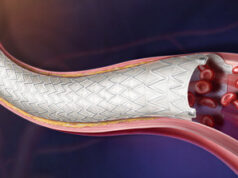
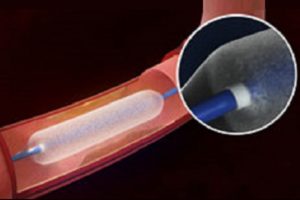
 The US Food and Drug Administration (FDA) has approved expanded sizes of the Lutonix 018 drug-coated balloon (DCB) to treat long superficial femoral artery lesions in patients with peripheral arterial disease.
The US Food and Drug Administration (FDA) has approved expanded sizes of the Lutonix 018 drug-coated balloon (DCB) to treat long superficial femoral artery lesions in patients with peripheral arterial disease.
According to a press release, the Lutonix 018 DCB provides clinicians with a variety of longer sizes allowing more options for 7mm diameter vessels. This includes four new lengths: 80mm, 100mm, 150mm and 220mm. The Lutonix 018 DCB is designed to perform over small guidewires, reduce guidewire exchanges and enable alterative access sites.
Lutonix 018 DCB is available in the USA, and is currently undergoing the regulatory approval process in Europe.