic announced the first implant in the world using the TREO FIT system. The procedure was performed by Benjamin W Starnes (Harborview...
This advertorial is sponsored by Shape Memory Medical.
A wide variety of vessels treated with no device-related adverse events is a key recent finding to emerge from the EMBO postmarket surveillance (EMBO-PMS) registry.
EMBO-PMS is a prospective, multicentre registry of the...
This advertorial, sponsored by Bentley, is only available in selected countries and geographies.
In November 2024, vascular surgeon Stéphan Haulon (Hôpital Marie Lannelongue, Paris, France) successfully performed the first-in-human implantation of Bentley’s recently CE-certified BeFlared fenestrated endovascular aneurysm repair...
Concept Medical has successfully enrolled the first patient in the MAGICAL BTK US investigational device exemption (IDE) pivotal trial, a press release reveals.
Following the recent IDE approval by the US Food and Drug Administration (FDA) for the MagicTouch PTA,...
The European Society for Vascular Surgery (ESVS) has developed new guidelines for the care of patients with vascular trauma. The 2025 clinical practice guideline document was recently published as an open-access article in press in the European Journal of...
A recent prospective, multicentre cohort study provides insights into early and late aortic-related mortality and rupture after fenestrated and branched endovascular aneurysm repair (F/BEVAR) of thoracoabdominal aortic aneurysms (TAAAs). Researchers claim these are likely to represent the most comprehensive...
Laura Capoccia (Frosinone, Italy) writes about vascular surgeons’ role in acute stroke management and the importance of collaboration with other specialties in this setting.
While stroke treatment has dramatically changed in recent years, still around 795,000 people experience a new...
The Centers for Medicare & Medicaid Services (CMS) is opening a national coverage analysis (NCA) on renal denervation, with a view to reviewing and developing a national Medicare coverage policy for the therapy for patients with hypertension.
The expected completion...
AngioDynamics has announced the publication of the results from the Acute Pulmonary Embolism Extraction Trial with the AlphaVac system—APEX-AV—in the Journal of the Society for Cardiovascular Angiography & Interventions (JSCAI).
The primary efficacy endpoint of the APEX-AV trial was the...
Inquis Medical has announced completion of patient enrolment in its AVENTUS clinical trial, a pivotal investigational device exemption (IDE) trial evaluating the safety and efficacy of the company’s Aventus thrombectomy system for the treatment of pulmonary embolism (PE).
A total...
Medtronic announced today that it has entered into an exclusive US distribution agreement with Contego Medical. Under the agreement, Medtronic will be the sole US distributor for Contego’s portfolio of commercially available products, which provide revascularisation treatment for carotid...
R3 Vascular today announced that it has received WCG Institutional Review Board (IRB) approval for the ELITE-BTK pivotal trial of its Magnitude drug-eluting next-generation bioresorbable scaffold for below-the-knee (BTK) peripheral arterial disease (PAD). This approval allows the trial to...
Gore has announced recent CE mark of a lower profile Viabahn VBX balloon expandable endoprosthesis (VBX stent graft).
Medical Device Regulation (MDR) approval of this innovation builds on a proven device which has become an important tool for treating complex...
In conversation with Vascular News about IVUS, Eric Secemsky (Beth Israel Deaconess Medical Center, Boston, USA, and a newly appointed editor of Vascular News North America) drilled down on the benefits of this technology in the vascular field.
Providing some...
Marianne Brodmann (Medical University of Graz, Graz, Austria) shared her opinions on some of the burning debates in peripheral arterial disease (PAD) treatment at the Paris Vascular Insights course (PVI; 12–14 December, Paris, France).
On the ongoing competition between sirolimus...
Fabrizio Fanelli (Careggi University Hospital, Florence, Italy) tells Vascular News that artificial intelligence (AI) has tremendous potential to help refine which treatment modality and which device can be useful for specific types of patients or lesions. Fanelli was speaking...
Stéphan Haulon, head of Cardiac and Vascular Surgery, Hôpital Marie Lannelongue, Paris, France, one of the co-directors of the Paris Vascular Insights course (PVI, 12–14 December, Paris, France) spoke to Vascular News about artificial intelligence (AI) in vascular surgery now...
Rachael Forsythe (Edinburgh, UK) writes about an “enriching and rewarding” trip across the USA as the 2024 American College of Surgeons (ACS) John D Corson international guest scholar and encourages other vascular surgeons to apply for the fellowship.
The ACS...
Argon Medical announces the first patient enrolment in the CLEAN-PE study. The prospective, multicentre CLEAN-PE study aims to evaluate the safety and efficacy of the Cleaner Pro thrombectomy system for removing blood clot from the lungs in patients diagnosed...
A retrospective study has indicated that carotid artery stenting (CAS)—when performed by fellowship‐trained neurointerventional physicians in comprehensive stroke centre (CSC) settings—was associated with a lower complication rate as compared to prior literature on CAS, carotid endarterectomy (CEA) and transcarotid...
Boston Scientific has announced it has entered into a definitive agreement to acquire Bolt Medical, the developer of an intravascular lithotripsy (IVL) advanced laser-based platform for the treatment of coronary and peripheral artery disease.
“Representing one of the fastest growing...
A new Vascular Quality Initiative (VQI) data analysis, recently published in the European Journal of Vascular and Endovascular Surgery (EJVES), has found no statistically significant differences between three carotid endarterectomy (CEA) shunting strategies regarding in-hospital stroke and death rate,...
Endologix today announced a leadership transition. Matt Thompson will step down as president and chief executive officer (CEO) and take on a new role as executive vice president and chief medical officer. John Liddicoat has been appointed as the...
The US Food and Drug Administration (FDA) has issued draft guidance that includes recommendations to support development and marketing of safe and effective artificial intelligence (AI)-enabled devices.
The guidance, if finalised, would be the first guidance to provide comprehensive...
A recent analysis of over 270,000 Medicare fee-for-service beneficiaries has found an increase in adverse outcomes and death after a US Food and Drug Administration (FDA) warning led to decreased use of paclitaxel-coated devices for peripheral revascularisation procedures.
Writing in...
Xeltis has announced the completion of enrolment in its EU pivotal trial for aXess, its restorative vascular access conduit, in adults with end-stage renal disease. The trial is taking place in 22 centres across Europe.
The aXess EU pivotal trial...
Stryker has announced a definitive agreement to acquire all of the issued and outstanding shares of common stock of Inari Medical for US$80 per share in cash, representing a total fully diluted equity value of approximately US$4.9 billion.
A Stryker...
Humacyte recently shared that the US Food and Drug Administration (FDA) has granted a full approval for the Symvess acellular tissue-engineered vessel.
A press release notes that Symvess is indicated for use in adults as a vascular conduit for extremity...
Merit Medical Systems announced today that the Wrapsody cell-impermeable endoprosthesis has received premarket approval from the US Food and Drug Administration (FDA). With this approval, a press release notes, Merit can begin commercialisation of the device in the USA...
Contego Medical has announced the successful completion of the first commercial cases with its newly US Food and Drug Administration (FDA)-approved Neuroguard IEP system.
A press release notes that John Gaughen Jr, an interventional neuroradiologist at Centra Medical Group (Lynchburg,...
Biotronik announced this week that it has enrolled the first patients in its BIO-OSCAR FIRST trial, a study designed to confirm the safety and clinical performance of the Oscar peripheral multifunctional catheter for dilation of lesions in the femoral,...
A rupture prediction-based algorithm is set to enhance patient selection for abdominal aortic aneurysm (AAA) repair. This is according to Randy Moore (University of Calgary, Calgary, Canada), who, at the 2024 VEITHsymposium (19–23 November, New York, USA), reported that...
Boston Scientific today announced the appointment of Kathleen Van Vlierberghe as the new vice president for the Peripheral Interventions division in Europe, Middle East and Africa (EMEA), effective 1 December 2024.
A news story on the company's website details that Van...
The six-month results of the WAVE (WRAPSODY Arteriovenous Access Efficacy) pivotal trial have reported superior target lesion primary patency (TLPP) with the WRAPSODY (Merit Medical) cell-impermeable endoprosthesis when compared to percutaneous transluminal angioplasty (PTA) when used in failing arteriovenous...
Sensome today announced positive results from two studies of its Clotild smart guidewire system demonstrating its ability to successfully identify fresh clot—thrombus rich in red blood cells (RBCs)—in peripheral arterial disease (PAD) and differentiate it from other tissue encountered...
A new photographic initiative reveals the scale of preventable amputations in the UK and the impact on both patients and the UK National Health Service (NHS).
AmpuNATION, a project led by Abbott, launches with a series of confronting portraits...
Biotronik today announced that it has concluded the BIO-OSCAR SOC study evaluating the baseline against which to measure the Oscar multifunctional catheter in treating complex peripheral arterial disease (PAD). The outcomes were presented at the Paris Vascular Insights (PVI) course...
This advertorial is sponsored by GE HealthCare.
In this article, Bjoern Suckow (Dartmouth Health, Lebanon, USA) discusses the Allia IGS 7 (GE HealthCare) and its role within his daily practice, sharing how the system has “technologically revolutionised” the way he...
Late-breaking data shared at VIVA 2024 (3–6 November, Las Vegas, USA), the first-in-human implantation of a new bridging stent, and an interview with Ramon Varcoe (Sydney, Australia) all feature in November’s top 10.
1. Educational supplement: Is it time to rethink...
Microbot Medical recently announced that it has submitted a 510(k) premarket notification to the US Food and Drug Administration (FDA) for its Liberty endovascular robotic system.
A press release details that Liberty is the world’s first single-use, fully disposable...
Tim Resch (University of Copenhagen, Copenhagen, Denmark) gives an update on the SUNDAY trial, which is set to provide some much-needed data on treatment options for uncomplicated type B aortic dissection (TBAD).
What is the SUNDAY trial?
The SUNDAY trial is...
Artivion recently announced that the US Food and Drug Administration (FDA) has granted a humanitarian device exemption (HDE) for use of the AMDS hybrid prosthesis (formerly known as the Ascyrus Medical dissection stent) in acute DeBakey type I dissections...
InspireMD today announced that the first patient has been enrolled in the company’s CGUARDIANS II clinical trial evaluating its CGuard Prime carotid stent system in patients undergoing carotid artery stenting (CAS) via the transcarotid artery revascularisation (TCAR) approach. The patient...
Terumo Interventional Systems has announced the launch and commercial availability of its R2P NaviCross peripheral support catheter in the USA as part of the company’s radial-to-peripheral (R2P) portfolio. The Terumo Interventional Systems has announced the launch and commercial availability...
FastWave Medical has announced the issuance of its sixth utility patent by the US Patent and Trademark Office (USPTO), the fourth patent the company has been granted this year.
FastWave Medical's intravascular lithotripsy (IVL) systems are designed to address calcified...
Viz.ai has announced new data supporting advancements in neurology and vascular care, with two studies—both set to be presented at the Radiological Society of North America (RSNA) annual meeting (1–5 December 2024, Chicago, USA)—showing positive outcomes and the real-world...
Bentley has announced the successful first-in-human implanation of its recently CE-certified BeFlared stent graft system, which the company states is the world’s first dedicated bridging stent for fenestrated endovascular aneurysm repair (FEVAR).
The procedure was performed by Stéphan Haulon,...
Newly released six-month results from the single-arm arteriovenous graft (AVG) cohort in the WAVE (Wrapsody arteriovenous access efficacy) trial showed target lesion and access circuit primary patency rates of nearly 82% and 68.8%, respectively.
The latest data on the Wrapsody...
Researchers have identified acute effects of cigarette and e-cigarette smoking on vascular function, even without nicotine. The results of the ongoing research are being presented next week at the annual meeting of the Radiological Society of North America (RSNA 2024; 1–5 December,...
Bentley recently announced that it has been granted TÜV certificates for two of its bridging stent products.
One certificate is for the Bentley BeGraft stent, which a press release notes makes it the world’s first on-label bridging device. The second...
Humacyte today announced the publication of clinical results evaluating the efficacy and safety of the acellular tissue engineered vessel (ATEV) in the repair of extremity civilian and military injuries in JAMA Surgery, an American Medical Association peer-reviewed journal.
The publication...
A new analysis has found arm veins to be the most suitable alternative conduits to great saphenous veins (GSV) in chronic limb-threatening ischaemia (CLTI) patients, particularly in below-knee bypass targets. Prosthetic grafts also represent an adequate option, the researchers...
During a presentation at the Egyptian Vascular and Endovascular Society (EVES) 2024 Summit (30 October–2 November, Cairo, Egypt) on managing abdominal aortic aneurysm (AAA), Mohamed Rizk (Ain Shams University, Cairo, Egypt) extolled the benefits of acquiring both open and...
Firas Mussa (Houston, USA) urges specialists to put biases aside when it comes to treatment options for uncomplicated type B aortic dissection (TBAD).
Surgeons in general and vascular surgeons in particular have not been at the forefront of generating evidence....
Gianluca Faggioli (University of Bologna, Bologna, Italy) and Gert J de Borst (UMC Utrecht, Utrecht, The Netherlands) went head-to-head at the recent European Society for Vascular Surgery (ESVS) annual meeting (24–27 September, Kraków, Poland) to debate a motion that...
This advertorial is sponsored by Cardionovum®
Matteo Tozzi (Varese, Italy) recently spoke to Vascular News about the role of drug-coated balloons (DCBs) in vascular access.
The Aperto® over-the-wire (OTW) DCB from Cardionovum—which is available in lengths of 20, 40 and 60mm...
Following his presentation last year of 12-month results from the “landmark” LIFE-BTK randomised controlled trial, Ramon Varcoe (Sydney, Australia) speaks to Vascular News about this research as part of his wider career in vascular surgery so far. The vascular...
This article forms part of an educational supplement sponsored by Shockwave Medical. Explore the full Shockwave L6 series here.
Ashish Patel (Guy’s and St Thomas’ NHS Foundation Trust and King’s College London, London, UK) outlines a case report demonstrating how the new...
This article forms part of an educational supplement sponsored by Shockwave Medical. Explore the full Shockwave L6 series here.
In this case report, Stefano Fazzini (University of Rome Tor Vergata, Rome, Italy) illustrates how the new Shockwave L6 offers an “ideal solution”...
This article forms part of an educational supplement sponsored by Shockwave Medical. Explore the full Shockwave L6 series here.
Mark Portou (Royal Free Hospital, London, UK) presents the case of a patient with chronic limb-threatening ischaemia (CLTI) in whom use of the...
This article forms part of an educational supplement sponsored by Shockwave Medical. Explore the full Shockwave L6 series here.
Charting the history of treatment for severely calcified iliac arteries, Michel Bosiers (University Hospital Bern, Bern, Switzerland) explains how the Shockwave L6—which is...
This educational supplement is sponsored by Shockwave Medical. Explore the full Shockwave L6 series here.
In this supplement:
Michel Bosiers (University Hospital Bern, Bern, Switzerland) explains how the new Shockwave L6 can change the game in the treatment of calcified...
Endologix has announced the final 36-month results from the DETOUR2 study—a prospective, single-arm, international, multicentre clinical evaluation of the novel Detour system for fully percutaneous femoropopliteal bypass procedures.
A press release details that the findings highlight the durable efficacy of...
Shockwave Medical, part of Johnson & Johnson MedTech, has announced the first clinical outcomes associated with the Shockwave Javelin peripheral intravascular lithotripsy (IVL) catheter, a novel, non-balloon-based lithotripsy platform designed to modify calcium and cross extremely narrowed vessels in...
Vascular News' live conference coverage, an interview with Kevin Mani (Uppsala University Hospital, Uppsala, Sweden), and several industry updates feature in October's top 10.
1. Cook Medical creates custom-made aortic device for fenestrated FET procedure
According to Cook Medical, a frozen...
Bolt Medical has announced the completion and results of the RESTORE ATK and RESTORE BTK pivotal clinical trials investigating the company's Bolt intravascular lithotripsy (IVL) above-the-knee (ATK) and below-the-knee (BTK) systems for the treatment of peripheral arterial disease (PAD)...
Results from two studies evaluating the utility of atherectomy for peripheral endovascular interventions have just been released as late-breaking data presentations at VIVA 2024 (3–6 November, Las Vegas, USA).
The Total REALITY study retrospectively compared directional atherectomy versus balloon predilatation...
Presented today, late-breaking data from the second year of the LIFE-BTK clinical trial demonstrate the long-term effectiveness of the US Food and Drug Administration (FDA)-approved Esprit BTK everolimus-eluting resorbable scaffold system (Abbott Vascular) in patients with the most severe...
Royal Philips has announced enrolment of the first patient in the US THOR IDE clinical trial, which will study an innovative combined laser atherectomy and intravascular lithotripsy (IVL) catheter.
Procedures that previously required the use of two different devices can...
R3 Vascular today announced that the US Food and Drug Administration (FDA) has granted the company investigational device exemption (IDE) approval to initiate its ELITE-BTK pivotal trial of the next-generation Magnitude drug-eluting bioresorbable scaffold for below-the-knee (BTK) peripheral arterial...
A retrospective economic analysis of the LIFE-BTK trial has demonstrated the one-year cost-effectiveness of an everolimus-eluting resorbable scaffold over angioplasty for the treatment of infrapopliteal artery disease, with investigators predicting more significant results in the longer term.
Sharing this finding...
The Centers for Medicare and Medicaid Services (CMS) has granted transitional pass-through (TPT) payment for Medtronic's Symplicity Spyral renal denervation (RDN) catheter and Recor Medical's Paradise ultrasound RDN (uRDN) system, according to press releases from the two companies.
TPT payment,...
Medtronic has announced new, long-term data from the SPYRAL HTN-ON MED clinical trial that showed subjects who underwent radiofrequency renal denervation with the Symplicity Spyral renal denervation system had significantly greater reductions in 24-hr ambulatory systolic blood pressure, and...
A debate at this year’s European Society for Vascular Surgery (ESVS) annual meeting (24–27 September, Kraków, Poland) centred on innovation in vascular surgery, with experts considering whether it will ‘change everything’ in the field. While Matt Thompson, chief executive...
A recently published retrospective study has shown that a deep learning model based on non-contrast computed tomography (CT) can detect endoleak after endovascular aneurysm repair (EVAR) “with high sensitivity”.
Writing in CardioVascular and Interventional Radiology (CVIR), Qingqi Yang, Jinglang Hu...
Surmodics has announced that early results of a subset of 60 real-world acute, subacute, and chronic limb ischaemia patients from its PROWL registry study were presented by Dean Ferrera (Powers Health, Munster, USA) at TCT 2024 (27–30 October, Washington,...
NOTE: This video is ONLY available to watch in selected countries and geographies
Ross Milner (University of Chicago, Chicago, USA) outlines how long-term data from the unique GREAT registry supports the use of Gore’s aortic solutions.
The worldwide GREAT registry...
Data from the prospective, multicentre, investigator-initiated SIRONA randomised clinical trial demonstrated MagicTouch (Concept Medical) sirolimus-coated balloons to be noninferior compared to paclitaxel-coated balloons with regards to the primary safety and efficacy endpoints in patients with femoropopliteal artery disease. Findings...
This advertorial is sponsored by Bentley.
Complex peripheral chronic total occlusions (CTOs) represent a daily challenge for interventionalists, necessitating a standardised approach for optimal patient outcomes. A team of experts has developed such a protocol—published in 2023 in JACC: Cardiovascular...
Mark Portou (London, UK) presents the case of a patient with chronic limb-threatening ischaemia (CLTI) in whom use of the new Shockwave L6 resulted in avoiding the need for a stent, thereby reducing risk to the patient and preserving...
In this case report, Stefano Fazzini (University of Rome Tor Vergata, Rome, Italy) illustrates how the new Shockwave L6 offers an “ideal solution” for severely calcified iliac lesions as part of an optimised approach to imaging and functional assessment.
He...
Ashish Patel (Guy’s and St Thomas’ NHS Foundation Trust and King’s College London, London, UK) outlines a case report demonstrating how the new Shockwave L6 catheter represents a “significant addition” to clinicians’ armamentarium for the endovascular treatment of aortoiliac...
