This article is an advertorial by Veryan Medical.
Authors: PA Gaines, T Sullivan, G Ansel, C Caro
Introduction
Medical advances in the treatment of femoropopliteal artery disease over the last decade reflect the on-going fight to maintain patency; and that battle is important for a number of reasons. Patency has a clear effect on the clinical success of treating both claudication and critical limb ischaemia; revascularisation to maintain patency adds risk to the patient and costs to the healthcare provider. Lumen loss after endovascular intervention has separate mechanisms depending upon whether it is early or late.1
Immediately after angioplasty, particularly in bulky and calcified disease, there may be recoil or dissection. Over the ensuing months there is a fibrotic adventitial response known as late negative remodelling, and an inflammatory response that leads to vascular smooth muscle cell proliferation, extracellular matrix formation and the development of neointimal hyperplasia. When the combination of remodelling and hyperplasia is excessive it is referred to as restenosis. The accepted treatment for recoil and flow limiting dissection observed at the time of intervention, and to prevent the constrictive effect of late negative remodelling, is to place a stent. Such an intuitive solution has resulted in clear benefit when compared to simple angioplasty.2,3
The more difficult problem of limiting restenosis now has two solutions which may be complementary—drugs and imparting swirling flow.
Using Swirling Flow® to benefit intervention in the femoropopliteal artery
It is clear that the distribution of atherosclerosis and the tendency of an artery to develop restenosis varies throughout the human arterial system; the femoropopliteal artery has both a high prevalence of atherosclerosis and a high tendency to develop restenosis.
Part of the reason for the variable distribution of native arterial disease was explained in the 1969 Nature article which drew attention to the relationship between high wall shear stress and a reduced tendency towards atherosclerosis.4
We all recognise that normal arterial blood flow is laminar. Less well known is that the normal pattern in the aorta and proximal branches is also spiral—referred to as swirling flow.5
Wall shear stress can be thought of as the velocity of blood against the internal wall of the vessel. Swirling flow increases wall shear stress when compared to non-swirling flow resulting in a reduced propensity to develop atherosclerosis when exposed to well recognised risk factors. Blood flow in the superficial femoral artery is naturally less swirling—hence the high prevalence of native disease (Figure 1).
Less well recognised is the effect of wall shear stress on the tendency to develop restenosis. Animal studies have shown that areas of low wall shear stress predict the development of neointimal hyperplasia in grafts and stents.6 Treating the superficial femoral artery of peripheral arterial disease patients with straight nitinol stents has been shown to impair wall shear stress.7 Evaluation of coronary stents in humans showed that the pattern of restenosis is determined by the distribution of wall shear stress.8,9
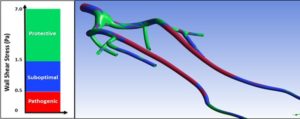
These observations led to the development of a self-expanding stent with a helical centreline that is capable of inducing swirling flow (Figure 2). The ability of this stent to reduce restenosis was first tested in a porcine model where a straight stent was placed in one carotid artery and a helical stent in the other of the same animal.

The study demonstrated that the helical centreline stent imparted helical curvature onto the artery and generated swirling flow; that the swirling flow significantly reduced the development of neointimal hyperplasia (Figure 3); and that a correlation between the degree of curvature and the reduction in neointimal hyperplasia was established. The BioMimics 3D stent was subsequently tested in the first randomised controlled trial (RCT) to directly compare two nitinol stents in the femoropopliteal segment.10

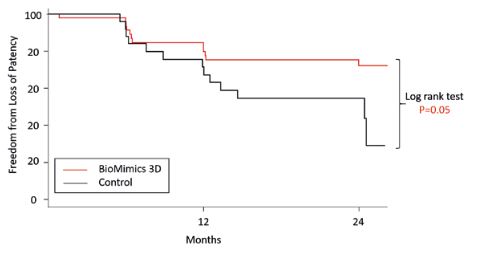
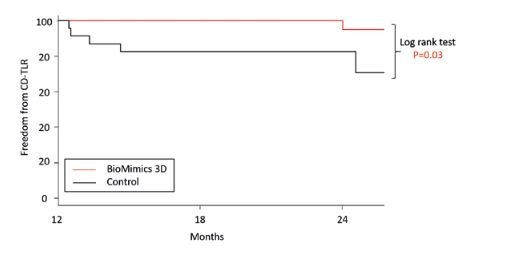
The MIMICS trial was a multicentre, core-lab controlled, prospective, randomised trial in which the BioMimics 3D stent was compared to a conventional straight stent control (Lifestent, Bard) in 76 patients with symptomatic occlusive disease of the superficial femoral and proximal popliteal arteries. Conventional radiographs and angiography confirmed that the BioMimics 3D stent imparts non-planar curvature to the diseased artery. Compared to the straight stent control the BioMimics 3D stent had significantly better primary patency through two years (Figure 4). There was no change in clinically driven target lesion revascularisation (CD-TLR) in the BioMimics 3D arm between 12 and 24 months whereas there was a three-fold increase in CD-TLR in the straight stent control arm over the same time period; a significant difference between the two stents (Figure 5).
