
Nicholas (Nick) Inston calls for “more creative solutions” to vascular access challenges, commending interventional radiologists’ innovative reputation as instrumental to increasing the number of treatment approaches available to physicians. Outlining the complexities of end-stage vascular access in an ever-increasing patient population, Inston expresses the need for a multidisciplinary team approach, claiming that the “outlook is improving” due to the fruitful cross-pollination of ideas and skills.
The age of the dialysis population continues to increase, and whilst some of this may be attributed to older patients initiating dialysis, the main component is improved survival in prevalent dialysis patients. As a consequence, the vascular access needs of this population become more challenging. As primary access sites become depleted, more creative solutions must be employed.
In a small but increasing group of patients, standard, multiple, autologous, and synthetic options may have all failed, and these patients may be deemed to have exhausted all vascular access options except for the use of a central venous catheter (CVC). This situation may be termed end-stage vascular access (ESVA).1
If a patient with a limited lifespan expresses a wish to remain on a CVC, this may be a suitable option. However, this group of patients are likely to require multiple interventions to maintain their CVC patency and may require kinase infusions and even emergency CVC exchange to maintain an ability to dialyse.2
In those patients keen to pursue definitive access options, classifying the underlying issues is helpful. This can be done on an anatomical basis such as the radiological classification outlined by the Society of Interventional Radiology,3 or by using a simplified, more basic classification, such as that described by Julien Al-Shakarchi (University Hospital Birmingham, Birmingham, UK).1
A simple classification for endstage vascular access failure is proposed based on a sequential broad categorisation of: stage 1) no upper limb vascular access option; stage 2) no lower limb vascular access options; stage 3) no vascular access options at any site.
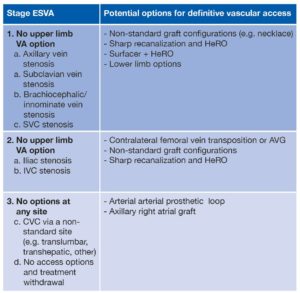
These can have the underlying anatomical problem added to allow more detailed definition (see Table 1).
An illustrative example may be a patient with tight superior vena cava (SVC) and brachiocephalic stenosis who has a right iliac vein occlusion and a left groin line. This would be classed as ESVA 2a, and possible solutions that could be considered are a left femoral vein transposition or loop thigh arteriovenous (AV) graft, attempts at recanalising the upper body central veins with recanalisation and stenting, or use of a Surfacer device (Bluegrass Vascular) and insertion of a HeRO (Merit Medical).4 Other options may be a non-standard graft configuration—here a right axillo-femoral graft may be possible. Peritoneal dialysis should not be forgotten.
When balancing each of these the risks and benefits should be carefully considered.
Lower leg access is not without complications. Femoral vein transposition, whilst having the advantage of use of the autologous vein and of reducing infection, is associated with lower limb congestion and venous insufficiency. Additionally, amputation has been reported with this method. Lower limb prosthetic grafts are associated with high rates of infection and low patency.5
Recanalisation of upper body central vessels may require expert interventional radiology skills and devices. The Surfacer device has potential, but robust data are not yet available to allow evaluation. Even with recanalised vessels the next step towards definitive access requires either extensive stenting or the use of a HeRO device. In patients with CVC infections (common in groin access), this is contraindicated. In such patients, a staged approach to recanalisation with initial upper body CVC, eradication of infection, and subsequent conversion to a HeRO graft may be a better approach.
Patients with arterial insufficiency can pose a problem and inadequate inflow may not only threaten the access, but also the limb. The resistance within longer grafts and HeRO grafts requires a suitable inflow including a reasonable blood pressure. Hypotension should be carefully evaluated when these options are being considered.
A further consideration is that of transplantation. Younger patients with ESVA may have had a long history of renal replacement and multiple CVCs. In the example above, a groin line in the only patent iliac vein poses a great threat to future kidney transplantation. It may be proposed that such patients are considered higher priority in organ allocation schemes as a working transplant is their optimal option. When considering such patients for lower limb access there is little to support avoidance of lower limb options and many transplants have been performed successfully above patent ipsilateral loop thigh grafts.