Vesalio has announced the initiation of a prospective, single-arm, multicentre study supporting the recently launched pVasc thrombectomy system for non-surgically removing peripheral occlusions. This multidisciplinary clinical research initiative aims to collect real-world insights on the pVasc system in treating...
In this issue:
Innovation in vascular surgery: Balancing progress with patient safety
New ESC guidelines combine peripheral arterial and aortic disease for first time
In profile: Ramon Varcoe (Sydney, Australia)
Advertorial: Andrej Schmidt (Leipzig, Germany) on how the BeBack catheter from Bentley is...
In this issue:
Innovation in vascular surgery: Balancing progress with patient safety
New ESC guidelines combine peripheral arterial and aortic disease for first time
In profile: Ramon Varcoe (Sydney, Australia)
Advertorial: Andrej Schmidt (Leipzig, Germany) on how the BeBack...
According to a new national survey released by the Society for Vascular Surgery (SVS), nearly one in three Americans at the highest risk for developing vascular diseases have not heard of any of the most common conditions, like peripheral arterial...
NOTE: This video is ONLY available to watch in selected countries and geographies
Dennis Gable (The Heart Hospital Baylor Plano, Plano, USA) elaborates on the unique imprint of the GREAT registry that has collected data from multiple continents, and...
CVRx, the company behind the Barostim baroreflex activation therapy system, recently announced that the American Medical Association (AMA) has accepted new Current Procedural Terminology (CPT) Category I codes for the device, which treats the symptoms of heart failure.
The decision...
This advertorial is sponsored by Boston Scientific.
During a satellite symposium session dedicated to real cases and trial outcomes which was held at the Cardiovascular and Interventional Radiological Society of Europe (CIRSE) annual congress (14–18 September, Lisbon Portugal), experts presented...
Endospan today announced the completion of enrolment for the primary arm of its TRIOMPHE investigational device exemption (IDE) clinical study investigating the Nexus aortic arch stent graft. The study is evaluating the safety and efficacy of the device for...
Pilot testing has shown that a new framework—dubbed Endo-STAR—can be used to describe and standardise endovascular interventions for peripheral arterial disease (PAD) within a randomised controlled trial (RCT) protocol and monitor adherence to the protocol over the course of...
Amplitude Vascular Systems (AVS) recently announced that it has enrolled the first patient in its US pivotal trial for pulsatile intravascular lithotripsy (IVL) therapy.
A press release notes that the POWER PAD II clinical study will evaluate the safety...
Microbot Medical has today announced that it has successfully completed enrolment and follow-up for all patients in its ACCESS-PVI human clinical trial.
The company also announced that it is accelerating its go-to-market strategy. It expects to begin building out...
Contego Medical today announced the US Food and Drug Administration's (FDA) premarket approval (PMA) of the Neuroguard IEP system for carotid revascularisation.
The company shares in a press release that clinical studies of the Neuroguard IEP system, including the PERFORMANCE...
Michael Lichtenberg (Arnsberg, Germany) discusses the results of the SUCCESS six-month all-comers registry with Vascular News at CIRSE 2024 after the late-breaking results were presented in the First@CIRSE session.
The real-world registry evaluated the SELUTION SLR™ PTA drug-eluting balloon (DEB;...
Ahmed Eleshra (Mansoura University, Mansoura, Egypt) and Sherif Elkerdawi (Wadi Elneel Hospital, Cairo, Egypt) write together about the upcoming Egyptian Vascular and Endovascular Society (EVES) 2024 Summit (30 October–1 November, Cairo, Egypt), touching on the state of vascular surgery...
Findings from a retrospective analysis presented at the recent European Society for Vascular Surgery (ESVS) annual meeting (24–27 September, Kraków, Poland) provide evidence that—for symptomatic carotid artery stenosis patients—endarterectomy “remains a useful and relevant intervention in the era of...
InspireMD has announced the US Food and Drug Administration (FDA) approval of the company’s investigational device exemption (IDE) application to initiate the CGUARDIANS II pivotal study of its CGuard Prime 80cm carotid stent system during transcarotid artery revascularisation (TCAR)...
Medinol has announced the successful first-in-human implantation of the ChampioNIR drug-eluting peripheral stent by Gerard S Goh and Thodur Vasudevan of the Alfred Hospital in Melbourne, Australia.
The ChampioNIR drug-eluting peripheral stent poses a transformational technology designed to improve patient...
New study data suggest that the interventional treatment of symptomatic carotid artery disease may result in an improved rate of age-adjusted overall survival compared to medical management. However, the same study—presented at this year’s European Society for Vascular Surgery...
Six-month data from Merit Medical's Wrapsody WAVE trial, an update on the NHS England Acute Aortic Dissection Toolkit, and a large meta-analysis of endovascular aneurym repair (EVAR) outcomes by age all caught Vascular News readers' attention in September.
1. Merit...
This week Cook Medical announced that it has created a custom-made device for the frozen elephant trunk (FET) aortic procedure.
A press release details that, earlier this year, cardiothoracic surgeon François Dagenais (Quebec Heart & Lung University Institute, Quebec City,...
Surmodics has announced that it has received US Food and Drug Administration (FDA) 510(k) clearance for its Pounce XL thrombectomy system.
The Pounce XL thrombectomy system is indicated for the non-surgical removal of thrombi and emboli from the peripheral arterial...
Access Vascular Inc (AVI) today announced findings from a proactive device evaluation showing a significant reduction in complications with a midline catheter made with its Mimix hydrophilic biomaterial (HBM), compared with a conventional chlorhexidine (CHG)-coated catheter.
The evaluation, results of...
Baylis Medical Technologies has today announced the commercial launch of the PowerWire Pulse radiofrequency (RF) guidewire in the USA.
A press release notes that the PowerWire Pulse RF guidewire is the newest addition to the PowerWire family of RF guidewires...
“We need to continue to build the scientific evidence base for what we do,” Kevin Mani (Uppsala, Sweden) tells Vascular News, underlining what he believes to be a key priority for vascular surgery in the years ahead. Research has...
A large meta-analysis of elective endovascular aneurysm repair (EVAR) outcomes by age has shown that three-to-five-year survival is very low in octogenarians, prompting researchers to caution against routine intervention in these patients.
Sebastian Vaughan-Burleigh, Dominic PJ Howard and colleagues (Oxford,...
Boston Scientific announced the close of its acquisition of Silk Road Medical, the company behind transcarotid artery revascularisation (TCAR).
"The integration of the TCAR platform into our portfolio means we can offer a treatment option for patients suffering from carotid artery disease that...
Royal Philips today announced the introduction of the 160cm US Food and Drug Administration (FDA)-approved version of its LumiGuide endovascular navigation wire. This enhanced LumiGuide guidewire, which utilises the company’s Fiber Optic RealShape (FORS) technology, was used for the...
Merit Medical has today announced positive six-month findings from the randomised arteriovenous (AV) fistula arm of its Wrapsody arteriovenous access efficacy (WAVE) pivotal trial. The data were shown at the Cardiovascular and Interventional Radiological Society of Europe (CIRSE) annual...
Shockwave Medical, part of Johnson & Johnson MedTech, has announced the full US launch of its Shockwave E8 peripheral intravascular lithotripsy (IVL) catheter, following clearance by the US Food and Drug Administration (FDA).
A company press release details that...
A dedicated toolkit has made it safer to be an aortic dissection patient in England today than it was before the project’s 2022 launch, but there is still work to be done, Rachel Bell (Freeman Hospital, Newcastle, UK) informed...
Penumbra today announced it has secured CE mark in Europe for two computer-assisted vacuum thrombectomy (CAVT) technologies—Lightning Flash 2.0 and Lightning Bolt 7.
“Based on outcomes from our clinical trials and from physicians using our devices globally, our CAVT technologies...
In a recently conducted survey, over 200 US advanced practice providers (APPs) in vascular surgery cited performing at the top of their licence as the most important determinant of job satisfaction. This and other key findings are set to...
In this short video, Michael Kostrzewa (Baden, Switzerland) and Rashid Akhtar (London, UK) discuss the latest approaches to the treatment of acute pulmonary embolism (PE) and deep vein thrombosis (DVT) using mechanical thrombectomy.
They examine which patients are eligible...
Commencement of the CALCIO trial—designed to collect real-world data on the use of intravascular lithotripsy (IVL) with the Shockwave Medical IVL system in patients with chronic limb-threatening ischaemia (CLTI)—was the most-read Vascular News story in August.
1. CIRSE begins CALCIO trial...
The Society for Vascular Surgery Vascular Quality Initiative (SVS VQI) clinical registry today announced the Carotid Care Quality Champion recognition programme.
The programme honours healthcare organisations across the USA dedicated to improving the safety and effectiveness of vascular care through...
“I would challenge anybody who says that VenaSeal is investigational. It’s a robust, well-evaluated, very well studied intervention that I think we would all agree has a part in the overall armamentarium for treating venous disease.” – Manj Gohel
During...
Vesalio announces the US commercialisation of pVasc thrombectomy system for non-surgically removing peripheral occlusions.
A recent press release outlined that pVasc targets the full range of emboli from soft acute clots to fibrin-rich, calcified ones. The company states that...
AngioDynamics today announced European CE mark approval of the Auryon atherectomy system, designed for the treatment of peripheral arterial disease (PAD), including chronic limb-threatening ischaemia (CLTI) and in-stent restenosis (ISR).
The Auryon atherectomy system uses solid-state laser technology to treat...
Matteo Tozzi (Varese, Italy) recently spoke to Vascular News about the role of drug-coated balloons (DCBs) in vascular access.
The full professor of vascular surgery at the University of Insubria started the use of Aperto® OTW DCB by Cardionovum® in his vascular...
In this issue:
European registry set to raise the evidence bar for deep venous intervention
"There's no meeting like it": Stage is set for CX Aortic Live 2024
In profile: Kevin Mani (Uppsala, Sweden)
Advertorial: Philips' FORS-powered LumiGuide has...
In this issue:
European registry set to raise the evidence bar for deep venous intervention
"There's no meeting like it": Stage is set for CX Aortic Live 2024
In profile: Kevin Mani (Uppsala, Sweden)
Advertorial: Philips' FORS-powered LumiGuide has...
The pilot phase of a new European Venous Registry (EVeR)—designed to illuminate the real-world outcomes of deep venous interventions over a 10-year time horizon—is set to launch this month (September 2024).
Developed and hosted by the European Society for Vascular...
Patients with peripheral arterial disease (PAD) reporting lower levels of social support experience worse health outcomes, a new Yale-led study finds.
Social support is thought to bolster cardiovascular health by facilitating health-promoting behaviours and acting as a buffer against the...
The European Society of Cardiology (ESC) has today published its 2024 guidelines for the management of peripheral arterial and aortic diseases (PAAD), evaluating these vascular conditions together as part of the same cardiovascular system.
The guidelines are aimed at...
In her new role as an academic clinical fellow in vascular surgery, our Launch Pad columnist Sarah Sillito (Newcastle, UK) writes about the importance of creating a research-confident workforce at the medical school level.
The first Wednesday in August marks...
iVascular has announced the first clinical outcomes of the Restorer bare metal stent (BMS) in iliac artery treatment.
The data were presented earlier this summer at the Leipzig Interventional Course (LINC 2024; 28–31 May, Leipzig, Germany) by Lieven Maene...
This advertorial is sponsored by Philips. FORS-powered
A new technology is set to change the endovascular game by offering 3D, full-colour guidance to replace the 2D, greyscale standard-of-care. The result? Increased visualisation without the use of fluoroscopy. This is one...
Nikos Giannikas and Panos Kitrou (Patras, Greece) write together about the role that endothelial tight junctions play in vascular access, evidence from previous studies, and a proposed upcoming study that aims to evaluate the expression of the main tight...
Michael Conte (San Francisco, USA) recently used his Veith Lecture at the 2024 Vascular Annual Meeting (VAM; 19–22 June, Chicago, USA) to call on young vascular surgeons on the peripheral arterial disease (PAD) frontlines to drive the space forward amid...
Researchers have reported a 94% technical success rate among other key findings from a recent international, multicentre, single-arm cohort study of physician-modified endografts (PMEGs) for thoracoabdominal and complex abdominal aortic aneurysm (AAA) repair. The work has been published in...
Hannah Burton (Bedford, UK) writes about a new study—recently documented in the European Journal of Vascular and Endovascular Surgery—on the fate of device packaging for endovascular aneurysm repair (EVAR).
Sustainable surgery is an ever-growing topic of interest due to the...
One-year limb salvage and quality of life (QoL) data from the STRIDE study, showing that frontline use of Penumbra’s Indigo system for patients with lower extremity acute limb ischaemia (LE-ALI) is safe and effective, has been published in the...
A new research letter underscores the need to improve long-term survival following lower extremity revascularisation for chronic limb-threatening ischaemia (CLTI), setting out a randomised controlled trial to examine the impact of coronary artery ischaemia testing in these patients.
The letter,...
A newly published US Medicare cohort study illuminates trends and factors associated with peripheral vascular interventions (PVIs)—including a sharp rise in the use of ambulatory surgical centres (ASCs) and office-based laboratories (OBLs) for treatment—from 2011 to 2022.
In the Journal...
A meeting centred around live case transmissions from across the world, CX Aortic Live 2024 (7–8 October, Vienna, Austria), will showcase the latest cardiac, vascular and endovascular aortic advances for a global, multidisciplinary audience.
Designed to bring together all...
Amplitude Vascular Systems (AVS), developer of the Pulse intravascular lithotripsy (IVL) platform, recently announced it has entered into a strategic agreement with the Jacobs Institute, a non-profit medical device innovation centre in Buffalo, USA, led by Adnan Siddiqui.
The partnership...
BioCardia has submitted a 510(k) for approval of its patented Morph DNA steerable introducer sheath to the US Food and Drug Administration (FDA).
This product family is intended to provide a pathway through which medical instruments, such as balloon dilatation...
China’s National Medical Products Administration (NMPA) has granted a Class III Medical Devices Certificate of Registration for the msRDN radiofrequency renal denervation system (SyMap Medical).
The msRDN system consists of a disposable renal artery radiofrequency ablation catheter, a console with...
Contego Medical recently announced that Mitchell Silver, an interventional cardiologist and vascular medicine specialist, has joined the team as chief medical officer (CMO).
A press release notes that Silver will play a pivotal role in shaping the company's clinical strategy...
Centerline Biomedical recently announced that its Intra-Operative Positioning System (IOPS) Viewpoint catheter has received US Food and Drug Administration (FDA) 510(k) clearance. The Viewpoint catheter is the most recent addition to the company's patented IOPS portfolio.
A press release details...
Several company updates including Series A funding for Endoron Medical, new insights on the role of machine learning in determining endovascular aneurysm repair (EVAR) suitability, and more from this year's Vascular Annual Meeting (VAM 2024; 19–22 June, Chicago, USA)...
Vascular Therapies has today announced completion of enrolment in the ACCESS 2 trial, a Phase III prospective randomised, multicentre clinical study of Sirogen for the surgical creation of arteriovenous (AV) fistula in patients requiring haemodialysis vascular access.
Vascular Therapies developed its...
A roundtable discussion at the 2024 Charing Cross (CX) Symposium (23–25 April, London, UK) saw a multidisciplinary panel of experts discuss a new multimodal treatment for diabetic wounds—AOTI’s topical oxygen therapy (TWO2).
Andrew Boulton (Manchester, UK), professor of medicine at...
InspireMD today congratulated the lead investigators on the completion of enrolment in the CREST-2 clinical trials.
The CGuard embolic prevention stent system (EPS), utilising InspireMD's MicroNet mesh, was included as a device option for stenting in the CREST-2 trials...
The Cardiovascular and Interventional Society of Europe (CIRSE) has announced that patient enrolment for the CALCIO trial has begun. CALCIO started patient enrolment on 15 July, 2024, with the goal of recruiting 400 chronic limb-threatening ischaemia (CLTI) patients over...
BrosMed Medical has announced the appointment of Scott J Addonizio as its chief operating officer (COO), effective immediately.
A press release details that Addonizio brings a wealth of experience and expertise to BrosMed, with a distinguished career spanning almost 30...
Silk Road Medical has announced the milestone of more than 100,000 transcarotid artery revascularisation (TCAR) procedures having been performed to date.
“A growing number of patients come to us looking for a solution that is minimally invasive and requires less...
PECA Labs has added Joseph E Bavaria (Jefferson Health and Sidney Kimmel Medical College, Philadelphia, USA) to its board of directors. Bavaria, a cardiac surgeon, will provide leadership to PECA as it continues to develop and commercialise cardiovascular implants...
The first patients in the USA have been treated in Recor Medical’s Global Paradise System US Post Approval Study (US GPS), a real-world study gathering data on the long-term safety and effectiveness of ultrasound renal denervation.
Antonio Gutierrez is the...
Patients who undergo carotid revascularisation at the weekend have increased odds of complications and mortality, with transfemoral carotid artery stenting (TFCAS) posing the highest risk and transcarotid artery revascularisation (TCAR) the lowest, compared to carotid endarterectomy (CEA). This is...
Researchers at the University Medical Center Groningen (Groningen, The Netherlands) and the University of Twente (Enschede, The Netherlands) have developed a prediction model that they claim could assist in the preoperative identification of patients who are unlikely to achieve...
iVascular has announced that "outstanding" results from the SOL Japan study were recently presented at the Japan Endovascular Treatment (JET) conference (14–16 June, Fukuoka, Japan).
SOL Japan is a clinical trial aimed at evaluating the efficacy and safety of...
CX co-chairs Dittmar Bӧckler (University Hospital Heidelberg, Heidelberg, Germany) and Andrew Holden (Auckland City Hospital, Auckland, New Zealand) discuss with Marc Schermerhorn (Beth Israel Deaconess Medical Center, Boston, USA) the unmet need in EVAR and the failure of some...
Michel Reijnen and Rianne van Rijswijk (both Rijnstate Hospital, Arnhem, The Netherlands) write about a new consortium focused on sac regression after endovascular aneurysm repair (EVAR).
Aneurysm shrinkage after EVAR has been identified as an important predictor for treatment success....
Life Seal Vascular recently announced that it has been awarded a National Science Foundation (NSF) Small Business Innovation Research (SBIR) grant. The funding will be used to accelerate the development of the company's endovascular aneurysm repair (EVAR) technology.
"We are...
NOTE: This video is ONLY available to watch in selected countries and geographies
Yoshimitsu Soga (Fukuoka, Japan) spoke to Vascular News at the Leipzig Interventional Course (LINC) 2024 (28–31 May, Leipzig, Germany) about the SOL-Japan trial.
The interventional oncologist at...
The top 10 most-read Vascular News stories in June featured two US Food and Drug Administration (FDA) investigational device exemption (IDE) approvals, Boston Scientific's announcement that it will acquire Silk Road Medical, and a series of highlights from this...
New research on Chat generative pre-trained transformer (GPT) technology and its Vascular Education and Self-Assessment Program (VESAP) success rate provides insight into the future of artificial intelligence (AI) in vascular surgery training and practice, investigators Michael Amendola (Richmond, USA)...
The inaugural president spoke to Vascular News about the newly formed International Society for Women Vascular Surgeons ahead of this year’s Women's Vascular Summit (3–4 May, Chicago, USA), outlining its genesis and goals, as well as some of...
Matteo Tozzi (Varese, Italy) and Kate Steiner (Stevenage, UK) sat down with Vascular News at this year’s Charing Cross (CX) International Symposium (23–25 April, London, UK) to provide their thoughts on the latest findings from the IN.PACT AV Access...
Transit Scientific announces the successful clinical deployment of XO Cath, an embolic delivery microcatheter intended for interventional procedures.
Richard Saxon (North County Radiology Associates, San Diego, USA), performed the inaugural procedure utilising the XO Cath 2.0Fr 130cm microcatheter with a...
Endoron Medical has announced the successful closing of its US$10 million Series A funding round. The round was led by Sofinnova Partners, with significant matching contributions from the European Innovation Council Fund (EIC Fund).
A press release notes that the...
The ATEV is an investigational product and has not been approved for sale by the FDA or any other regulatory agency.