The complementary effect of swirling flow and drug coated balloons
The pivotal trials showed improved performance over simple angioplasty but this was in a well-defined set of lesions and the importance of that in terms of generalisability of the value of DCBs deserves some attention.11–15 Severe calcification and an inability to completely pre-dilate the lesion were exclusions in these studies and effectively removed those lesions from any analysis. Furthermore, since only 12–26% of lesions were total occlusions this non-calcified, simple disease was unlikely to recoil after angioplasty resulting in a bailout stent rate of only 2.5–7%. When the same DCBs are used in more routine clinical practice, and documented within the Global Registries, the lesion patency and CD-TLR rates remain good.16,17 This is because these more clinically generalisable cohorts are in fact measuring the outcome of DCB plus stent. This everyday disease is more complex than those recruited to the pivotal trials resulting in an average stent rate of 28–35.5%. The stent rate is clearly related to both lesion length and the CTO rate. In IN.PACT Global for instance, when the length of lesion exceeds 25cm, the stent rate is 53%, and in total occlusions the stent rate is 47%.17
The Kaplan Meier survival estimates from the pivotal trials demonstrate that the improvement in patency and reduction in CD-TLR over simple angioplasty occur between six and 12 months, but after this time the Kaplan Meier curves are the same.18 This trend is seen also in the global registries.16,17 It is clear that there are limitations to the use of DCBs in the femoropopliteal segment:
1. A ‘DCB-only’ approach is not appropriate to clinical practice outside pivotal trials. Whilst the bail-out stent rate was low in the pivotal trials the global registries demonstrate that a much higher use of stents is required to maintain high patency and low CD-TLR rates.
2. Loss of patency is a combination of recoil, late negative remodelling and restenosis. DCBs alone clearly do not provide the scaffolding that a stent can to overcome the recoil and remodelling. Indeed, DCBs appear to have been tested in an environment that would avoid likely recoil; lesions were short and both calcified lesions and lesions that demonstrated recoil after the initial pre-dilatation were excluded in the pivotal trials.
3. The antiproliferative effects are necessarily time limited and yet contemporary data show that loss of patency due to restenosis in the femoropopliteal segment occurs out to three to four years.19 Something other than the drug is required to affect these late events.
It would appear therefore that in common clinical practice a stent is required in a significant proportion of DCB-treated lesions. If a stent is used to support a DCB it would appear sensible to use a device with good outcomes that would not only provide the scaffolding that DCB-only therapy lacks, but could extend the period of low CD-TLR past the initial 12 months when DCBs are effective. The BioMimics 3D stent was seen in a RCT to have a better patency and a reduced CD-TLR rate when compared to a straight nitinol stent. Furthermore, as there was no CD-TLR between 12 and 24 months in the BioMimics 3D arm of the RCT, this suggests that the use of a proven DCB and BioMimics 3D might be the ideal combination.
Continuing the BioMimics programme
The clinical validation of swirling flow as a key component in a combined approach to limit restenosis is continuing across a range of studies in the BioMimics Clinical Programme.
Following on from MIMICS, MIMICS-2 is a prospective, multicentre, interventional study designed to evaluate the safety and effectiveness of BioMimics 3D in the treatment of patients with symptomatic femoropopliteal disease. The study is being conducted under a US Food and Drug Administration (FDA) Investigational Device Exemption (IDE), with Japanese Pharmaceuticals and Medical Devices Agency (PMDA) concurrence under the “Harmonisation By Doing” initiative, in order to provide safety and effectiveness data that are intended to support marketing applications for BioMimics 3D in both the USA and Japan. Twelve-month results will be announced at the Charing Cross International Symposium in April (CX Symposium; 24–27 April, London, UK).
In addition, MIMICS-3D is an ongoing prospective, multicentre, observational registry evaluating the safety, effectiveness and device performance of BioMimics 3D within a real-world clinical population in a minimum of 500 patients across Europe.
This combined clinical database of over 1000 cases is intended to provide significant validation of the unique helical shape of BioMimics 3D and the clinical benefits of swirling flow.













