PQ Bypass has announced the enrolment of the first patients in the pivotal DETOUR II trial in the USA. The trial is a prospective, single-arm clinical trial designed to evaluate the DETOUR System for percutaneous femoropopliteal bypass.
The study will enrol up to 292 patients at up to 40 centres in the USA and Europe with patient follow-up out to 36 months. The trial also includes a prospective economic study to collect quality-of-life outcome measures and cost data, including re-hospitalisations.
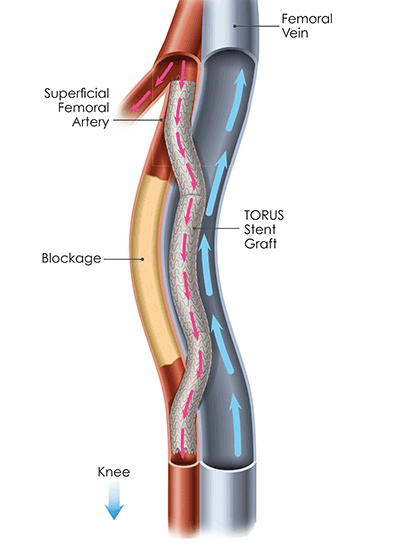
Vasili Lendel and André Paixao initiated the DETOUR II trial in the USA at Arkansas Heart Hospital in Little Rock, enrolling two patients with complete blockages in the superficial femoral artery (SFA) longer than 30 centimetres.
“We are excited to start treating patients in the DETOUR II investigational device exemption (IDE) trial, studying a procedure that may improve upon existing treatment options for patients with long-segment femoropopliteal disease,” says Sean P Lyden, professor and chairman of the Department of Vascular Surgery, Sydell and Arnold Miller Family Heart & Vascular Institute at Cleveland Clinic, and national co-principal investigator of the trial. “The DETOUR II trial is designed to provide definitive safety and effectiveness data on the new percutaneous bypass procedure, which has the potential to provide the effectiveness of open surgery through a minimally-invasive endovascular approach for these challenging lesions.”
Enabled by PQ Bypass’ patented DETOUR System, the DETOUR procedure creates an endovascular bypass, using proprietary stent grafts, that originates in the SFA, travels through the femoral vein, then returns into the popliteal artery. This new pathway is designed to re-direct oxygen-rich blood around the diseased part of the vessel, restoring blood flow to the lower leg and foot of the patient.
“DETOUR is an innovative, yet intuitive endovascular procedure to create a bypass in a channel already provided by the body, but without performing open surgery,” said Jihad Mustapha, CEO of Advanced Cardiac and Vascular Amputation Prevention Centers, clinical associate professor of medicine at Michigan State University College of Osteopathic Medicine, and national co-principal investigator of the DETOUR II study. “We look forward to working with the trial sites and, ultimately, to seeing the results from this landmark study.”
The lack of an optimal treatment alternative for long-segment femoropopliteal occlusions is a persistent issue for patients with advanced PAD. Published literature has demonstrated that conventional endovascular approaches result in at least 30¬-40% lower patency in these blockages when compared to the same treatments used for shorter lesions. Studies have also shown that these patients could experience a 40% rate of re-treatment within 12 months.
In March 2017, PQ Bypass received CE mark approval for the DETOUR System. In November 2017, PQ Bypass received conditional approval of their IDE for the DETOUR System. The DETOUR System is an investigational device in the USA.