NOTE: This video is ONLY available to watch in selected countries and geographies
Humacyte's Acellular Tissue Engineered Vessel, a...
Avicenna.AI has received Medical Device Regulation (MDR) certification for five of its algorithms from British Standards Institution (BSI) Medical Devices—a development that means the company's product portfolio is fully compliant with the European Union's (EU) MDR 2017/745, which is...
New data presented at the 2024 Vascular Annual Meeting (VAM; 19–22 June, Chicago, USA) have revealed poor overall rates of medical therapy adherence in moderate asymptomatic carotid stenosis patients—with smoking cessation representing a “ripe target” for improvement moving forward.
“The...
Humacyte recently announced it has been granted Regenerative Medicine Advanced Therapy (RMAT) designation from the US Food and Drug Administration (FDA) for its investigational acellular tissue engineered vessel (ATEV), designed to treat patients with advanced peripheral arterial disease (PAD)....
Artivion recently announced it has amended its credit facility and option purchase agreements with Endospan, developer of the Nexus stent graft system. In 2019, Artivion provided a credit facility to Endospan and entered into an option agreement to purchase...
This advertorial is sponsored by Terumo Aortic.
On 9 April 2024, a significant milestone was achieved as Terumo Aortic introduced the first implantation of the Fenestrated TREO device in Asia. The procedure was performed at Tan Tock Seng Hospital (TTSH)...
Alexander Mallios (Paris, France) and Andrew Holden (Auckland, New Zealand; one of the co-chairs of the Charing Cross Symposium) share their views on the first and only consistently positive randomised controlled trial results that are “unequivocally” in favour...
Gore has announced the first commercial use of the Gore Tag thoracic branch endoprosthesis (TBE) in Canada.
The news came as Canadian government health authority Health Canada approved the first-of-its-kind endovascular device, which is indicated to treat lesions of the...
A new technology is set to change the endovascular game by offering 3D, full-colour guidance to replace the 2D, greyscale standard-of-care. The result? Increased visualisation without the use of fluoroscopy. This is one of the key messages to have emerged from a...
A recent study suggests that endovascular treatment of the common femoral artery (CFA) is associated with an increased rate of long-term CFA-specific reintervention, regardless of indication. Nicholas Wells, a medical student at Yale School of Medicine in New Haven,...
Results of a sub-study from the pivotal investigational device exemption (IDE) trial assessing Gore’s Excluder conformable abdominal aortic aneurysm (AAA) endoprosthesis have shown that the device is safe and effective in hostile angulated neck anatomy, with the results...
High stroke and death rates relating to carotid stenting in the Pacific Northwest may be driven by the selection of high-risk patients with less than 80% stenosis, and reducing the frequency of stenting in this high-risk group—along with better...
A study examining outcomes between different carotid revascularisation procedures has concluded that transcarotid artery revascularisation (TCAR) and carotid endarterectomy (CEA) both represent a “reasonable choice” in severe chronic kidney disease (CKD) and haemodialysis patients—but transfemoral carotid artery stenting (TF-CAS)...
“Our findings ultimately suggest that vascular evaluation within six weeks of wound appearance can significantly supplement wound healing in wound centres,” said Saranya Sundaram, vascular surgery resident (PGY-3) at the Medical University of South Carolina in Charleston, USA, presenting...
The US Food and Drug Administration (FDA)-approved Gore Excluder Thoracoabdominal Branch Endoprosthesis (TAMBE) has been shown to be safe and effective at 30 days for the treatment of patients with complex aortic aneurysms involving the visceral aorta.
This is according...
The Society for Vascular Surgery (SVS) has announced Linda Harris as its next vice president, meaning that she will become only the second woman president in the history of the SVS.
The announcement was made during the SVS Annual...
Edited case using LumiGuide, powered by Fiber Optic RealShape (FORS) technology.
For physicians who regularly perform complex aortic procedures, high radiation and limited visibility is one of the challenges. In this video, recorded at the 46th Charing Cross International Symposium...
Cordis has announced positive 24-month results from the Selution SFA Japan trial. The prospective, multicentre, single-arm trial is designed to assess the safety and efficacy of the Selution SLR drug-eluting balloon (DEB) for the endovascular therapy of de novo...
Boston Scientific has announced that it has entered into a definitive agreement to acquire Silk Road Medical, a medical device company involved in producing products to be deployed during transcarotid artery revascularisation (TCAR). The purchase price is US$27.50 per...
Tara Mastracci (Barts Health NHS Trust, London, UK) writes about the recent creation of a European Society for Vascular Surgery (ESVS) Equality, Diversity and Inclusion (EDI) task force.
The news of the ESVS’ decision to create an EDI task force,...
This advertorial is sponsored by PolyNovo.
In this article, Victoria Bristow (Cambridge, UK) discusses the NovoSorb Biodegradable Temporising Matrix (BTM) from PolyNovo and shares a case report demonstrating its use in the treatment of a patient with chronic limb-threatening ischaemia...
Xeltis has announced that it has gained approval from the US Food and Drug Administration (FDA) for an investigational device exemption (IDE) submission to begin enrolling patients into a pivotal study for aXess.
aXess is a restorative conduit that enables...
Robert Shahverdyan (Hamburg, Germany) encourages interventionalists to try the new Flex Vessel Prep system (VentureMed) to experience the benefits that arrive in the wake of using this three-bladed device. The device creates symmetrical microincisions when deployed, rotated and pulled...
Interested in medicine from a young age, Bijan Modarai (London, UK) is now professor of vascular surgery at King’s College London and a consultant vascular and endovascular surgeon at Guy’s and St Thomas’ NHS Foundation Trust, where he co-manages...
Vicente Riambau (Barcelona, Spain) describes the unique features and improvements of the iCover balloon expandable covered stent, notably its low profile, radiopaque markers, and total encapsulation with polytetrafluoroethylene (ePTFE), which give the device an edge over its competitors and...
Amplitude Vascular Systems (AVS) announced today that it has received an investigational device exemption (IDE) from the US Food and Drug Administration (FDA) to begin its pivotal trial for pulsatile intravascular lithotripsy (IVL) therapy.
The POWER-PAD-II clinical study will...
The concept of a dedicated cardiovascular centre and whether this is best for optimal patient care was the topic of a lecture delivered by Alan Lumsden (Houston, USA) at the 2023 Vascular Societies’ Annual Scientific Meeting (VASAM; 22–24 November,...
Baylis Medical Technologies today announced the completion of its first clinical use of the PowerWire Pro radiofrequency (RF) guidewire to safely cross a chronically occluded peripheral stent. The procedure was performed in April 2024 by Mark Iafrati (Vanderbilt University...
Anahita Dua (Boston, USA) spoke to Vascular News at the 2023 Vascular Societies’ Annual Scientific Meeting (VSASM; 22–24 November, Dublin, Ireland) on some of the key messages from her presentations at the conference.
One presentation centred around the concept of “strong and...
Sabine Steiner (University of Leipzig, Leipzig, Germany) took to the podium at this year’s Leipzig Interventional Course (LINC 2024; 28–31 May, Leipzig, Germany) to share, for the first time, two-year data from the BEST SFA randomised controlled trial (RCT)...
Andrew Holden (Auckland, New Zealand), one of the co-chairs of the Charing Cross (CX) International Symposium, presented the final five-year data on the vital status of patients enrolled in the IN.PACT AV Access pivotal trial in April 2024 as...
Shape Memory Medical recently announced that the first patient has been treated in the AAA-SHAPE pivotal trial.
AAA-SHAPE (Abdominal aortic aneurysm sac healing and prevention of expansion) is a prospective, multicentre, randomised, open-label trial to determine safety and effectiveness of...
Cook Medical have announced that Beacon tip sizing catheters are now available in the USA and Canada. The Beacon catheter is available in a variety of lengths and tip configurations, a recent press release adds.
The Beacon catheter is used...
Johnson & Johnson today announced it has completed its acquisition of Shockwave Medical. Shockwave is now part of Johnson & Johnson and will operate as a business unit within Johnson & Johnson MedTech.
Joaquin Duato, chairman and chief executive officer...
FastWave Medical has announced the 30-day results of its first-in-human (FIH) study using the company's differentiated peripheral intravascular lithotripsy (IVL) technology.
This was a prospective, single-arm study to assess the safety and feasibility of the FastWave IVL system in...
New data from the C-GUARDIANS pivotal investigational device exemption (IDE) trial support consideration of carotid artery stenting (CAS) with the CGuard embolic prevention stent system (EPS; InspireMD) as a "frontline therapeutic option for appropriate patients being considered for carotid...
In this issue:
Common ground: High-quality data and individualised care emerge as non-negotiables in CX 2024 aortic debates
Studies highlight need for tailored treatment options for women with peripheral arterial disease
In profile: Bijan Modarai (London, UK)
Feature: Experts...
In this issue:
Common ground: High-quality data and individualised care emerge as non-negotiables in CX 2024 aortic debates
Studies highlight need for tailored treatment options for women with peripheral arterial disease
In profile: Bijan Modarai (London, UK)
Feature: Experts...
New multisociety clinical practice guidelines for the management of lower extremity peripheral arterial disease (PAD) have been published online in the Journal of the American College of Cardiology (JACC). One of the document’s key takeaway messages is that care...
Majorie van Helvert (University of Twente, Enschede, The Netherlands) and Michel Reijnen (Rijnstate Hospital, Arnhem, The Netherlands) are currently conducting research on ultrafast contrast-enhanced ultrasound blood flow quantification in peripheral arterial disease (PAD) patients after endovascular treatment. Early findings...
New two-year follow-up data from the BIOMAG-I first-in-human trial confirms a reliable and predictable long-term safety profile for Freesolve, the third-generation resorbable magnesium scaffold (RMS), establishing it as a true alternative to contemporary drug-eluting stents (DES). The results were...
FastWave Medical has been issued its fifth utility patent by the United States Patent and Trademark Office (USPTO), a press release reveals.
"FastWave's IVL systems have been designed with physician input to deliver meaningful improvements for the management of...
This advertorial, sponsored by Teleflex, is only available in selected countries and geographies.
Ozan Yazar is a vascular and endovascular surgeon with international training acquired in various countries including Belgium, The Netherlands, and Germany. He is currently working at...
New clinical results highlight the need for inclusive approaches and comprehensive examinations of treatment options for peripheral arterial disease (PAD), including endovascular therapy and revascularisation. The data were presented recently at the Society for Cardiovascular Angiography & Interventions (SCAI)...
Reflow Medical has announced completion of enrolment in the DEEPER REVEAL clinical trial to evaluate the Reflow Spur stent.
The company notes in a press release that the Spur—designated a Breakthrough Device by the US Food and Drug Administration...
Our Launch Pad columnist Sarah Sillito (Newcastle, UK), who ran the Rouleaux Club Introduction to Vascular Surgery Course at the recent Charing Cross (CX) International Symposium (23–25 April, London, UK), writes about the importance of there being “mutual respect...
L-R: Dittmar Böckler, Eric Verhoeven and Hence Verhagen
“Centralisation and interdisciplinary teams—they are the keys,” Dittmar Böckler (University Hospital Heidelberg, Heidelberg, Germany) concluded during a roundtable discussion at the 2024 Charing Cross (CX) International Symposium (23–25 April, London, UK)...
Experience wall-to-wall thrombectomy in action with Manuela Konert from Leipzig, Germany! In this brief video, Konert walks through a deep vein thrombosis (DVT) case step by step using the ClotTriever system, providing valuable clinical insights along the way. Highlighting...
Terumo Aortic today announced that it has received the prestigious King’s Award for Enterprise in the Innovation category for its global custom solutions programme. This is the company’s first King's Award for Enterprise, having previously been the recipient of...
This advertorial, sponsored by Gore, is only available in selected countries and geographies.
Following the European launch of the recently CE-marked GORE TAG Thoracic Branch Endoprosthesis (TBE) device, Joseph Bavaria (University of Pennsylvania, Philadelphia, USA) gives Vascular News an...
This advertorial, sponsored by Gore, is only available in selected countries and geographies.
The GORE TAG Thoracic Branch Endoprosthesis (TBE) recently became the first device of its kind to be approved in Europe with a CE mark under the...
This advertorial, sponsored by Gore, is only available in selected countries and geographies.
A satellite session at the 2024 Charing Cross (CX) International Symposium (23–25 April, London, UK) focused on a new treatment option for left subclavian artery (LSA)...
Gore has announced US Food and Drug Administration (FDA) approval of an expanded indication for the Gore Excluder conformable abdominal aortic aneurysm (AAA) endoprosthesis. The device is now indicated for patients with aortic neck angulation ≤90° and a minimum...
Patients undergoing cyanoacrylate glue closure for superficial venous disease reported higher periprocedural satisfaction than those who were treated via surgical stripping, data from a pair of randomised controlled trials (RCTs) assessing the VenaSeal system (Medtronic) reveal.
The modality also showed...
Expanse Ice recently announced that its Ice aspiration system has received 510(k) clearance from the US Food and Drug Administration (FDA).
A press release notes that the Ice system is specifically designed to address the complex challenges associated with peripheral...
Abbott has announced that the US Food and Drug Administration (FDA) has approved the Esprit BTK everolimus-eluting resorbable scaffold system (Esprit BTK system), a breakthrough innovation for people with chronic limb-threatening ischemia (CLTI) below-the-knee (BTK).
The Esprit BTK System is...
Provisio Medical has announced US Food and Drug Administration (FDA) 510(k) clearance of the Provisio sonic lumen tomography (SLT) intravascular ultrasound (IVUS) system.
According to a press release, SLT technology addresses a critical unmet need for vascular specialists by...
The Dragons' Den-style competition returned on the final day of CX 2024 during the Innovation Showcase programme, introducing next-generation technology to delegates in attendance. “You have made our job as Dragons very difficult,” the judges said, as they commended...
Following audience polling, Nathalie Yonan (Newcastle upon Tyne, United Kingdom) and Balraj Maan (Willington, United Kingdom) were announced as the winners of the Worrying Foot Competition at this year's Charing Cross (CX) International Symposium (23–25 April, London, UK). The...
Presenting podium-first data on specialist chronic limb-threatening ischaemia (CLTI) clinics and rates of amputation, Assad Khan (Leeds, United Kingdom) provided an argument for “favourable” outcomes—including limb salvage—in a dedicated setting on the final day of the CX Symposium.
Briefly establishing...
In a podium-first presentation on the final day of the 2024 Charing Cross (CX) International Symposium (23–25 April, London, UK), Sean Lyden (Cleveland Clinic, Cleveland, USA) disclosed an interim analysis from the JAGUAR trial and, in doing so, gave...
In the Vascular Trauma Controversies session at this year's Charing Cross (CX) International Symposium (23–25 April, London, UK), one of the highlights was seen early on in proceedings, with a debate on whether there is a role for resuscitative...
“The incidence of acute aortic dissection in England is rising,” Arun Pherwani (University Hospitals of North Midlands NHS Trust, Stoke-on-Trent, UK) told the 2024 Charing Cross (CX) International Symposium (23–25 April, London, UK). Pherwani delivered this statement among other...
In the UK National Institute for Health and Care Research (NIHR) Health Technology Assessment (HTA)-funded BASIL-3 randomised controlled trial (RCT), neither drug-coated balloon (DCB) angioplasty with or without bare metal stent nor drug-eluting stent (DES), when used in the...
Balloon occlusion is one of the most hotly contested topics when it comes to vascular surgery in the setting of emergency trauma and, with this in mind, the Vascular Trauma Controversies programme at CX 2024 intends to move the...
A first-time presentation at the 2024 Charing Cross (CX) International Symposium (23–25 April, London, UK) of five-year data from the IN.PACT AV Access study evaluating the IN.PACT AV (Medtronic) drug-coated balloon (DCB) led principal investigator and CX co-chair Andrew...
Penumbra has announced the US Food and Drug Administration (FDA) clearance and launch of Lightning Flash 2, the next generation computer assisted vacuum thrombectomy (CAVT) system to remove venous thrombus and treat pulmonary embolism (PE), a press release states.
Lightning...
During today’s (Tuesday) Aortic Techniques & Technologies session, Hence Verhagen (Rotterdam, Netherlands) delivered a podium-first presentation on long-term health-related quality of life (HRQoL) after acute type B aortic dissection (ATBD), sharing data from a cross-sectional survey study.
“This is the...
“Unfortunately, we are doing worse for our patients today,” were the sobering thoughts of Eric Secemsky (Boston, United States) during a late-breaking presentation in this morning’s Peripheral Arterial Controversies programme. Among several presentations on paclitaxel safety, he reported that:...
This advertorial, sponsored by Gore, is only available in selected countries and geographies.
“This is a milestone for arch repair,” Dittmar Böckler (Heidelberg, Germany) says of the first ever implantation of the Gore TAG thoracic branch endoprosthesis (TBE) in...
“These will be important, salutary lessons. Should a controversy arise again involving a proven efficacious therapy, we now know that stopping access to that therapy may result in unwanted and unexpected negative effects on patient care.” These are the...
Transcarotid artery revascularisation (TCAR) features in three of March's most popular Vascular News stories, with a talk on the future of aortic repair and the announcement of a new peripheral arterial disease (PAD) vascular covered stent trial also making...
Nectero Medical recently announced the closing of its US$96 million Series D financing round.
Financing was led by Norwest Venture Partners, with large investments from Boston Scientific Corporation, BioStar Capital, Cadence Healthcare Ventures, Aphelion Capital and other firms.
A press...
This advertorial, sponsored by BD, is only available in selected countries and geographies.
This year marks a significant milestone as BD celebrates 25 years of its RotarexS rotational atherothrombectomy system, a journey that started in the Swiss mountains with...
Johnson & Johnson is to acquire Shockwave Medical, it has been announced today.
Under the terms of the transaction, Johnson & Johnson will acquire all outstanding shares of Shockwave for US$335.00 per share in cash, corresponding to an enterprise value...
Erin Murphy (Atrium Health’s Sanger Heart and Vascular Institute, Charlotte, USA), one of the CX Symposium cochairs and a CX Executive Board member, along with fellow CX Executive Board members Stephen Black (Guy’s and St Thomas’ Hospital; King’s College...
Silk Road Medical has announced the launch of its Enroute transcarotid neuroprotection system Plus (NPS Plus), which the company describes as a key component of its transcarotid artery revascularisation (TCAR) portfolio.
As per a Silk Road press release, this next-generation device...
Debates will shed new light on controversies in the treatment of aortic disease at the 2024 Charing Cross (CX) Symposium, alongside practical sessions with a focus on techniques and technologies in both aortic endovascular and open surgical repair.
Running over...
Issue highlights:
Endovascular showdown: CX 2024 sets the stage for BASIL-3 first-time data presentations
A year in profiles: Rachel Bell, Jürgen Falkensammer, Peter Schneider, and Daniel Clair
EVC 2024: Personalised, precision medicine is key to the future of aortic...
Issue highlights:
Endovascular showdown: CX 2024 sets the stage for BASIL-3 first-time data presentations
A year in profiles: Rachel Bell, Jürgen Falkensammer, Peter Schneider, and Daniel Clair
EVC 2024: Personalised, precision medicine is key to the future of aortic...
During the Society of Interventional Radiology (SIR) 2024 annual meeting (23–28 March, Salt Lake City, USA), newly presented data from a subgroup analysis of the STRIDE study showed that Penumbra’s Indigo aspiration system used in a single session without...
In a recent interview, Marie Josee van Rijn from Rotterdam, the Netherlands and Emma Wilton from Oxford, UK, provided insight into the transformative power of interventional approaches in the treatment of DVT. They also set the stage for the...
Medical imaging AI company Avicenna.AI has announced 510(k) clearance from the US Food and Drug Administration (FDA) for its CINA-iPE artificial intelligence (AI)-powered tool that detects incidental pulmonary embolism (PE) during routine computed tomography (CT) scans.
The CINA-iPE algorithm identifies lung blood...
Getinge and Cook Medical today announced an exclusive sales and distribution agreement for the iCast covered stent system, which recently received US Food and Drug Administration premarket approval for the treatment of symptomatic iliac arterial occlusive disease.