For patients where attempts to gain access are unsuccessful—stage 3 ESVA—there are still solutions that can be found.
This group traditionally were treated with a translumbar or transhepatic CVC. For many this was seen as a pre-terminal option, although data support reasonable short and mid-term survival.6 A relatively simple definitive option in these patients is the arterial arterial prosthetic loop (AAPL), which is an interposition graft in the subclavian/axillary artery.7
This unique configuration requires special consideration when cannulated and cannot be used for drug delivery but has been shown to have good patency with low complications.
Smaller series of axillary atrial grafts are published and advocated by some authors, however complications are greater and more significant.6
In deciding treatment options, an access life plan should be defined, as highlighted in the recent Kidney Disease Outcomes Quality Initiative (KDOQI) guidelines from the National Kidney Foundation.8 Where multiple options exist, these should be considered and staged with the simplest and safest option first, whilst retaining the possibility of further interventions later down the line.
The best approach to managing these difficult cases is with a multidisciplinary team, using a treatment utilising their experience and expertise, and chosen based on its applicability to the patient. The availability of surgical and radiological expertise is required not only for the index procedure, but also for many of these patients’ subsequent interventions and salvage procedures, conducted to maintain patency. An identified pathway should be defined with access to suitable facilities to allow this.
ESVA provides a very challenging problem for the vascular access surgeon, and the bad news is the incidence is likely to increase. Fortunately, there are an increasing number of possibilities for treatment with innovative devices and techniques. Historically, the prognosis was bleak for patients with ESVA, but by combining surgery and interventional radiology skills, the outlook is improving. As always in access, the approach used should be the right access, for the right patient, at the right time. This applies especially to ESVA, where the whole team’s input and detailed discussion with the patient are essential.
Nicholas Inston is a consultant transplant and vascular access surgeon at the University Hospital Birmingham NHS Trust, Birmingham, UK.
References
- Al Shakarchi J, Nath J, McGrogan D, Khawaja A, Field M, Jones RG, et al. End-stage vascular access failure: can we define and can we classify? Clin Kidney J. 2015;8(5):590-3.
- Kumwenda M, Dougherty L, Spooner H, Jackson V, Mitra S, Inston N. Managing dysfunctional central venous access devices: a practical approach to urokinase thrombolysis. Br J Nurs. 2018;27(2):S4-S10.
- Dolmatch BL, Gurley JC, Baskin KM, Nikolic B, Lawson JH, Shenoy S, et al. Society of Interventional Radiology Reporting Standards for Thoracic Central Vein Obstruction: Endorsed by the American Society of Diagnostic and Interventional Nephrology (ASDIN), British Society of Interventional Radiology (BSIR), Canadian Interventional Radiology Association (CIRA), Heart Rhythm Society (HRS), Indian Society of Vascular and Interventional Radiology (ISVIR), Vascular Access Society of the Americas (VASA), and Vascular Access Society of Britain and Ireland (VASBI). J Vasc Interv Radiol. 2018;29(4):454-60 e3.
- Davis KL, Gurley JC, Davenport DL, Xenos ES. The use of HeRo catheter in catheter-dependent dialysis patients with superior vena cava occlusion. J Vasc Access. 2016;17(2):138-42.
- Parekh VB, Niyyar VD, Vachharajani TJ. Lower Extremity Permanent Dialysis Vascular Access. Clin J Am Soc Nephrol. 2016;11(9):1693-702.
- Inston N, Khawaja A, Mistry H, Jones R, Valenti D. Options for end stage vascular access: Translumbar catheter, arterial-arterial access or right atrial graft? J Vasc Access. 2019:1129729819841153.
- Stephenson MA, Norris JM, Mistry H, Valenti D. Axillary-axillary interarterial chest loop graft for successful early hemodialysis access. J Vasc Access. 2013;14(3):291-4.
- KDOQI clinical practice guideline for vascular access: 2018. 2018.












