Zoom in on a tête-à-tete between Marianne Brodmann (Graz, Austria) and Giovanni Torsello (Munster, Germany) who talk through the latest clinical evidence that directly compares drug-eluting stents in a head-to-head fashion.
Brodmann and Torsello talk through thorny issues such as which patients qualify as “high-risk” patients who would benefit most from the use of paclitaxel-coated devices in light of the most recent US FDA guidance on the topic. They also outline the poor outcomes obtained before the era of drug-elution with bare metal technologies.
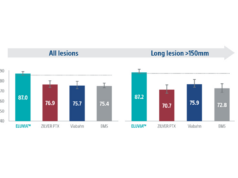
Torsello highlights the latest results from drug-eluting stent trials, particularly the IMPERIAL trial that compares the Eluvia drug-eluting stent platform (Boston Scientific) with the Zilver PTX drug-eluting stent platform (Cook Medical), emphasising the importance of polymer-mediated, low dose, sustained local drug release over time.
Both look to the near future and the upcoming EMINENT trial that will compare Eluvia with best practice US FDA approved bare metal stents, and comment on the importance of long-term data from the Muenster Eluvia registry which is testing this drug-eluting stent system in a complex patient cohort with lesions at high risk of restenosis to provide evidence of its clinical benefits in this patient population.
This video is sponsored by Boston Scientific.