Cook Medical will...
At the Charing Cross (CX) Symposium 2024 (23–25 April, London, UK), Andrew Bradbury (University of Birmingham, Birmingham, UK) and the BASIL-3 team of triallists will address the pressing question of which endovascular strategy wins in the disputed femoropopliteal segment....
Elixir Medical has announced Breakthrough Device designation by the US Food and Drug Administration (FDA) for its novel DynamX BTK System, an implant for use in the treatment of narrowed or blocked vessels below-the-knee (BTK) in patients with chronic...
Predicting longevity in patients who may be candidates for aortic repair is challenging. This, coupled with a high and unchanging long-term postoperative mortality rate, begs the question: are patients living long enough to benefit from aortic repair?
Bijan Modarai (King’s...
Cook Medical and Bentley today announced a distribution agreement for the BeBack crossing catheter in the USA, with Cook assuming commercial responsibilities for the Bentley product in the coming months.
The BeBack device is designed for steering through chronic total occlusions...
Peca Labs has received an additional CE mark for its exGraft family of vascular grafts, expanding its on-label use.
ExGraft vascular grafts are intended for use as vascular prostheses for replacement or bypass of diseased or injured vessels, and...
Data from a new study that maps out the levels at which adverse postoperative events decrease following transfemoral carotid artery stenting (TfCAS) based on depth of physician experience with the procedure represent an “absolute minimum threshold of learning,” the...
Aquedeon Medical has announced the initiation of an investigational device exemption (IDE) clinical trial to study the Duett vascular graft system.
A press release reveals that, this February, the first enrolled patient underwent open surgical aortic arch reconstruction at the...
Medtronic has received a license from Health Canada for its Symplicity Spyral multi-electrode renal denervation (RDN) system.
This is indicated for the management of essential hypertension in patients for whom blood pressure remains uncontrolled despite lifestyle modifications and guideline-driven medical...
Hans-Henning Eckstein, the vascular surgeon who played a leading role in the SPACE and SPACE 2 randomised controlled trials on the treatment of carotid stenosis and founder of the Munich Vascular Conference (MAC), died on 24 February at the...
Baylis Medical Technologies today announced the 510(k) clearance and launch of the PowerWire Pro radiofrequency (RF) guidewire in the USA, facilitating venous stent recanalisation for total occlusions.
Patients with stenotic or occluded peripheral vessels often undergo stent placement to restore...
Biotronik has been granted Breakthrough Device designation (BDD) from the US Food and Drug Administration (FDA) for the Freesolve below-the-knee (BTK) resorbable magnesium scaffold (RMS).
The Freesolve BTK RMS is designed for individuals suffering from chronic limb-threatening ischaemia (CLTI).
To qualify...
Healthcare Inroads (HCIR)—which aims to deliver value-driven solutions to medical technology companies, healthcare delivery systems, and investors worldwide—has announced its global launch.
HCIR was formed by the merger of Healthcare Insights and Inroad Medical and is a partnership of clinicians...
Eric Ducasse (University of Bordeaux, Bordeaux, France) spoke with Vascular News at this year’s Leipzig Interventional Course (LINC 2023; 6–9 June, Leipzig, Germany) on the use of artificial intelligence (AI) in vascular interventions.
He said there is potential for AI—and...
A large, single-centre analysis has found that, despite parity between the two procedures in terms of perioperative clinical outcomes, carotid endarterectomy (CEA) was associated with a reduced rate of late stroke/death compared to transcarotid artery revascularisation (TCAR).
Writing in...
Switzerland-based startup firm Nanoflex Robotics has announced that it recently installed its first remote-ready robotics system for neurovascular procedures at the Jacobs Institute (Buffalo, USA).
The Nanoflex Robotics system uses a compact magnetic field generator and a navigation control unit...
February's top 10 includes an update from the UK Medicines and Healthcare products Regulatory Agency (MHRA) on the use of paclitaxel-coated devices for peripheral arterial disease (PAD), a talk delivered at the Critical Issues America (CIA) annual meeting (2–4...
Vascular News is pleased to announce Sarah Sillito (Newcastle, UK) as our new Launch Pad columnist. She replaces Claire Dawkins, who helped launch the column—dedicated to issues important to vascular trainees—back in April 2022. Dawkins was recently promoted to...
FastWave Medical has announced the issuance of its third utility patent by the US Patent and Trademark Office (USPTO).
The newly granted patent supports the differentiated design of FastWave's next-generation intravascular lithotripsy (IVL) system, which aims to address challenges associated...
Silk Road Medical has announced the launch of its tapered Enroute transcarotid stent system to hospitals in the USA, expanding upon the company’s prior Enroute transcarotid stent system and offering additional configurations to better tailor the transcarotid artery revascularisation...
Gianmarco de Donato (Siena, Italy) speaks to Vascular News at Paris Vascular Insights (PVI) 2023 (8–10 November, Paris, France) about acute limb ischaemia (ALI), detailing that his treatment approach has changed considerably over the past 10 years. Nowadays, Donato...
BD has announced the enrolment of the first patient in the AGILITY investigational device exemption (IDE) study, which will assess the safety and effectiveness of the BD vascular covered stent for the treatment of peripheral arterial disease (PAD).
The...
Florian Enzmann (Innsbruck, Austria) speaks to Vascular News about PROMOTE ALI—a prospective, multicentre observational study focused on lower limb ischaemia.
Acute limb ischaemia (ALI) is “not very well investigated,” the head of endovascular surgery at the Medical University Innsbruck states,...
This year’s International Stroke Conference (ISC; 7–9 February, Phoenix, USA) hosted discussions on the current state of play in carotid stenosis treatment—and, most notably, advances that may erode the dominance of long-established carotid revascularisation procedures in the USA.
Session moderator...
Shape Memory Medical has announced that it has completed $38 million in Series C financing, which has been led by Earlybird Venture Capital, along with participation from both new and existing investors.
Shape Memory stated in a press release that...
ViTAA Medical Solutions has announced the appointment of Frank J Veith, professor of surgery at the Cleveland Clinic Lerner College of Medicine of Case Western Reserve University (Cleveland, USA) and New York University Medical Center (New York City, USA),...
Endologix has announced the initiation of the Percutaneous transmural arterial bypass (PTAB)1 postmarket study. This study marks the beginning of a comprehensive postmarket study aimed at evaluating the real-world performance of the Detour system in patients undergoing treatment for...
New guidelines from the European Association for Cardio-Thoracic Surgery (EACTS) and the Society of Thoracic Surgeons (STS) have, for the first time, recognised the aorta as “an organ in its own right”.
“Recognising the aorta as an organ puts it...
Royal Philips has announced the launch of their image-guided therapy mobile C-arm system 9000—Zenition 90 Motorized, designed to help surgeons deliver high-quality care to more patients at the European Congress of Radiology (ECR) annual meeting (February 28–3 March, 2024,...
Royal Philips has announced major enhancements to its image guided therapy system—Azurion—with the launch of its new Azurion neuro biplane system.
Featuring enhanced imaging and gantry positioning flexibility, this new angio suite system is designed to streamline neurovascular procedures and...
“Specialist CLTI clinics are inclusive, safe, effective, efficient, and alleviate pressure on vascular units,” Assad Khan (Leeds Vascular Institute, Leeds, UK) stated at the 2023 Vascular Society of Great Britain and Ireland’s (VSGBI) annual scientific meeting (22–24 November,...
Andres Schanzer (UMass Memorial Medical Center, Worcester, USA) has hailed it “one of the most exciting changes” seen in imaging during the course of his career. Philips’ LumiGuide “human GPS” technology—which uses light reflected along an optical fibre inside...
“The future of vascular surgery is incredibly bright,” Daniel Clair (Vanderbilt University Medical Center , Nashville, USA) opines in this interview with Vascular News on his career in the field so far. Having always known he wanted to be...
Cagent Vascular has announced a series C financing close in excess of US$30 million. US Venture Partners (USVP) led the round. Participation included new investor Blue Ridge Medical and existing investors, including Sectoral Asset Management.
“We are pleased with the...
IsomAb Ltd, a UK-based biotechnology company, has announced the closing of a £7.5 million (approximately US$9.4 million) seed financing round, led by Broadview Ventures, with further backing from existing investor, SCVC and participation from MEIF Proof of Concept & Early...
A randomised controlled trial (RCT) of open repair versus endovascular aneurysm repair (EVAR) for the treatment of abdominal aortic aneurysms (AAAs), as well as head-to-head trials for additional information on EVAR and complex repairs, are warranted. This was the...
In this issue:
LIFE-BTK breathes life into drug-eluting resorbable scaffolds in breakthrough for below-the-knee arteries
Could metformin be first-ever medical treatment that is effective at managing aneurysm disease?
In profile: Daniel Clair (Nashville, USA)
Launch Pad: Introducing our new...
In this issue:
LIFE-BTK breathes life into drug-eluting resorbable scaffolds in breakthrough for below-the-knee arteries
Could metformin be first-ever medical treatment that is effective at managing aneurysm disease?
In profile: Daniel Clair (Nashville, USA)
Launch Pad: Introducing our new...
January's top 10 highlights new guidelines from the European Society for Vascular Surgery (ESVS), an expert consensus roundtable on the benefits of intravascular ultrasound (IVUS) in lower extremity revascularisation procedures, and US FDA approval for the Gore Excluder thoracoabdominal...
Efemoral Medical today announced that the US Food and Drug Administration (FDA) has granted its novel Efemoral vascular scaffold system (EVSS) Breakthrough Device status for the treatment of de novo or restenotic lesions of the infrapopliteal arteries in patients...
This advertorial, sponsored by Gore, is only available in selected countries and geographies.
Michel Reijnen (Arnhem, The Netherlands) discusses the clinical relevance of type 2 endoleaks in abdominal aortic aneurysms (AAAs).
Type II endoleaks (T2ELs) are the most prevalent of...
Sensome has announced enrolment of the first patients into a feasibility clinical study using the Clotild smart guidewire in peripheral arterial disease (PAD). Clotild was designated a breakthrough medical device for use in brain arteries by the US Food and...
Humacyte has announced that the US Food and Drug Administration (FDA) has accepted and granted priority review to Humacyte’s Biologics License Application (BLA) seeking approval of the Human Acellular Vessel (HAV) in urgent arterial repair following extremity vascular trauma...
The multi-society PAD Pulse Alliance is taking part in a peripheral arterial disease (PAD) public awareness campaign called “Get a Pulse on PAD”.
The group—formed of the Society for Vascular Surgery (SVS), the Association of Black Cardiologists (ABC), the Society...
Innova Vascular has announced successful early commercial use of the company's Laguna thrombectomy system. Physicians at UCLA Medical Center in Los Angeles, USA, and Providence St Joseph in Orange, USA, who were the first to use this system in...
The use of a relatively novel scoring system, referred to as Symptomatic carotid atheroma inflammation lumen-stenosis (SCAIL), has produced favourable predictive qualities versus the Oxford carotid stenosis tool (OCST) and Essen stroke risk score (ESRS). According to researchers, the...
Following a review, the UK Medicines and Healthcare Products Regulatory Agency (MHRA) has updated guidance on the use of paclitaxel-coated devices stating that such devices can be considered for the treatment of peripheral arterial disease (PAD), including intermittent claudication...
The REDUCED-1 study, a US Food and Drug Administration (FDA) investigational device exemption (IDE)-approved pilot study of the treatment of hypertension with the proprietary Therapeutic Intra-Vascular Ultrasound (TIVUS) system (SoniVie), has completed enrolment.
“Initial results from the ongoing REDUCED-1 study...
Gore today announced the first ever implantation of the Gore Tag thoracic branch endoprosthesis (TBE) in Europe. The patient was treated by Dittmar Böckler, chief of the Department of Vascular Surgery and Endovascular Surgery at University Hospital Heidelberg in...
Cook Medical has announced the first patient treated in the clinical study of the ZENITH FENESTRATED+ Endovascular Graft (ZFEN+) in the USA. The procedure was performed on January 29, 2024 at The University of Texas Health (UTHealth) in Houston, Texas, by...
Rijnstate Hospital in Arnhem, The Netherlands, recently announced that the first patient has been enrolled in the HERCULES trial comparing endosuture aneurysm repair (ESAR) to standard endovascular aneurysm repair (EVAR) in the treatment of abdominal aortic aneurysms (AAAs) with...
Gore has announced recent US Food and Drug Administration (FDA) approval of a lower profile Viabahn VBX balloon expandable endoprosthesis (VBX stent graft).
"Our team is pleased to be the first commercial implanter of the new lower profile VBX stent...
Endospan shared 30-day results from the first 22 patients enrolled in the TRIOMPHE investigational device exemption (IDE) study in a late-breaking presentation at the 60th annual meeting of the Society of Thoracic Surgeons (27–29 January, San Antonio, USA).
TRIOMPHE...
Gore has announced US Food and Drug Administration (FDA) approval for the Gore Excluder thoracoabdominal branch endoprosthesis (TAMBE), which the company states is the first off-the-shelf endovascular solution for the treatment of complex aneurysmal disease involving the visceral aorta.
A...
The updated European Society for Vascular Surgery (ESVS) 2024 clinical practice guidelines for the care of patients with aneurysms of the abdominal aorta and iliac arteries have been published online this month in the European Journal of Vascular and...
Vascular News caught up with European Society for Vascular Surgery (ESVS) president Ian Loftus (London, UK) at the Vascular for Society of Great Britain and Ireland’s (VSGBI) 2023 annual scientific meeting (VSASM; 22–24 November, Dublin, Ireland) to discuss the...
“Everyone in this room, whether you believe it or not, is a leader and therefore a role model,” Neeraj Bhasin (Huddersfield, UK) told the Vascular Society of Great Britain and Ireland’s (VSGBI) 2023 annual scientific meeting (VSASM; 22–24 November,...
AngioDynamics today announced that the US Food and Drug Administration (FDA) has cleared the Auryon XL catheter, a 225cm radial access catheter, for use with the Auryon atherectomy system in the treatment of peripheral arterial disease (PAD).
According to an AngioDynamics...
Surmodics has announced successful early clinical use of the company’s Pounce LP (low-profile) thrombectomy system. The Pounce LP system, which received US Food and Drug Administration (FDA) clearance in June 2023, is currently in limited market evaluation (LME), with full...
The final publication of midterm results from the observational CARAS study suggest that cerebral ischaemic event (CIE) incidence in asymptomatic carotid stenosis patients “should not be underestimated”, so say authors Rodolfo Pini (University of Bologna, Italy) and colleagues, relaying...
Medtronic recently announced that Simona Zannetti has been appointed as the general manager of its aortic business.
Zannetti previously spent 21 years at Medtronic. She returns after serving as senior vice president of clinical research at Penumbra, where she helped...
An analysis of data from the SPYRAL HTN-ON MED trial of the Symplicity Spyral (Medtronic) radiofrequency denervation system for the treatment of uncontrolled hypertension has found that the technique can be expected to provide “good value for money” as...
Summa Therapeutics has announced that the first-in-man injectable angioplasty procedures for patients with below-knee peripheral arterial disease (PAD) were performed successfully using its Finesse Injectable balloon catheter platform.
The Summa Finesse injectable catheter is the industry’s first hybrid diagnostic and...
Nectero Medical today announced initiation of a Phase II/III clinical trial (stAAAble) to investigate the safety and efficacy of the Nectero Endovascular Aneurysm Stabilisation Treatment (Nectero EAST) system in patients with infrarenal abdominal aortic aneurysms (AAAs), maximum diameter 3.5–5cm.
A...
FastWave Medical has announced the successful completion of enrolment for its first-in-human (FIH) study with the company's peripheral intravascular lithotripsy (IVL) technology.
The prospective, single-arm study aims to assess the safety and performance of FastWave's IVL system in patients with...
Reflow Medical recently announced it has received CE mark certification in the European Union for the Bare Temporary Spur stent system. The device is intended to treat de novo or restenotic lesions in the infrapopliteal arteries with a commercially...
Laminate Medical has announced the first implantation of their VasQ device in the USA, performed by Ari Kramer (Spartanburg Regional Hospital, Spartanburg, USA). The VasQ technology was granted US Food and Drug Administration (FDA) approval in September 2023, and...
Gore has announced that the first patients have been enrolled in the Gore VBX FORWARD clinical study, a global prospective, multicentre, randomised controlled trial to compare the Gore Viabahn VBX balloon expandable endoprosthesis (VBX stent graft) to bare metal...
“I don’t reflect back enough on how much things have changed,” Cynthia Shortell (Durham, USA) tells Vascular News at the 2023 VEITHsymposium (14–18 November, New York, USA), speaking on the representation of women in vascular surgery over the course...
Carotid artery stenting (CAS) delivered via transradial artery access has demonstrated no increased risk of stroke, death, myocardial infarction, transient ischaemic attack, or access site complications, as compared to the more traditional transfemoral approach. That is the concluding finding...
A dedicated session at the 2023 VEITHsymposium (14–18 November, New York, USA) aimed to unpack the ways in which clinical practice and attitudes in the field of chronic limb-threatening ischaemia (CLTI) have changed since the BEST-CLI trial was published...
The Intervention Master Aortic Course (IMAC) Board provides an overview of the most recent iteration of its annual event, which in 2023 took place in Cairo, Egypt. The event featured a Charing Cross (CX) session introduced by the late...
Extolling the benefits of evidence-based practice, timely surgical intervention and intensive medical therapy, Dominic PJ Howard (Oxford, UK) spoke at the Vascular Society of Great Britain and Ireland's (VSGBI) 2023 annual scientific meeting (VSASM; 22–24 November, Dublin, Ireland) on...
Endovascular Engineering (E2) has announced US Food and Drug Administration (FDA) investigational device exemption (IDE) approval for its ENGULF US pivotal trial.
The study will evaluate the safety and effectiveness of the Hēlo thrombectomy system in treating pulmonary embolism (PE)....
A recent study published in Circulation concerning patients with chronic limb-threatening ischaemia (CLTI) in the USA, has found that patients of low-income status and those who attended “safety-net” hospitals—facilities that treat patients regardless of insurance status or ability to...
Cook Medical has announced that its Slip-Cath Beacon Tip hydrophilic selective catheter is now available for use in the USA and Canada. The company shares in a press release that it is proud to bring back this product—a hydrophilic...
Proceedings from an expert consensus roundtable that discussed the benefits of intravascular ultrasound (IVUS) in lower extremity revascularisation procedures were released today in the Journal of the Society for Cardiovascular Angiography & Interventions (JSCAI), Journal of Vascular and Interventional...
Principal investigators of the LIFE-BTK randomised controlled trial delve into the technological improvements underpinning the Esprit drug-eluting resorbable scaffold (DRS; Abbott) from its previous iteration used in the coronaries, while discussing how this trial places this device at the...
"Despite significant technological improvements since its inception, endovascular repair of abdominal aortic aneurysms (AAAs) is still associated with some of the same problems we experienced nearly 30 years ago," Michel Reijnen (Arnhem, The Netherlands) remarked at the VEITHsymposium 2023...
Endologix recently announced that the US Centers for Medicare & Medicaid Services (CMS) has granted a Transitional Pass-Through (TPT) payment for the Detour system, effective since 1 January 2024.
The TPT payment was created to facilitate patient access for qualifying...
Gore has announced the first patient implantation of the Gore ascending stent graft in the ARISE II trial, describing this as an exciting step in the development of treatments for pathologies involving the ascending aorta using endovascular repair rather...
“We’re going to see more and more amputations if we don’t figure out the right post procedure thromboprophylaxis regime ASAP,” Anahita Dua (Boston, USA) warned during a lecture on innovative approaches to preventing amputation at the Vascular Society of...
Peter Schneider (University of California San Francisco, San Francisco, USA) speaks to Vascular News about his life and career. Trained during the era of open surgery only, Schneider saw the benefits of endovascular intervention early on, writing Endovascular Skills...
Microbot Medical, developer of the Liberty endovascular robotic surgical system, has entered into a collaboration agreement with Corewell Health.
The objective of the collaboration, which will take place in multiple phases, is to enable telerobotics between remote centres by utilising...
Shape Memory Medical announced this week that it has entered into a sublicense agreement with a strategic global medical device company.
Under the agreement, Shape Memory Medical will sublicense its proprietary shape memory polymer technology for a narrow high-growth...
NOTE: This video is ONLY available to watch in selected countries and geographies
Vascular surgeon Raphael Coscas (Paris, France) and interventional cardiologist, Antoine Sauguet (Toulouse, France) and discuss the design and first results from the RADIANCY premarket clinical study,...
Further reports on the use of atherectomy in the USA this week appeared in ProPublica. The nonprofit investigative news outlet shared three articles in its latest round of investigations looking into Medicare claims data.
The articles, led by a story...
Humacyte today announced that it has submitted a Biologics License Application (BLA) to the US Food and Drug Administration (FDA) seeking approval of the Human Acellular Vessel (HAV) in urgent arterial repair following extremity vascular trauma when synthetic graft...
Vivasure Medical has announced the first patient treated with PerQseal Elite, the latest generation of the company’s fully absorbable large-hole closure system.
The first successful implants were completed by Nicolas Van Mieghem at Erasmus University Hospital in Rotterdam, The Netherlands,...
The European Union Horizon Europe, with joint funding from UK Research Innovation, has awarded the NetZeroAICT Consortium €6 million in funding to develop a novel artificial intelligence (AI)-based computed tomography (CT) technology that has the potential to promote climate...
Micro Medical Solutions (MMS), an innovator in the field of microvascular intervention to improve clinical outcomes and quality of life in patients with chronic limb-threatening ischaemia (CLTI), announced that it has completed its USA pivotal clinical study enrolment evaluating...
Kaneka Corporation has acquired all shares of Japan Medical Device Technology Co and has made it a wholly owned subsidiary.
Kaneka manufactures and sells endovascular catheters, which are devices used in the treatment of cardiac, peripheral vascular, and cerebrovascular diseases....
Summa Therapeutics, a specialty angioplasty balloon medical device company, announced today that its innovative line of injectable angioplasty balloon catheters has been cleared for marketing by the US Food and Drug Administration (FDA) for the treatment of peripheral artery...
There is a global interest in assessing whether Metformin, which has a long track record of safety and efficacy, is relatively inexpensive and is taken by millions of people every day for type 2 diabetes, has an effect on...
Daniel Clair (Vanderbilt University Medical Center, Nashville, USA) speaks to Vascular News at this year’s VEITHsymposium (14–18 November, New York, USA) on deep vein arterialisation with the LimFlow system (LimFlow). The technology aims to fill a treatment gap...
Inari Medical has announced the first patient enrolment in PEERLESS II. This prospective, global, multicentre randomised controlled trial (RCT) compares the outcomes of intermediate-risk acute pulmonary embolism (PE) patients treated with the FlowTriever system against those treated with traditional...
Penumbra today announced that the first patient has been enrolled in STORM-PE, a prospective, multicentre, randomised controlled trial (RCT) evaluating anticoagulation alone versus anticoagulation plus Lightning Flash for the treatment of pulmonary embolism (PE).
In partnership with PERT Consortium,...
Biotronik has announced the presentation of two-year results from the investigator-initiated BIOPACT randomised controlled trial (RCT) by principal investigator Koen Deloose (AZ Sint-Blasius Hospital, Dendermonde, Belgium) at the Paris Vascular Insights (PVI) 2023 congress (8–10 November, Paris, France). The...
Inari Medical has announced that it has completed its acquisition of chronic limb-threatening ischaemia (CLTI) treatment maker LimFlow.
The Irvine, California–based company earlier this month announced plans to acquire LimFlow for up to US$415 million. The deal came just weeks...
Lawrence Garcia (St Francis Hospital, New York, USA) speaks to Vascular News at VIVA 2023 (30 October–2 November, Las Vegas, USA) on lessons learned from a series of failed below-the-knee (BTK) trials that preceded the recent “first victory” of...
Vincent Rowe (University of California Los Angeles, Los Angeles, USA) speaks to Vascular News at VIVA 2023 (30 October–2 November, Las Vegas, USA) on the importance of patient-specific decisions for the management of vascular wounds.
He also considers how multidisciplinary...
Peter Schneider (University of California San Francisco, San Francisco, USA) recently presented 36-month data from Surmodics' TRANSCEND clinical trial at the VEITHsymposium 2023 (14–18 November, New York, USA).
The TRANSCEND trial is a prospective, multicentre, single-blind randomised controlled trial to...
Cordis has announced the completion of patient enrolment in the RADIANCY premarket clinical study. The prospective, multicentre, single-arm study is designed to evaluate the safety and efficacy of the S.M.A.R.T. Radianz vascular stent system when used with the Brite...
Medtronic has announced approval from the USA Food and Drug Administration (FDA) for the Symplicity Spyral renal denervation (RDN) system for the treatment of hypertension. The company has said that it will immediately begin commercialisation following the approval.
“Medtronic has...
This advertorial is sponsored by Cardionovum.
Cardionovum’s CE-marked Aperto drug-coated balloon (DCB) is, according to expert opinion and a growing evidence base, spearheading a revolution in the realm of vascular access maintenance for dialysis patients.
This high-pressure DCB has been specifically...
Xeltis has announced "highly encouraging" 12-month results from its first-in-human (FIH) aXess vascular conduit trial. The data were presented by Frans Moll (University Medical Center Utrecht, Utrecht, The Netherlands) at this year's VEITHsymposium (14–18 November, New York, USA).
At 12...
An ongoing UK National Institute for Health and Care Research (NIHR)-funded randomised controlled trial seeks to illuminate the value of an image fusion guidance technology for endovascular aortic repair.
In a presentation on the trial at this year’s VEITHsymposium (14–18...
Two-year data from the SWING trial, a first-in-human study of the safety and performance of the Sundance sirolimus drug-coated balloon (DCB; Surmodics), have been presented at this year's VEITHsymposium (14–18 November, New York, USA).
The SWING trial is a 35-patient...
New analyses from the VOYAGER PAD clinical trial in both high-risk and fragile patients and those with and without comorbid coronary artery diseases (CAD) were presented at the American Heart Association (AHA) 2023 Scientific Sessions (11–13 November, Philadelphia, USA).
The...
Vivasure Medical has enrolled the 100th patient in its PATCH clinical study, a multicentre, single-arm, pivotal study evaluating the safety and efficacy of the Vivasure PerQseal closure device system.
The US Food and Drug Administration (FDA) granted investigational device exemption...
Shape Memory Medical, developer of custom shape memory polymers for endovascular markets, announced that one-year data from the AAA-SHAPE safety trial will be presented during the 50th annual VEITHsymposium (14–18 November, New York City, USA) on Tuesday, November 14,...
A new study published in BJS Open has found that revascularisation treatment to restore blood flow to the legs of patients with severely blocked arteries is no more expensive than carrying out an amputation.
The peer-reviewed paper is the first to compare...
Cordis has announced that its board of directors has appointed Scott Drake as chief executive officer (CEO) effective immediately. Drake most recently held the position of executive chairman of the Cordis board of directors. He will succeed Shar Matin,...
Recor Medical has become the first company in the USA to have a device-based therapy approved for the treatment of hypertension, after it was announced that the US Food and Drug Administration (FDA) has approved the company’s Paradise ultrasound...
Patients undergoing computer-assisted vacuum thrombectomy with the Indigo Aspiration System (Penumbra) for pulmonary embolism (PE) showed 2.7% rates of both major adverse events (MAEs) and major bleeding at 48 hours post-procedure alongside “a significant reduction” in right ventricle/left ventricle...
One-year results from the PROMISE II US pivotal trial demonstrate the durability of outcomes of transcatheter arterialisation of the deep veins (TADV) using the LimFlow system (LimFlow) and that the procedure is safe and reproducible.
Daniel Clair (Vanderbilt University Medical...
Endologix has announced results from a pooled analysis of the DETOUR1 and DETOUR2 studies evaluating percutaneous transmural arterial bypass (PTAB) with the Detour system.
A press release details that PTAB with the Detour system offers a novel approach to treating...
InspireMD recently presented 30-day results from the C-GUARDIANS US investigational device exemption (IDE) clinical trial evaluating its CGuard embolic prevention stent (EPS) system for stroke prevention at VIVA 2023 (30 October–2 November, Las Vegas, USA). The presentation, which was...
Contego Medical has announced the presentation of late-breaking clinical results from the PERFORMANCE II carotid stent trial at VIVA 2023 (30 October–2 November, Las Vegas, USA). PERFORMANCE II is a prospective, multicentre study evaluating the safety and effectiveness of...
Nectero Medical today announced that the US Food and Drug Administration (FDA) has granted Breakthrough Therapy designation for the Nectero Endovascular Aneurysm Stabilisation Treatment (Nectero EAST) system to treat patients with infrarenal abdominal aortic aneurysms (AAAs), maximum diameter 3.5–5cm....
LimFlow announced today that it has entered into a definitive agreement to be acquired by Inari Medical.
A press release details that, under the terms of the agreement, LimFlow will receive US$250 million in cash at closing and be eligible...
Surmodics has announced the commercial launch of its most advanced hydrophilic medical device coating technology, the Preside hydrophilic coatings.
As per a company press release, Preside hydrophilic coatings complement Surmodics’ existing Serene hydrophilic coatings by providing customers with a unique,...
Endovascular Engineering (E2) has announced positive initial results from its ENGULF study, demonstrating successful outcomes among the first 15 patients evaluated in the trial using the Hēlo thrombectomy system for the treatment of acute sub-massive pulmonary embolism.
The ENGULF Study...
New research from the FLASH registry shines a light on the effectiveness of large-bore mechanical thrombectomy in managing high-risk pulmonary embolism. The study, titled "Mechanical thrombectomy for high-risk pulmonary embolism: Insights from the US cohort of the FLASH registry," provides valuable insights into this life-saving...
Results from a first-in-human, prospective, single-arm study of the Akura Medical thrombectomy system (Akura Medical) for pulmonary embolism (PE) were revealed this week at The VEINS 2023 (28–30 October, Las Vegas, USA).
Presenter Jay Mathews (Manatee Memorial Hospital, Bradenton, USA)...
The open-label phase of the DEXTERITY-AFP trial investigating the Bullfrog microinfusion device—which involves the perivenous injection of the anti-inflammatory drug dexamethasone to improve patency and post-thrombotic syndrome (PTS) six months after thrombus removal in symptomatic deep vein thrombosis (DVT)...
A secondary analysis of existing clinical trial data has indicated that new ischaemic brain lesions detected via diffusion-weighted magnetic resonance imaging (DW-MRI) following carotid artery revascularisation—either with carotid artery stenting (CAS) or carotid endarterectomy (CEA)—do not appear to have...
David Dexter (Sentara Vascular Specialists, Norfolk, USA) shared one-year data from the CLOUT registry this week at The VEINS 2023 (28–30 October, Las Vegas, USA).
Long-term, prospective outcome data after mechanical thrombectomy for deep vein thrombosis (DVT) are lacking. The...
Getinge has announced commercial availability of the iCast covered stent system in the USA for the treatment of iliac arterial occlusive disease. In March 2023, the iCast covered stent received premarket approval from the US Food and Drug Administration...
Vein360 has announced it received US Food and Drug Administration (FDA) 510(k) clearance for reprocessing the Philips Visions PV 0.035 digital intravascular ultrasound (IVUS) catheter in June of 2023 and the Visions PV 0.018 digital IVUS catheter in August...
In this issue:
Obituary: Roger M Greenhalgh 6th February 1941 – 6th October 2023
Forty five-year legacy of CX vascular education continues: The new CX Symposium co-chairs look ahead to CX 2024
In profile: Peter Schneider (San Francisco, USA)
Claire Dawkins (Newcastle,...
In this issue:
Obituary: Roger M Greenhalgh 6th February 1941 – 6th October 2023
Forty five-year legacy of CX vascular education continues: The new CX Symposium co-chairs look ahead to CX 2024
In profile: Peter Schneider (San Francisco, USA)
...
Rachel Bell (Newcastle, UK) shares her thoughts on a recent UK survey that highlights the prevalence of sexual misconduct within surgery and advises what should be done to address the issue.
Sexual harassment and misconduct in medicine is sadly too...
A survey of more than 1,400 individuals in the UK reveals that, in the past five years, nearly a third of female surgeons reported having been sexually assaulted by a colleague and over two-thirds reported having been the target...
Data from the REAL-PE study were presented this week at TCT 2023 (23–26 October, San Francisco, USA) demonstrating that patients treated for pulmonary embolism (PE) with the Ekos endovascular system (Boston Scientific) had lower rates of adverse events, including...
Thrombolex has announced never-before-reported major reductions in obstruction in all of the segmental pulmonary arteries (PA), based on independent core lab data analysis of 107 patients from 18 sites in the USA, with acute intermediate-risk pulmonary embolism (PE), using...
Speaking at TCT 2023 (23–26 October, San Francisco), vascular surgeon Ramon Varcoe (Sydney, Australia) reflects on the wider impact of the results of the LIFE-BTK trial on chronic limb-threatening ischaemia (CLTI) treatment.
Vascular surgeon Peter Schneider (San Francisco, USA) gives some context to results from the LIFE-BTK trial after they were presented at TCT 2023 (23–26 October, San Francisco), highlighting previous failed trials in the BTK segment.
Results of the LIFE-BTK randomised controlled trial have just been presented at TCT 2023 (23–26 October, San Francisco, USA). The data show that, in patients with chronic limb-threatening ischaemia (CLTI) due to infrapopliteal artery disease, an everolimus-eluting resorbable scaffold...
Data from a patient-level meta-analysis—a factor in the US Food and Drug Administration’s (FDA) recent change of position on the use of paclitaxel-coated devices to treat peripheral arterial disease (PAD)—have been made public for the first time at TCT...
Robocath has today announced the results of its CARE clinical trial focusing on robotic carotid stenting—the first phase of a research programme launched in July 2021 by Robocath and Rennes University Hospital (Rennes, France), which, in the long term,...
Evident Vascular, a medical technology startup developing an intravascular ultrasound (IVUS) platform leveraging artificial intelligence to enable superior imaging and streamlined workflows, recently announced that it has launched with a US$35 million Series A financing from Vensana Capital.
“The impetus...
Cardio Flow recently announced it has received US Food and Drug Administration (FDA) 510(k) clearance for the company’s FreedomFlow orbital atherectomy peripheral platform.
The company details in a press release that the FreedomFlow platform is designed with a modern mechanism...
Alcami has welcomed the recent announcement by Nectero Medical that it has received clearance from the US Food and Drug Administration (FDA) of their investigational new drug (IND) application to initiate a Phase II/III trial of the Nectero Endovascular...
New research examining the safety and efficacy of using radial access for peripheral artery interventions has found that radial access allows early ambulation and same-day discharge with no serious adverse events.
These are the conclusions of a study published in...
NOTE: This video is ONLY available to watch in selected countries and geographies
The EffPac trial was a prospective, multicentre, randomised controlled trial (RCT) that enrolled 171 patients of Rutherford category two to four with medium-length femoropopliteal lesions. Patients...
While sex disparities regarding the outcomes of carotid revascularisation have “long been a concern”, new prospective data published in the Journal of Vascular Surgery (JVS) indicate that no such disparity exists between male and female patients treated with transcarotid...
Discussions on the role of aritifical intelligence in the vascular field and emerging deep venous valve technologies caught readers' attention in September.
1. Humacyte announces positive top line results from Phase 2/3 trial of Human Acellular Vessel in treatment of...
Three newly appointed Charing Cross International Symposium (CX) co-chairs will ensure Roger Greenhalgh’s inspiring legacy of vascular education continues at CX 2024 with the 46th CX Symposium. Dittmar Böckler (Heidelberg, Germany), Andrew Holden (Auckland, New Zealand) and Erin Murphy...
Contego Medical has announced enrolment of the first patient in the prospective, multicentre PERFORMANCE III trial aimed at further evaluating the safety and effectiveness of the Neuroguard integrated embolic protection (IEP) direct system—a carotid stent system designed for direct transcarotid...
Roger Malcolm Greenhalgh, the surgeon internationally renowned for his unparalleled contribution to vascular education, training and research, died peacefully on 6th October, aged 82. At the time of his death, he was emeritus Professor of Surgery at Imperial College...
Artivion has announced presentation of interim results from the Ascyrus medical dissection stent (AMDS) PERSEVERE clinical trial in a late-breaking science session at the 37th European Association for Cardio-Thoracic Surgery (EACTS) annual meeting (4–7 October, Vienna, Austria).
Fernando Fleischman (Keck...
An investigator-sponsored clinical study conducted at the Mayo Clinic in Rochester, Minnesota, of the investigational Human Acellular Vessel (HAV) in patients with chronic limb-threatening ischemia (CLTI) was presented at the Midwestern Vascular Surgical Society (MVSS) annual meeting in Minneapolis,...
MedAlliance has been acquired by Cordis for a 2022 investment of US$35 million and a 2023 upfront closing payment of US$200 million, together with regulatory achievement milestones of up to US$125 million and commercial milestones of up to US$775...
A vascular surgery team from the University of Southern California (USC) in Los Angeles demonstrated the feasibility of multidisciplinary direct revascularization of segmental arteries to prevent spinal cord ischemia (SCI) using novel endovascular or extra-anatomic bypass techniques in high-risk...
Laminate Medical Technologies has announced their flagship device, the VasQ External Vascular Support, has been cleared by the US Food and Drug Administration (FDA) for use to create arteriovenous fistulas (AVFs) for dialysis access. The device, designated by the...
Shockwave Medical has announced that Nick West has joined the company in the role of associate chief medical officer (CMO). He will report to current CMO Keith Dawkins, and will be based in Santa Clara (USA) with plans to...
Biotronik recently announced two-year-results from the BIOLUX P-III BENELUX all-comers registry, presented by principal investigator Frank Vermassen (UZ Ghent, Ghent, Belgium) at the Cardiovascular and Interventional Radiological Society of Europe (CIRSE) annual congress 2023 (9–13 September, Copenhagen, Denmark). The prospective,...
The pursuit of a cure for deep venous valvular reflux—long considered to be the “holy grail” of deep venous disease—is underway, with new technologies set to address a longstanding unmet clinical need across the globe.
Chronic venous disease affects nearly...
Can we trust artificial intelligence (AI)? Will AI replace physician judgement? Experts recently addressed these and other key questions amidst an expansion of AI technologies in the vascular space.
AI is “here to stay”. This is according to Randy Moore...
Nicolas J Mouawad (McLaren Health System, Bay City, USA) urges vascular surgeons to “get out of their comfort zone” and become more involved in pulmonary embolism (PE) care.
With over one million cases of deep vein thrombosis (DVT) and/or PE...
“With effective use of apron and ceiling and table shields, operator doses can be reduced to the equivalent of one to two days of natural background radiation,” investigators write in the conclusion of a recently published study.
Richard W...
Humacyte has announced positive top line results from its V005 Phase 2/3 trial of the Human Acellular Vessel (HAV) in vascular trauma repair.
A press release reports that the single-arm clinical trial was a success and showed that the...
The US Food and Drug Administration (FDA) today announced approval of LimFlow therapy—a novel and minimally invasive procedure designed to bypass blockages in arteries of the legs and restore blood flow for many thousands of people suffering from chronic...
The Society for Vascular Surgery’s Patient Safety Organization (SVS PSO) has launched a national smoking cessation initiative. It includes tools and resources to help physicians, surgeons and other healthcare professionals to help their patients kick the smoking habit....
FastWave Medical recently announced the swift closure of an oversubscribed multi-million dollar private financing. A press release states that this achievement, secured in just a few weeks, highlights the growing interest and confidence in FastWave's ability to execute its...
Endologix has announced that the US Centers for Medicare & Medicaid Services (CMS) recently granted a New Technology Add-on Payment (NTAP) for the Detour system, a US Food and Drug Administration (FDA)-designated Breakthrough Device, as part of its Fiscal...
The Society for Vascular Surgery (SVS), American Venous Forum (AVF), and American Vein and Lymphatic Society (AVLS) have released the second and final part of new guidelines for the management of varicose veins of the lower extremities. The recommendations,...
Shape Memory Medical recently announced that the US Food and Drug Administration (FDA) has granted investigational device exemption (IDE) for the company to begin a prospective, multicentre, randomised, open-label trial to determine safety and effectiveness of the Impede-FX RapidFill...
David Bosanquet (South East Wales Vascular Network, Cardiff, UK) is chief investigator of a new randomised trial—PLACEMENT—which addresses the topic of pain control around the time of a major amputation. Here, he speaks to Vascular News about the importance...
This advertorial, sponsored by Gore, is only available in selected countries and geographies.
In conversation with CX Vascular, Dennis Gable (The Heart Hospital Baylor Plano, Plano, USA) and Ross Milner (University of Chicago, Chicago, USA) outline how long-term data...
“The median thrombus age of DVT patients treated in our centre is 14 days – says Dr Andrew Wigham from Oxford University Hospitals. – We know that traditional treatment options such as thrombolytics and other thrombectomy devices are less...
In this issue:
Paclitaxel-coated devices: US FDA removes red flag after review finds data do not support mortality risk
“A hugely exciting time”: Experts focus in on artificial intelligence in the vascular field
In profile: Jürgen Falkensammer (Vienna, Austria)
Journal...
In this issue:
Paclitaxel-coated devices: US FDA removes red flag after review finds data do not support mortality risk
"A hugely exciting time": Experts focus in on artificial intelligence in the vascular field
In profile: Jürgen Falkensammer (Vienna, Austria)
...
The first US patient has been treated as part of the SOCRATES trial (Short neck AAA randomised trial—ESAR and FEVAR), which compares the safety and performance of endosuture aneurysm repair (ESAR) with fenestrated endovascular aneurysm repair (FEVAR) for the...
NOTE: This video is ONLY available to watch in selected countries and geographies
The benefits of Shockwave Medical’s new Shockwave L6 Peripheral Intravascular Lithotripsy (IVL) catheter—designed for use in large, calcified vessels (available in 8-12 mm diameter sizes), —were highlighted...
The UK’s Medicines and Healthcare products Regulatory Agency (MHRA) has designated three new approved bodies to increase the country’s capacity to certify medical devices.
TÜV SÜD, Intertek, and TÜV Rheinland UK join the four current UK Approved Bodies, almost doubling...
“A truly novel and potentially groundbreaking treatment strategy for patients with small to mid-sized infrarenal abdominal aortic aneurysms,” is how Michael R. Jaff (Boston, USA) describes the Nectero Endovascular Aortic Stabilization Treatment (Nectero EAST®) System from Nectero Medical in...
This advertorial is sponsored by BD.
There has been major progress in the treatment of peripheral arterial disease (PAD) globally in the last few years, “but there are still issues that need to be addressed”, Marianne Brodmann (Medical University of...
This advertorial is sponsored by Boston Scientific.
Data on hundreds of thousands of patients who have received lower limb endovascular treatment provide confidence in paclitaxel safety. This central message from a Boston Scientific symposium at the recent Leipzig Interventional Course...
The Society for Cardiovascular Angiography & Interventions (SCAI) has welcomed the review of scientific data related to the premarket approval applications for two renal denervation systems, describing this as a “step forward to advancing access to additional therapies for...
The US Food and Drug Administration (FDA) Circulatory Systems Devices Panel met yesterday (23 August) to review evidence of the safety and efficacy of Medtronic’s Symplicity Spyral renal denervation system, with committee members split on the motion that the...
Nectero Medical today announced that the US Food and Drug Administration (FDA) has granted Fast Track designation for the Nectero Endovascular Aneurysm Stabilisation Treatment (Nectero EAST) system to treat patients with infrarenal abdominal aortic aneurysms (AAAs) of maximum diameter 3.5–5cm....
Aquedeon Medical has announced its receipt of US Food and Drug Administration (FDA) investigational device exemption (IDE) approval to conduct a staged pivotal clinical trial for the Duett vascular graft system in the USA. The study will be initiated...
The US Food and Drug Administration (FDA) Circulatory Systems Devices Panel of the Medical Devices Advisory Committee yesterday (22 August) voted that there is sufficient data to support the use of the Paradise ultrasound renal denervation system (Recor Medical)...
Cydar Medical has announced the successful treatment of the first patient in a strategic collaboration with Medtronic. The collaboration, which spans 40 sites across the USA, UK and Europe, involves the integration of the company’s Cydar Maps software with...
NOTE: This video is ONLY available to watch in selected countries and geographies
Experts gathered at the Society for Vascular Surgery’s 2023 Vascular Annual Meeting (VAM) in National Harbor, Maryland (June 14–17) to discuss the latest innovation from Shockwave...
Clinical outcomes of Humacyte's investigational Human Acellular Vessel (HAV) were presented at the recent Military Health System Research Symposium (14–17 August, Kissimmee, USA), following a year-long humanitarian program in Ukraine. The bioengineered, implantable human tissue-based device was provided to...
The Society for Cardiovascular Angiography & Interventions (SCAI) has released a position statement on renal denervation, in which it emphasises the importance on patient selection, optimal techniques, competence, training, and institutional recommendations in the use of the technique as...
A new study in the Journal of Vascular Surgery (JVS) finds that a patient’s postcode is a crucial indicator for advanced stages of an abdominal aortic aneurysm (AAA) at the time of endovascular aneurysm repair (EVAR) with a higher...
MedAlliance has announced completion of patient enrolment in the SAVE clinical trial with the Selution Sustained Limus Release (SLR) 018 drug-eluting balloon (DEB) for the treatment of failed arteriovenous fistulas (AVFs) in renal dialysis patients. Selution SLR is a...
July's top 10 highlights the US Food and Drug Administration's long-awaited update on paclitaxel-coated devices to treat peripheral arterial disease, an interview with Rachel Bell (Newcastle, UK), and some key industry updates.
1. FDA removes red flag for paclitaxel-coated devices...
“PERT care is the coalition of the willing,” quipped Dennis Gable, a vascular surgeon at Texas Vascular Associates in Plano, USA. He was referring to team-based care for pulmonary embolism (PE) patients and how, unlike acute stroke intervention and...
Merit Medical has announced that is has completed enrolment in its WRAPSODY arteriovenous access efficacy (WAVE) pivotal study. Merit’s WAVE study is a prospective, randomised, controlled, multicentre study comparing the Merit WRAPSODY cell-impermeable endoprosthesis (CIE) to percutaneous transluminal angioplasty...
Alucent Biomedical has announced that the US Food and Drug Administration (FDA) granted an investigational device exemption (IDE) for a US clinical study of AlucentNVS, a unique light-activated, drug-coated balloon catheter technology.
AlucentNVS combines an intravascular device with a photochemical...
Pathfinder Medical has been awarded a Smart grant for a £1.1 million (US$1.4 million) project by Innovate UK, the UK's innovation agency. A press release details that the grant will support Pathfinder’s novel project to develop a minimally invasive...
The characterisation of aortic tissue by means of three key biomechanics-based biomarkers bundled into a compound Regional Areas of Weakness (RAW) Map showed “very good performance” as part of an artificial intelligence (AI)-based prediction of faster than average growth...
A specialist in complex open aortic surgery, Rachel Bell speaks to Vascular News about her career so far. Raised in Yorkshire, UK, she moved to London for medical school before recently making the move back to the north of...
RapidAI today announced US$75 million in Series C funding led by Vista Credit Partners, a subsidiary of Vista Equity Partners and strategic financing partner focused on the enterprise software, data and technology markets.
A press release details that the funding aims to...
Humacyte today announced completion of enrolment in its Phase II/III vascular trauma trial (V005) that is expected to support a Biologics License Application (BLA) filing for the company's Human Acellular Vessel (HAV) in vascular trauma repair.
A press release details...
The Society for Vascular Surgery (SVS) Foundation has announced the appointment of Bernadette Aulivola and Vikram Kashyap to its board of directors. Both will serve two-year terms and, with the board's collective knowledge and experience, will further bolster the...
Medical device company Access Vascular has announced the publication of a peer-reviewed study of its HydroPICC peripherally inserted central catheter (PICC) in the Journal of Materials Science: Materials in Medicine. The retrospective study found that HydroPICC “significantly reduced” clot formation...
Nectero Medical today announced that the US Food and Drug Administration (FDA) has granted investigational new drug (IND) clearance for the company to initiate a prospective, multicentre, randomised clinical trial (the stAAAble study) to evaluate the safety and efficacy...
Endologix has shared in a press release that the first patients underwent percutaneous transmural arterial bypass (PTAB) using the Detour system since US Food and Drug Administration (FDA) approval of the system was granted. This marks the official start...
In an official Society response, SVS president Joseph Mills tackles recent coverage in the mainstream media of inappropriateness in vascular care.
There have been several recently published articles by respected news media outlets highlighting the deeply concerning issue of the...
The president of the Society for Cardiovascular Angiography & Interventions (SCAI), George Dangas (Icahn School of Medicine at Mount Sinai, New York, USA), has welcomed the US Food and Drug Administration (FDA) decision to remove restrictions on the use...
In June 2023, the Cardiovascular Interventional Radiological Society of Europe (CIRSE) published an editorial in Cardiovascular Interventional Radiology (CVIR). Written on behalf of CIRSE’s Endovascular Subcommittee, the authors “advocate that the benefits of paclitaxel-coated device used in the femoropopliteal...
“There is an increasing awareness that we need to get aneurysm sacs to regress after endovascular aneurysm repair ,” Andrew Holden (Auckland, New Zealand) stated during a recent discussion with Michel Reijnen (Arnhem, The Netherlands) on potential solutions in...
Highlights from this year’s Society for Vascular Surgery (SVS) Vascular Annual Meeting (VAM 2023; 14–17 June, National Harbor, USA) feature prominently in June's top 10.
1. “Sonolysis is back” after safely reducing endarterectomy stroke risk in randomised trial
Positive results from a clinical...
At this year’s Leipzig Interventional Course (LINC 2023; 6–9 June, Leipzig, Germany), Sabine Steiner (Leipzig, Germany) shared first results from the BEST superficial femoral artery (SFA) pilot study—a prospective, multicentre randomised trial comparing the efficacy and safety of a...
Biotronik has announced one-year subgroup results from the investigator-initiated BIOPACT randomised controlled trial (RCT), which were presented by principal investigator Koen Deloose (AZ Sint-Blasius, Dendermonde, Belgium) at this year’s Leipzig Interventional Course (LINC 2023, 6–9 June, Leipzig, Germany).
The randomised...
In an interview with Venous News at this year’s Leipzig Interventional Course (LINC 2023; 6–9 June, Leipzig, Germany), Raghu Kolluri (Columbus, USA) outlines the “vast list” of options that are now available for the treatment of patients who require...
The release of a letter from the US Food and Drug Administration (FDA) concerning the use of paclitaxel-coated devices for the peripheral arterial disease (PAD) marks an important chapter in a long-running story.
December 2018
Meta-analysis finds a higher risk of...
Following yesterday's news that the US Food and Drug Administration (FDA) has changed its stance on the use of paclitaxel-coated devices to treat peripheral arterial disease (PAD), Boston Scientific has announced its position on the update. The company's full...
Cagent Vascular has announced the results of its below-the-knee (BTK) RECOIL study. This core lab-adjudicated Recoil analysis— the first of its kind, according to the company— evaluated vessel recoil in lesions treated with Serranator versus plain balloon angioplasty.
The study...
In a letter to healthcare providers dated 11 July 2023, the US Food and Drug Administration (FDA) communicates that the risk of mortality associated with paclitaxel-coated devices to treat peripheral arterial disease (PAD) is no longer supported based on...
Shape Memory Medical has announced that it has entered into a sublicense agreement with "a global medtech market leader" in a press release. Under the agreement, Shape Memory Medical will sublicense its proprietary shape memory polymer technology for a...
Why is clot removal crucial for patients suffering from DVT? What are the major benefits of a lytic-free thrombectomy? Watch Michael Lichtenberg (Arnsberg, Germany) present the outcomes of the Arnsberg ClotTriever Study which confirms the safety and efficacy of...
Terumo Aortic have today announced that the Japanese Pharmaceuticals and Medical Devices Agency (PMDA) has granted approval of the Thoraflex Hybrid frozen elephant trunk (FET) device for commercial sale in Japan for the treatment of patients with complex...
As announced at the Society for Vascular Surgery (SVS) Vascular Annual Meeting (VAM 2023; 14–17 June, National Harbor, USA), the Advance Serenity hydrophilic percutaneous transluminal angioplasty (PTA) balloon catheter product line is now available with even more options. Interventionists...
The US Food and Drug Administration (FDA) has granted approval for Cook Medical to initiate an investigational device exemption (IDE) study on the Zenith fenestrated+ endovascular graft (ZFEN+).
The clinical study will assess the safety and effectiveness of the ZFEN+...
A new study from Keck Medicine at the University of Southern California (USC; Los Angeles, USA) has uncovered “significant racial disparities” in the diagnosis, treatment and outcomes of peripheral artery disease (PAD) among Black and white patients in the USA.
“We discovered that Black...
“It is time to question whether patients with end-stage below-the-knee peripheral arterial disease should not be treated primarily with deep vein arterialisation,” Arne Schwindt (Münster, Germany) avers in an interview with Vascular News.
Schwindt was speaking at this year’s Leipzig...
A temporary mechanical support with antiproliferative properties might combine two particular treatment qualities to fill a gap in the treatment armamentarium for peripheral arterial disease, both above and below the knee. Ramon Varcoe (Sydney, Australia) moderates a panel with...
Surmodics has announced the receipt of US Food and Drug Administration (FDA) approval for the SurVeil drug-coated balloon (DCB).
A company press release notes that the SurVeil DCB may now be marketed and sold in the USA to physicians for...
The local delivery of a glucose-derived compound in small- and medium-sized abdominal aortic aneurysms (AAAs) has been deemed safe, with “promising” early efficacy data indicating its potential in stabilising or slowing AAA sac growth. According to Stephen Cheng (University...
The findings of a recent study on reinterventions and sac dynamics after fenestrated endovascular aneurysm repair (FEVAR) with a physician-modified endograft (PMEG) for index aneurysm repair and following prior EVAR led researchers to conclude that “vigilant” surveillance and a...
Endospan has announced the initial experience with its custom-made Nexus Duo aortic arch stent graft system. Theodosios Bisdas (Athens Medical Centre, Athens, Greece) presented the multicentre European experience of the first ten patients treated with Nexus Duo at this...
The principal investigators behind the BEST-CLI trial struck a conciliatory tone during the inaugural Frank J Veith Distinguished Lecture at this year’s Society for Vascular Surgery (SVS) Vascular Annual Meeting (VAM 2023; 14–17 June, National Harbor, USA) in which...
“The endothelial cell is the superhero of the vascular system,” Kathryn L Howe (University of Toronto, Toronto, Canada) argued during an invited research presentation at this year’s Society for Vascular Surgery (SVS) Vascular Annual Meeting (VAM 2023; 14–17 June,...
Endologix has announced the 24-month results of the DETOUR2 study, presented at VAM 2023 (14–17 June, National Harbor, USA), the annual meeting of the Society for Vascular Surgery (SVS), by one of the study’s principal investigators, Sean Lyden (Cleveland...
Long-term overall survival (OS) and amputation-free survival (AFS) are outcomes that rebound in claudicants who quit smoking prior to elective surgery—and they mirror those of never smokers. But patients who do not kick the habit have significantly worse outcomes,...
NOTE: This video is ONLY available to watch in selected countries and geographies
The combined use of a drug-eluting device and a vascular scaffold “seems to be, in the longer run, the perfect solution for complex lesion treatment”. This...
Gore announced today the initiation of the Gore VBX FORWARD clinical study to compare the VBX stent graft to bare metal stenting for patients with complex iliac occlusive disease.
This prospective, multicentre, randomised controlled trial will include up to 40...
Surmodics announced in a press release that it has received US Food and Drug Administration (FDA) 510(k) clearance for its Pounce LP (low profile) thrombectomy system.
Introduced in 2021, the Pounce thrombectomy system is intended for the non-surgical removal...
Terumo Aortic announced in a press release the launch of the company’s global post-approval study (PAS) known as EXTEND for Thoraflex Hybrid, the only frozen elephant trunk (FET) device approved by the US Food and Drug Administration (FDA) for...
Eric Secemsky (Boston, USA) and Edward Choke (Singapore) discuss the role of sirolimus-coated balloons in the treatment of peripheral arterial disease (PAD), particularly below the knee, at the Charing Cross (CX) Symposium (25–27 April, London, UK).
With high amputation and...
Sharing first-time data from the SOCRATES (ShOrt neCK AAA RAndomized Trial) design, Giovanni Torsello (St Franziskus Hospital, Münster, Germany) expanded on the intentions of the prospective, global, multicentre, randomised trial which he stated is the “first comparative” study to...
Endologix has announced in a press release that the US Food and Drug Administration (FDA) has granted approval for the Detour system to treat patients with complex peripheral arterial disease (PAD).
Patients with long blockages of the superficial femoral artery...
The Connect The World Session: Japan at this year’s Leipzig Interventional Course (LINC 2023, 6–9 June, Leipzig, Germany)—in collaboration with the Japanese Endovascular Treatment Conference (JET)—sparked discussion surrounding the latest data in femoropopliteal interventions. Giving platform to multiple...
Delivering first-release data, Scott O Trerotola (University of Pennsylvania, Philadelphia, USA) conveyed the “consistent results” in a real-world environment for the Lutonix arteriovenous (AV) post-approval study (PAS) for drug-coated balloon (DCB)-AV on the afternoon of the second day at...
Bentley announced at this year's Leipzig Interventional Course (LINC 2023; 6–9 June, Leipzig, Germany) that it has reached an important milestone: having sold the 300,000th BeGraft. This milestone was celebrated at the Bentley booth at LINC.
After launching the first...
Gore has announced that the first US patient has been enrolled in a prospective, non-randomised, multicentre, single-arm study with five-year follow-up to evaluate the investigational Gore Viafort vascular stent for the treatment of symptomatic iliofemoral venous obstruction.
The first US...
Following his first-to-podium presentation on statin treatment after aortic repair at the 2023 Charing Cross (CX) International Symposium (25–27 April, London, UK), Kevin Mani (Uppsala, Sweden) joined Janet Powell (London, UK) at the CX Vascular Live studio to discuss...
“I see a future where this technology may be used to treat people early and avoid the devastating long term consequences of venous insufficiency” Ramon Varcoe (Sydney, Australia) opined in a CX Vascular Live discussion with Erin Murphy (Charlotte,...
The US Food and Drug Administration (FDA) has released draft guidance with updated recommendations for good clinical practices (GCPs) aimed at modernising the design and conduct of clinical trials.
In a statement, the regulator said that the updates are intended...
Konstantinos Spanos (Larissa, Greece) recently shared new meta-analysis data from comparative studies evaluating balloon-expandable versus self-expandable stent grafts for branched endovascular aneurysm repair (BEVAR). Following his first-to-podium presentation at the 2023 Charing 2023 Charing Cross (CX) International Symposium (25–27...
Presenting first-time data from large multicentre European studies at this year's Leipzig Interventional Course (LINC 2023; 6–9 June, Leipzig, Germany), Arne Schwindt (St Franziskus Hospital Münster, Münster, Germany) and Ralf Langhoff (Sankt Gertrauden Hospital, Berlin, Germany) delivered results that...
While the exact location of atherosclerotic plaque ruptures—a common cause of myocardial infarction (MI) and stroke—has previously been unknown, researchers at Lund University (Lund, Sweden) have now managed to successfully map this phenomenon. In addition, the team has identified...
Biotronik today announced the limited release of its Oscar (One Solution: Cross. Adjust. Restore) multifunctional peripheral catheter and start of promotional activities at this year's Leipzig Interventional Course (LINC 2023; 6–9 June, Leipzig, Germany).
As indicated per Instructions for...
Positive results from a clinical trial involving a novel therapeutic procedure known as sonolysis—deployed during a carotid endarterectomy (CEA)—were presented at the recent European Stroke Organisation Conference (ESOC; 24–26 May, Munich, Germany).
“The randomised clinical trial confirmed the results of...
Medtronic has announced that Ken Washington has been appointed chief technology and innovation officer.
Washington joins Medtronic from Amazon where he served as vice president and general manager of consumer robotics, and will lead technology development across industries including robotics,...
Penumbra has announced the US Food and Drug Administration (FDA) clearance and launch of Lightning Bolt 7, which the company claims is the most advanced and powerful arterial thrombectomy system on the market.
Lightning Bolt 7 introduces a new method...
NOTE: This video is ONLY available to watch in selected countries and geographies
Could drug-coated balloons (DCBs) be “the perfect device” for the treatment of patients with neointimal hyperplasia? Yes, opines Matteo Tozzi (University of Insubria, Varese, Italy) at...
This advertorial is sponsored by Shockwave Medical.
Mazin Foteh, MD, contrasts the benefits of Shockwave Medical’s new Shockwave L6 Peripheral Intravascular Lithotripsy Catheter alongside those of its sister device, the Shockwave M5+ Peripheral Intravascular Lithotripsy Catheter.
The Shockwave L6 device is...
A recent study has concluded that the “novel” technique of serration angioplasty with the Serranator balloon (Cagent Vascular) for the treatment of chronic limb-threatening ischaemia (CLTI) in the pedal arteries is “safe and effective”. Following his presentation of this...
This advertorial is sponsored by Inari Medical
“We need to eliminate symptoms as fast as possible—it is not OK just to make things a little better,” says Rick de Graaf (Clinical Centre of Friedrichshafen, Friedrichshafen, Germany), setting out why intervention...
Shape Memory Medical has announced the publication of results from the company’s prospective, open-label, single-arm, safety study of the Impede embolisation plug in peripheral vascular embolisation. The study was published this month in the peer-reviewed journal, Vascular, ahead of...
Twelve-month results from the SELUTION SFA trial have been presented for the first time at the Japan Endovascular Treatment (JET) Conference (26–28 May, Tokyo, Japan). The objective of this study was to assess the safety and efficacy of the...
Thirona recently announced its new artificial intelligence (AI)-based algorithm for pulmonary artery-vein phenotyping, LungQ AVX. The results from multiple validation studies were presented at the American Thoracic Society (ATS) 2023 International Conference (19-24 May, Washington, DC, USA).
A press release...
Cora Therapeutics has announced the presentation of the results of a clinical trial at the Canadian Association of Interventional Radiologists (CAIR) annual meeting (25–27 May, Quebec City, Canada), assessing the efficacy of its proprietary antioxidant complex in safeguarding DNA...
On 24 May, the US Food and Drug Administration (FDA) granted an investigational device exemption (IDE) approval for Concept Medical's MagicTouch percutaneous transluminal angioplasty (PTA) sirolimus-coated balloon (SCB) for treatment of the superficial femoral artery (SFA).
This marks the fourth...
Interim results from the ECST-2 randomised controlled trial (RCT) have shown no evidence that carotid stenosis patients with a low-to-intermediate stroke risk, treated with optimised medical therapy, will benefit from additional carotid revascularisation via carotid endarterectomy (CEA) or carotid...
Speaking on the FLAME, FLASH and PEERLESS trials that each collected data on the FlowTriever (Inari Medical) device for the treatment of pulmonary embolism (PE), Ripal Gandhi (Miami Cardiac & Vascular Institute & Miami Cancer Institute, Miami, USA) elaborated...
A new study using data from millions of patients hospitalised across the USA has determined whether population density and associated urban versus suburban versus rural environments predispose patients with peripheral vascular disease for chronic poor blood flow in their...
This advertorial is sponsored by Cardionovum®.
In 2023, there are a number of different options available for treating the vascular access complications of thrombosis and stenosis, Matteo Tozzi (University of Insubria, Varese, Italy) tells Vascular News. He highlights that perhaps...
Data from a cohort of one million patients with chronic limb-threatening ischaemia (CLTI) were recently presented as late-breaking clinical research at the Society for Cardiovascular Angiography & Interventions (SCAI) 2023 Scientific Sessions (18–20 May, Phoenix, USA). Using data from Medicare,...
Presenting “remarkable results” from the JETi registry—a prospective, multicenter, observational study which collected real-world data on the safety, performance and clinical benefits of the JETi peripheral thrombectomy system—speaker Mahmood K. Razavi (Children’s Health of Orange County, Orange, USA) relayed...
Steven Abramowitz (Washington DC, USA) explains the significance of the CLOUT registry—the only registry capturing data on mechanical thrombectomy in deep venous thrombosis (DVT). CLOUT confirms excellent safety results and effectiveness of the ClotTriever system in real-world DVT patients....
Artificial intelligence (AI) is “here to stay”. This was the closing statement from Randy Moore (Calgary, Canada), vascular surgeon and chief medical officer (CMO) of ViTAA Medical Solutions, during a recent CX Vascular Live roundtable discussion. Moore was joined...
Endologix recently announced the completion of the 150th case in the JAGUAR study.
This randomised controlled trial is evaluating Endologix’s Alto abdominal stent graft system, comparing its effectiveness to other commercially available endovascular aneurysm repair (EVAR) devices in the...
Six-month outcomes from FLASH registry have shown that patients with pulmonary embolism who were treated with mechanical thrombectomy showed significant improvement in symptoms, quality of life and cardiac functions.
The findings were presented as late-breaking clinical research at the Society...
This advertorial, sponsored by Inari Medical, is only available in selected countries and geographies.
During a recent webinar hosted by Inari Medical, a multidisciplinary group of physicians focused in on how to select eligible patients for the endovascular treatment of...
A new analysis of chronic limb-threatening ischaemia (CLTI) treatment outcomes was presented today as late-breaking clinical research at the Society for Cardiovascular Angiography & Interventions (SCAI) 2023 Scientific Sessions (18–20 May, Phoenix, USA), a press release reports.
Following the initial...
Both open and endovascular surgical approaches are likely to produce improved outcomes compared to ligation in the treatment of ruptured extracranial carotid artery aneurysms. This was the salient point delivered by Maarit Venermo (Helsinki University Hospital, Helsinki, Finland) at the...
Bentley has announced the US launch of its BeBack crossing catheter, which is designed for the treatment of heavily calcified lesions. This is the company's first product to be launched in the USA.
A press release notes that, after the...
In this issue:
Extensive coverage of CX 2023, including a full report of the BASIL-2 randomised controlled trial results
New guidelines: Chris Twine (Bristol, UK) and Stavros Kakkos (Patras, Greece) highlight key takeaways from soon-to-be-published recommendations on antithrombotic therapy for...
In this issue:
Extensive coverage of CX 2023, including a full report of the BASIL-2 randomised controlled trial results
New guidelines: Chris Twine (Bristol, UK) and Stavros Kakkos (Patras, Greece) highlight key takeaways from soon-to-be-published recommendations on antithrombotic therapy...
A recent proposal has called for “whole-person, multidisciplinary interventions” after an interrogation of the interplay between lower extremity peripheral arterial disease (PAD) and mental health impacts. Published in the Journal of The American College of Cardiology, researchers have put...
For the treatment of juxtarenal aortic aneurysms, reinterventions after physician-modified endovascular grafts (PMEGs) are non-detrimental to long-term survival. This is one of the main findings of a recently published study by Ayumi Tachida (University of Washington School of Medicine,...
With proper patient selection, short-term outcomes of thoracic endovascular aortic repair (TEVAR) in acute uncomplicated acute type B aortic dissection (uTBAD) are similar irrespective of the timing of treatment after dissection. This is according to a study recently published...
A nationwide cohort study of over 9,000 patients shows that the “controversial” practice of arteriotomy closure for carotid endarterectomy (CEA) is associated with an increased risk of ipsilateral stroke and “should be avoided”. Instead, authors Magnus Jonsson (Karolinska University...
The 2023 Charing Cross (CX) International Symposium (25–27 April, London, UK) saw a first-to-podium presentation by Andrew Holden (Auckland City Hospital, Auckland, New Zealand) in which he presented new data from the IN.PACT AV Access trial of the IN.PACT...
April's top 10 sees a product announcement from Terumo Aortic take the top spot, with highlights from the 2023 Charing Cross (CX) International Symposium (25–27 April, London, UK) also proving popular among readers.
1. Terumo Aortic announces first implant of...
Judges of the CX 2023 Dragon’s Den-style contest — the finale of the Day 3 CX Innovation Showcase programme — described the field of entrants to this year’s edition of the competition as the strongest line-up in its history.
The...
Representatives from multiple specialties highlight a pressing need to drive up the quality of research and care for patients who require the “life-changing” intervention that is an amputation of the leg.
The UK government’s National Institute for Health and Care...
Terumo Aortic today announced that the US Food and Drug Administration (FDA) has granted approval of the RelayPro thoracic stent graft device for the treatment of dissection and transection in the USA.
A Terumo Aortic press release notes that RelayPro...
Xeltis has presented “highly-encouraging” six-month data from its first-in-human (FIH) aXess vascular graft trial at the 13th Congress of the Vascular Access Society (VAS; 27–29 April, Porto, Portugal).
A press release outlines that six-month data from the FIH trial found...
The first US patient has been enrolled in the SELUTION4SFA sirolimus drug-eluting balloon (DEB) study by Arthur Lee (Cardiac & Vascular Institute, Gainesville, USA).
This study evaluates SELUTION SLR in the treatment of occlusive disease of the superficial femoral...
“The time has definitely come to look at the evidence, and redo these studies,” posited Alun Davies (Imperial College London, London, UK), putting forward his argument that the NASCET and ECST clinical trials “need to be reconducted” at this...
Vivasure Medical has announced it has enrolled the first patients in the company’s PerQseal PATCH clinical study, a multicentre, single-arm, pivotal study evaluating the safety and efficacy of the Vivasure PerQseal closure device system. The first patients were enrolled...
Cook Medical recently announced that the first patient has been treated in a clinical study to evaluate a new venous valve designed for treating chronic venous insufficiency (CVI). The patient was treated by principal investigator Mauricio Alviar (Clinica de...
Endologix has announced the online publication of the final five-year results of the LEOPARD trial in the Journal of Vascular Surgery (JVS). The study's findings showed that there was no significant difference in aneurysm-related outcomes between patients randomised to...
This advertorial is sponsored by Boston Scientific.
During a recent satellite symposium, which took place at the 2022 Cardiovascular Interventional Radiological Society of Europe (CIRSE) annual meeting (10–14 September, Barcelona, Spain), experts emphasised the clinical value of drug-eluting technologies (DET)...
A question from Manj Gohel (Cambridge University Hospitals NHS Foundation Trust, Cambridge, UK) on what the Charing Cross (CX) audience should take back to their multidisciplinary team meetings from the first-time presentation of BASIL-2 led chief investigator Andrew Bradbury...
A debate between two prominent carotid interventionists—Peter Schneider (University of California San Francisco, San Francisco, USA) and Domenico Valenti (King's College London, London, UK)—at the 2023 Charing Cross (CX) International Symposium (25–27 April, London, UK) revealed that the vascular...
Penumbra recently announced the launch of the latest addition to its RED family of catheter devices for mechanical thrombectomy, the RED 43 reperfusion catheter.
According to the company, this device is intended to take distal aspiration “to the next level”...
A first-to-podium presentation at the 2023 Charing Cross (CX) International Symposium (25–27 April, London, UK) saw Hence Verhagen (Erasmus Medical Center, Rotterdam, The Netherlands) present unique 10-year data from the ENGAGE OUS registry on the Endurant abdominal aortic aneurysm...
This advertorial is sponsored by Gore.
At this year’s Charing Cross (CX) International Symposium (25–27 April, London, UK), Andrew Holden (Auckland City Hospital, Auckland, New Zealand), presented results from the longest follow-up study to date evaluating the GORE® VIABAHN® VBX...
This advertorial is sponsored by Gore.
Even though it is celebrating its aortic portfolio reaching its 25-year milestone, Gore believes that this is only the beginning when it comes to being an aortic ally. The GORE® EXCLUDER® AAA Endoprosthesis and...
This advertorial is sponsored by Gore.
During a satellite symposia at the 2023 Charing Cross (CX) International Symposium (25–27 April, London, UK), speakers addressed the “key factor” of conformability in the endovascular repair of multiple vascular pathologies.
First to address the...
Statin treatment after aortic repair is associated with improved long-term survival, while dose does not matter. This was the key message from a first-to-podium presentation delivered by Kevin Mani (Uppsala University, Uppsala, Sweden) at the 2023 Charing Cross (CX)...
A question from Manj Gohel (Cambridge University Hospitals NHS Foundation Trust, Cambridge, UK) on what the Charing Cross (CX) audience should take back to their multidisciplinary team meetings from the first-time presentation of BASIL-2 led chief investigator Andrew Bradbury...
Bentley has bought its main supplier, the Switzerland-based Qmedics, Bentley has recently announced in a press release. It also announced its plan of initial public offering (IPO), which is planned for autumn/winter 2023/2024, depending on market conditions.
Qmedics manufactures approximately...
Terumo Aortic today announced the first North American implant of a custom-made hybrid device, Thoracoflo.
A press release notes that the device is used to treat patients with thoracoabdominal aortic disease using a less invasive surgical technique than traditional open...
Surmodics has announced enrolment of the first patient in PROWL, the Pounce thrombectomy system retrospective registry. PROWL is an open-label, retrospective, multicentre US registry of the Pounce system for the non-surgical removal of emboli and thrombi in the peripheral...
Surmodics has announced that Ankur Lodha (Cardiovascular Institute of the South, Lafayette, USA) and Pradeep Nair (Cardiovascular Institute of the South, Houma, USA) were the first physicians to use the company’s Sublime radial access microcatheter.
The Sublime microcatheter is in...
Lithotripsy may hold the key to enabling more carotid artery disease patients who require calcification treatment to undergo stent placement via a transcarotid artery revascularisation (TCAR) procedure, as per single-centre experiences presented at the recent Society for Clinical Vascular...
In this issue:
What to expect from CX 2023
A year in profiles: Alexander Zimmermann, Joseph Coselli, Markus Steinbauer and Jon Boyle
Medical devices: Key updates on regulatory changes in Europe
EVC 2023: STEVECO trial calls into question randomisation in...
In this issue:
What to expect from CX 2023
A year in profiles: Alexander Zimmermann, Joseph Coselli, Markus Steinbauer and Jon Boyle
Medical devices: Key updates on regulatory changes in Europe
EVC 2023: STEVECO trial calls into question randomisation in...
An emerging programme at Houston Methodist aims to help prod those practicing in the vascular surgical space deeper into the field of robotic surgery.
The first ah-ha during a talk at a national meeting being given by one of the...
The Society for Vascular Surgery Vascular Quality Initiative (SVS VQI) clinical registry recently announced it has achieved another milestone, with more than 1,000 centres enlisted in VQI registries. This achievement will strengthen vascular care, improve outcomes and underscore the...
The US launch of a new peripheral intravascular lithotripsy (IVL) catheter, new data on large bore mechanical thrombectomy in high-risk pulmonary embolism (PE) patients, and changes to the EU Medical Devices Regulation (MDR) caught readers’ attention in March.
1. Shockwave...
AVS, an early-stage medical device company focused on safely and effectively treating severely calcified arterial disease with its pulsatile intravascular lithotripsy (PIVL) therapy, has announced that it closed an additional US$8.8 million in new financing, bringing its Series B...
A new investigation into the association between end-stage renal disease (ESRD) and major adverse limb events (MALEs) has found that not only is it strong, but ESRD is particularly associated with below-knee amputation. The details of the study were...
This advertorial is sponsored by Philips.
Two early adopters of Fiber Optic RealShape (FORS; Philips) share their expert opinions on the developing technology, highlighting its future potential in reducing radiation and increasing procedural efficiency.
The European Society for Vascular Surgery (ESVS)...
This educational supplement, sponsored by Artivion, is only available in selected countries and geographies.
The E-liac stent graft system (Artivion/Jotec), according to the clinical data and expert experience, has the potential to overcome the pressing challenge of preserving the...
The CX 2023 Venous & Lymphatic programme is set to be a “highlight” of this year’s meeting, CX co-chair and executive board member Erin Murphy (Sanger Heart and Vascular Institute, Atrium Health, Charlotte, USA) tells Vascular News.
This year’s programme...
“When you save a limb, you save a life” Marianne Brodmann (Graz, Austria) tells CX Live. There are many people who never get seen by a vascular specialist as often there is not a great deal of knowledge among...
Results from the PROMISE II pivotal trial investigating transcatheter arterialisation of the deep veins using the LimFlow system in so-called no-option chronic limb-threatening ischaemia (CLTI) patients were today published in the New England Journal of Medicine (NEJM).
The publication confirmed...
Dual antiplatelet therapy (DAPT) has demonstrated improved clinical outcomes, including reduced in-hospital stroke and mortality risks, compared to other medication regimens in carotid artery disease patients who undergo stent placement via a transcarotid artery revascularisation (TCAR) procedure.
This is the...
Joseph Coselli (Houston, USA) reflects on the return of in-person meetings and the value they offer in terms of exchanging new information, learning from other healthcare professionals, expanding collective knowledge of the field and ultimately “improving what physicians can...
Tilo Kölbel (Hamburg, Germany) speaks to CX Live about new, innovative developments within devices and recent results of large studies producing data that poses “interesting” updates to the field of vascular surgery.
Focusing in on the aortic community and areas...
Navigating cases of abdominal aortic aneurysm (AAA) with short neck anatomy pose a serious challenge to vascular surgeons, but the list of potential options available to carry out repairs are numerous—and decision-making can be optimised by using a bespoke...
W L Gore & Associates (Gore) has announced that the first US patient has been enrolled in a prospective, non-randomised, multicentre, single-arm study with five-year follow-up to evaluate the investigational Gore Viafort vascular stent for the treatment of symptomatic...
Data and discussion on revascularisation treatment strategies for patients with chronic limb-threatening ischaemia (CLTI) will take centre stage at the CX 2023 Consensus update, with results from the BASIL-2 (Bypass versus angioplasty in severe ischaemia of the leg-2) randomised...
Palma Shaw (Syracuse, USA) discusses how the World Federation of Vascular Societies (WFVS) collaborates to determine what the key needs for trainees and education across the world are. “We bring global vascular societies together and engage in projects in...
Barbara Rantner (Munich, Germany) talks about the “fantastic experience” of being back in person at CX 2022, where she notes that attendees were able to “meet experts from all over the world” and discuss relevant issues “not only from...
Getinge's iCast covered stent system has received premarket approval from the US Food and Drug Administration (FDA) for the treatment of patients with iliac arterial occlusive disease.
Iliac arterial occlusive disease is a type of peripheral arterial disease (PAD), which...
A recent study comparing outcomes of endovascular aneurysm repair (EVAR) patients has reported no statistically significant differences in mortality or secondary rupture rates between standard Cook, Medtronic and Gore endografts, suggesting similar safety in a real-world setting.
Michael O Falster...
The Society for Vascular Surgery (SVS) and the American College of Surgeons (ACS) have launched the “Vascular Verification Program (Vascular-VP)," an ACS Quality Program developed in partnership with the SVS.
The newly launched inpatient programme reviews not just safety processes...
A range of experts, including Koen Deloose (Dendermonde, Belgium), Thomas Zeller (Bad Krozingen, Germany), Marianne Brodmann (Graz, Austria) and Antonio Micari (Messina, Italy), talk to Vascular News about how the latest data supporting the cost-effectiveness of the IN.PACT Admiral drug-coated balloon...
Viz.ai recently announced it has received US Food and Drug Administration (FDA) 510(k) clearance for its algorithm intended to detect suspected abdominal aortic aneurysm (AAA). Viz AAA is the first FDA-cleared AI-powered solution for the detection and triage of...
Koen Deloose (Dendermonde, Belgium) talks to Vascular News about the key one-year data from the BIOPACT randomised controlled non-inferiority trial, which evaluated the safety and efficacy of the Passeo-18 Lux drug-coated balloon (DCB) catheter (Biotronik) compared to the IN.PACT Admiral...
A total of £10 million has been awarded to the Medicines and Healthcare products Regulatory Agency (MHRA)—an executive agency of the UK Department of Health and Social Care (DHSC)—to help bring innovative new medicines and medical technologies to UK...
Merit Medical has announced the expansion of itsSwiftNinja steerable microcatheter product line. New sizes include a low-profile 2.4Fr distal diameter option in 125-cm and new longer 150-cm lengths.
The 180-degree articulating microcatheter is designed to provide access to challenging peripheral...
Vesteck scientific advisory board members along with key opinion leaders from around the world have successfully completed the first three in-human cases with the Suture-Tight catheter, a press release reports.
Dai Yamanouchi (University of Wisconsin, Madison, USA) and Dainis Krievins...
Shockwave Medical today announced the full US commercial availability of the Shockwave L6 peripheral intravascular lithotripsy (IVL) catheter following clearance by the US Food and Drug Administration (FDA).
A press release details that the Shockwave L6 catheter is purpose-built to...
Vivasure Medical has announced that the US Food and Drug Administration (FDA) has granted an investigational device exemption (IDE) to advance the company’s PATCH clinical study, a multicentre, single-arm, pivotal study evaluating the safety and effectiveness of the Vivasure...
Armando Mansilha (Porto, Portugal) tells CX Live about the recent publication of the first European training requirements in vascular surgery. Published in International Angiology, the guidance was approved by the (UEMS) comprising 40 national associations and 43 specialist sections...
Recruitment is now complete with more than 100 patients as planned per protocol in the branched endovascular aneurysm repair (BEVAR) study that uses Bentley's BeGraft peripheral Plus balloon expandable covered stent as a bridging stent in complex aortic aneurysms repairs.
Sebastian Büchert...
Distal embolic protection using a filter has been associated with improved transfemoral carotid artery stenting (tfCAS) outcomes in terms of in-hospital stroke and death risks—underpinning current Society for Vascular Surgery (SVS) guidelines recommending routine use of distal embolic protection...
A new study from the University of Sheffield, published in the British Journal of Surgery (BJS), has found clear evidence of socioeconomic disparities in operation rates and survival after surgery for abdominal aortic aneurysm (AAA).
The study, funded by the...
When receiving treatment for abdominal aortic aneurysm (AAA), female patients have a higher risk for kidney damage after endovascular repair, a Michigan Medicine study finds.
Researchers from Michigan Medicine designed an algorithm that can be used prior to endovascular AAA...
Michel Reijnen (Arnhem, The Netherlands) presents the results of a study that sought to assess the potential cost-effectiveness of a urea-based drug-coated balloon (DCB; IN.PACT Admiral, Medtronic) as standard of care treatment for those with chronic limb threatening ischaemia...
Speaking to CX Live, Eric Secemsky (Boston, USA) delves into the emerging use of intravascular ultrasound (IVUS) in the peripheral procedural phases—outlining contemporary studies providing evidence of its benefit, such as the reduction of restenosis risk when IVUS is...
The European Union’s Council of Ministers has today adopted a resolution to extend the deadline for the certification of medical devices under the Medical Devices Regulation (MDR).
Producers of medical devices will have until 31 December 2027 for higher risk...
Data from a new prespecified analysis of the phase III VOYAGER PAD clinical trial show that low-dose rivaroxaban plus aspirin resulted in a 33% reduction in acute limb ischaemia and a 15% reduction in major adverse limb and cardiovascular...
Large bore mechanical thrombectomy with the FlowTriever system (Inari Medical) in patients with high-risk pulmonary embolism (PE) was associated with a significantly lower occurrence of meaningful in-hospital adverse clinical outcomes compared to other contemporary treatments, data presented at the...
Robert A Lookstein, who is executive vice chair, Diagnostic, Molecular and Interventional Radiology at the Icahn School of Medicine at Mount Sinai Hospital (New York, USA) today presented the results of a subanalysis of Thrombolex’s National Heart, Lung and...
In a first-in-human, compassionate-use case approved by the US Food and Drug Administration (FDA), University of Michigan Health (Ann Arbor, USA) interventional radiologists David M Williams and Minhaj S Khaja successfully used VeinWay's Traversa for venous recanalisation to save...
February's top 10 includes the announcement that Abbott is to acquire Cardiovascular Systems, results of the PRESERVE study on the safety and effectiveness of inferior vena cava filters in treating venous thromboembolism, and an interview with past president of...
ReCor Medical and its parent company, Otsuka Medical Devices, recently announced that primary endpoint results from the RADIANCE II pivotal trial were published in the Journal of the American Medical Association (JAMA). Study results showed that the Paradise ultrasound...
As the new kid on the block, endovascular treatment of the common femoral artery (CFA) just does not have that long-term outcome data yet,” Narayanan Thulasidasan (London, UK) tells Vascular News. Thulasidasan discusses some of the benefits of endovascular therapy...
Giacomo Isernia (Perugia, Italy) sits down with Vascular News to discuss the challenges posed by calcium when it comes to endograft delivery. Calcium “limits the ability to choose the proper graft for each patient,” states Isernia. “This is a pity as...
Treatment of common femoral artery (CFA) stenosis, either with an open or endovascular approach, is the subject of much debate, Raphaël Coscas (Paris, France) tells Vascular News at the Paris Vascular Insights (PVI) conference (23–25 November 2022, Paris, France). “Open surgery...
Bella Huasen (Preston, UK) talks to Vascular News about some of the difficulties of treating occlusive disease in the femoropopliteal segment and why it is “essential to have tools that will allow you to deal with these challenges”.
Huasen outlines some of...
This article forms part of an educational supplement sponsored by Shockwave Medical. Explore the full Compliance is Key series here.
Raphaël Coscas (Ambroise Paré Hospital, AP-HP and Paris-Saclay University, Paris, France) illustrates the use and benefits of Shockwave Intravascular Lithotripsy (IVL;...
This article forms part of an educational supplement sponsored by Shockwave Medical. Explore the full Compliance is Key series here.
Giacomo Isernia and Gioele Simonte (Azienda Ospedaliera di Perugia, Perugia, Italy) outline a branched endovascular aneurysm repair (BEVAR) case in...
This article forms part of an educational supplement sponsored by Shockwave Medical. Explore the full Compliance is Key series here.
In this case report, Narayanan Thulasidasan (Guy’s & St Thomas’ NHS Foundation Trust, London, UK) demonstrates how the M5+ Intravascular...
This article forms part of an educational supplement sponsored by Shockwave Medical. Explore the full Compliance is Key series here.
Bella Huasen (Lancashire Teaching Hospitals NHS Foundation Trust, Preston, UK) outlines a recent femoropopliteal occlusion case, during which Shockwave Intravascular...
This article forms part of an educational supplement sponsored by Shockwave Medical. Explore the full Compliance is Key series here.
Stefano Fazzini (Tor Vergata Hospital, Rome, Italy) goes into the details and the data behind Shockwave Intravascular Lithotripsy (IVL; Shockwave...
This educational supplement is sponsored by Shockwave Medical. Explore the full Compliance is Key series here.
In this supplement:
Stefano Fazzini (Tor Vergata Hospital, Rome, Italy) goes into the details and the data behind Shockwave Intravascular Lithotripsy (IVL; Shockwave Medical)—a...
Few adverse events are connected to the use of inferior vena cava (IVC) filters to help prevent deep vein blood clots from developing into pulmonary embolisms (PEs), according to the findings of the Predicting the safety and effectiveness of...
David Gillespie (Boston, USA) talks to Vascular News about the PRESERVE study—established by the Society for Vascular Surgery and Society of Interventional Radiology—which is the largest prospective study to investigate real-world outcomes with contemporary use of inferior vena cava...
Humacyte, a clinical-stage biotechnology platform company developing universally implantable bioengineered human tissues and advanced tissue constructs and organ systems at commercial scale, today announced the publication of “Six-year outcomes of a phase two study of human-tissue engineered blood vessels...
NOTE: This video is ONLY available to watch in selected countries and geographies
Robert Shahverdyan (Hamburg, Germany), Matteo Tozzi (Varese, Italy), Alexandros Mallios (Paris, France), Ounali Jaffer (London, UK) and Narayan Karunanithy (London, UK) each give their take on...
BioGenCell has announced that it is recruiting for an international clinical trial to assess the company's stem cell-based therapy for chronic limb-threatening ischaemia patients who are at risk for limb amputation.
The clinical trial is currently taking place in medical...
Almost one-half of the patients receiving a peripheral vascular intervention (PVI) in the Society for Vascular Surgery (SVS) Vascular Quality Initiative (VQI) registry between 2017 and 2018 were not on guideline-directed medical therapy (GDMT). Kim G Smolderen and Carlos...
Shape Memory Medical has announced the first patient treated in Germany as part of the EMBO postmarket surveillance registry (EMBO-PMS), the company’s prospective, multicentre registry study of its Impede and Impede-FX embolisation plugs when used for peripheral vascular embolisation.
The...
The publication of a consensus statement from the European Society of Cardiology (ESC) Council on Hypertension and the European Association of Percutaneous Cardiovascular Interventions (EAPCI) is among the final steps in the evaluation of renal denervation as a device-based...
Xeltis has raised €32 million in a Series D2 equity fundraise, backed by a syndicate of current and new investors, which the company says will enable it to progress its clinical programmes into pivotal trials.
Investors include Grand Pharma, DaVita...
Following his 2021–2022 presidency of the Vascular Society of Great Britain and Ireland (VSGBI), Jon Boyle (Cambridge University Hospitals NHS Trust, Cambridge, UK) speaks to Vascular News about his career so far. He also shares his thoughts on the...
W L Gore & Associates today announced that it is initiating the EMBRACE registry to capture real-world data about the Gore Viabahn VBX balloon expandable endoprosthesis used as a bridging stent in conjunction with a branched/fenestrated stent graft.
This multicentre,...
Renal denervation represents another treatment option in patients with uncontrolled resistant hypertension and may be used in selected patients deemed intolerant to antihypertensive drugs.
These are among the messages of a new consensus statement published in EuroIntervention following a review...
NOTE: This video is ONLY available to watch in selected countries and geographies
Iliac side branch devices (ISBs) are “gamechangers in infrarenal therapy of abdominal aortic aneurysms (AAAs),” Mario Lescan (Tübingen, Germany) tells Vascular News. Lescan states that ISBs “allow...
Biotronik has announced the US Food and Drug Administration (FDA) 510(k) clearance and CE mark of its Oscar (One Solution: Cross. Adjust. Restore) multifunctional peripheral catheter.
The company notes in a press release that Oscar is intended for percutaneous...
Prominent venous disease experts discuss venous stenting, appropriate care, and the pursuit of refined data and better education in a space where part of the problem involves practitioners moving “freely from being able to do arterial intervention and suddenly...
In this issue:
CX 2023 preview: Headline peripheral session to spotlight audience view on new BASIL-2 findings
Journal highlight: Individual patient data meta-analysis probes safety of carotid endarterectomy in the elderly
In profile: Jon Boyle (Cambridge, UK)
Andrej Schmidt (Leipzig, Germany)...
In this issue:
CX 2023 preview: Headline peripheral session to spotlight audience view on new BASIL-2 findings
Journal highlight: Individual patient data meta-analysis probes safety of carotid endarterectomy in the elderly
In profile: Jon Boyle (Cambridge, UK)
Andrej Schmidt...
In a recent study of patients with intermittent claudication (IC) caused by isolated superficial femoral artery (SFA) lesions, researchers found that primary stenting conferred benefits in health-related quality of life (HRQoL) at 36 months from treatment compared with best...
Data from the UK-COMPASS trial provide new insights into the management of complex aneurysm treatment in England and, according to lead investigator Srinivasa Rao Vallabhaneni (University of Liverpool, Liverpool, UK), underscore the need for appropriate patient and technique selection...
Cydar Medical recently announced it has successfully completed a US$11.5 million (£9.3 million) Series A funding round including a US$3.7 million (£3 million) cornerstone investment by Pembroke Venture Capital Trust (VCT). The company also revealed that Ken Hitchner has...
Demonstrating the “susceptibility” of care delivery to regional market competition, M Libby Weaver (University of Virginia, Charlottesville, USA) presented the case that intermittent claudication (IC) is a “novel and underdefined” driver of practice variation in management of patients at...
The Magic Touch percutaneous transluminal angioplasty (PTA) sirolimus drug-coated balloon catheter (DCB) has received investigational device exemption (IDE) for the treatment of below-the-knee (BTK) atherosclerotic lesions in peripheral arterial disease (PAD). Concept Medical received its first IDE approval for the...
Speaking at the 2023 Critical Issues America (CIA) annual meeting (Coral Gables, USA; 10–11 February), vascular surgeon Caitlin Hicks (Baltimore, USA) notes recent findings concerning tibial interventions among patients with claudication.
January's top 10 highlights an individual patient data meta-analysis on the safety of carotid endarterectomy in the elderly and a new randomised trial on the best treatment option for severe occlusive aortoiliac disease among other key updates in the...
Abbott and Cardiovascular Systems (CSI) have announced a definitive agreement for Abbott to acquire CSI. Under terms of the agreement, CSI stockholders will receive US$20 per common share at a total expected equity value of approximately US$890 million.
CSI is...
NOTE: This video is ONLY available to watch in selected countries and geographies
In this Vascular News roundtable, Andrew Holden (moderator; Auckland, New Zealand) is joined by Alexandros Mallios (Paris, France), Kate Steiner (Stevenage, UK) and Tobias Steinke (Düsseldorf, Germany) who...
At the 2022 Vascular Society of Great Britain and Ireland (VSGBI) annual scientific meeting (23–25 November, Brighton, UK), Srinivasa Rao Vallabhaneni (University of Liverpool, Liverpool, UK) delivered a first-time presentation of the UK-COMPASS study results. In this interview with Vascular News,...
Northeast Scientific announced this week it has received US Food and Drug Administration (FDA) 510(k) clearance for reprocessing the intravascular ultrasound (IVUS) Eagle Eye Platinum RX digital catheter (Philips).
CEO and founder, Craig Allmendinger shared his thoughts on the impact...
The UK National Vascular Registry (NVR) recently published its 2022 annual report—the 10th since the registry was launched in 2013—highlighting important outcome data for vascular procedures performed in the period between 2019 and 2021.
The report, which was prepared by...
The external validity—in other words, the transferability to real-world clinical practice—of contemporary randomised controlled trials (RCTs) investigating carotid artery stenosis interventions “varies considerably”, as per a recent analysis published in the European Journal of Vascular and Endovascular Surgery (EJVES)...
This advertorial is sponsored by Bentley.
Crossing chronic total occlusion (CTO) lesions are challenging procedures. The BeBack crossing catheter—Bentley’s first product to be available in both Europe and the USA following the company’s acquisition of Upstream Peripheral Medical Technologies’ GoBack...
A systematic review and meta-analysis have demonstrated “convincing evidence” that sex differences exist in carotid atherosclerosis, with all types of plaque features—including those relating to size, composition, and morphology—found to be either larger or more common in men than...
Cagent Vascular has announced the results of a comparative subanalysis of the PRELUDE-below-the-knee (BTK) study versus plain balloon angioplasty.
The study was led by Marianne Brodmann (Medical University of Graz, Graz, Austria). This subanalysis compared their PRELUDE-BTK subset to a...
Mid-term results from a phase II study of surgical bypass using the Human Acellular Vessel (HAV; Humacyte) demonstrated an overall secondary patency rate at 72 months of 60% measured by Kaplan Meier. There was no evidence of graft rejection...
Surmodics recently announced it has received a letter from the US Food and Drug Administration (FDA) related to its premarket approval (PMA) application for the SurVeil drug-coated balloon (DCB).
In the letter, the FDA indicated that the application is not...
A newly published meta-analysis of individual patient data has found that older patients with symptomatic carotid disease are likely to benefit as much from timely intervention as younger patients. Speaking to Vascular News in light of this key finding,...
The UK’s national abdominal aortic aneurysm (AAA) screening programme is fast approaching its 10th anniversary—a milestone that Akhtar Nasim (Sheffield Vascular Institute, Sheffield, Sheffield, UK) and Meryl Davis (Royal Free London NHS Foundation Trust, London, UK) believe provides an...
In a newly released “focused update” to their 2019 recommendations, the European Society for Vascular Surgery (ESVS) abdominal aortic aneurysm (AAA) guidelines writing committee has published advice on the surveillance and management of patients treated with the Nellix endovascular...
Viz.ai has announced the launch of Viz Vascular Suite—artificial intelligence (AI)-powered software enabling vascular care teams to automatically detect and triage care for suspected pulmonary embolism, right heart strain, aortic dissection, and abdominal aortic aneurysm (AAA). The company submitted...
Athanasios Saratzis (University of Leicester, Leicester, UK) speaks to Vascular News about the EVOCC trial, which he hopes will dispel patient and physician “uncertainty” around the best treatment option for severe occlusive aortoiliac disease. This randomised study recently received...
Cardiovascular Systems Inc (CSI) recently announced initiation of the KAIZEN clinical study of its Diamondback 360 peripheral orbital atherectomy system (OAS) for the treatment of calcified plaque in patients with peripheral arterial disease (PAD). The study is intended to...
Medtronic has announced the first patient enrolment in the ADVANCE trial, a head-to-head randomised controlled trial of two leading aortic stent graft systems, the Medtronic Endurant II/IIs stent graft system and Gore Excluder AAA device family systems. The ADVANCE...
Closer ties between cardiologists, cardiac surgeons and vascular surgeons will be a hallmark of the future treatment of diseases of the aorta, a leading figure behind new guidelines for the diagnosis and management of aortic disease tells Vascular News.
Jointly...
Today, Penumbra announced the US Food and Drug Administration (FDA) clearance and launch of its Lightning Flash mechanical thrombectomy system.
"Lightning Flash features Penumbra's novel Lightning intelligent aspiration technology, now with dual clot detection algorithms," the company notes in a...
Lydus Medical is pleased to announce that the Vesseal has received US Food and Drug Administration (FDA) 510(k) clearance. The Vesseal is a microvascular anastomosis suture deployment system, for standardised omnivessel anastomoses, enabling simple, fast, safe, and effective procedures.
An...
AVS has announced that it raised US$20 million in Series B financing, which the company says will accelerate clinical trial timelines for its device for peripheral application in pulsatile intravascular lithotripsy (PIVL) cases and advance development and preclinical work...
Cardiovascular Systems (CSI) announced that Innova Vascular (Innova) has submitted a 510(k) premarket notification to the US Food and Drug Administration (FDA) for its thrombectomy devices intended to treat peripheral vascular disease.
CSI intends to acquire and commercialise each of...
Aisha Shaikh (Memorial Sloan Kettering Cancer Centre, New York, USA) has staked out a claim that, though hypotension was “associated” with arteriovenous access (AV) thrombosis, it should be far from the only consideration in choosing an access modality. She...
ReCor Medical and its parent company Otsuka Medical Devices have announced the appointment of Lara Barghout as president and chief executive officer of ReCor.
Barghout will lead the ReCor business strategy and organisation in the global commercialisation of ReCor’s Paradise...
Despite his initial intention to become a transplant surgeon, Markus Steinbauer (Krankenhaus Barmherzige Brüder, Regensburg, Germany) recalls how the rise of endovascular techniques, and the possibilities for procedural advancement they posed in vascular surgery, led him to change career...
This advertorial is sponsored by Bentley.
Stéphan Haulon (Hôpital Marie Lannelongue, GHPSJ, Paris, France) has extensive clinical experience using the BeGraft (Bentley, Hechingen, Germany) as a bridging stent in fenestrated endovascular aneurysm repair (FEVAR) procedures. He has conducted a pioneering study...
Corindus has been rebranded to Siemens Healthineers Endovascular Robotics and will sit as a dedicated business within the Advanced Therapies area of Siemens Healthineers, the company has announced.
This brand unification is the final step of the company integration process...
VentureMed Group, a privately held medical device innovator in access management for arteriovenous (AV) fistulas and grafts and vessel preparation for interventional treatment of peripheral arterial disease (PAD) announced data presented at the VEITHsymposium, (15–19 November, New York, USA)....