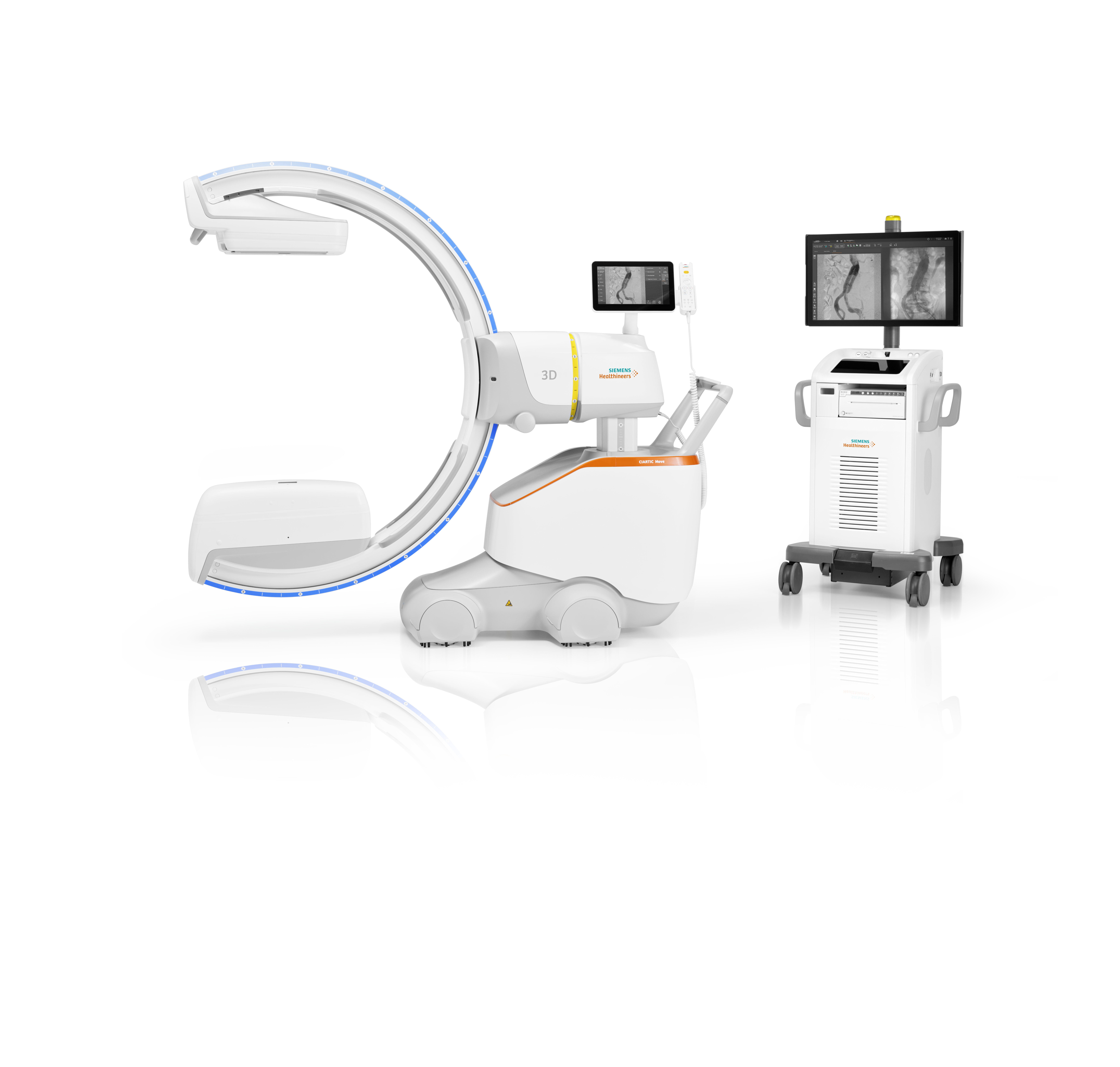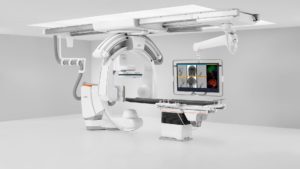
The US Food and Drug Administration (FDA) has cleared the Artis icono, a family of angiography systems from Siemens Healthineers that permit a range of minimally invasive procedures to be performed in a single interventional suite.
The Artis icono biplane system is engineered for optimal utilisation in neuroradiology and abdominal imaging, while the Artis icono floor is a floor-mounted, single-plane system for vascular, interventional cardiology, surgical, and oncology procedures. Both systems in the Artis icono family expand the reach of precision medicine.
At its core, the Artis icono platform delivers the new OPTIQ image chain, which fundamentally redesigns image processing for 2D imaging. OPTIQ increases image quality across a wide range of C-arm angles and patient weights, regulating acquisition parameters to automatically achieve optimal image contrast at patient radiation doses that conform to the As Low As Reasonably Achievable (ALARA) guiding principal for radiation safety. The platform also improves the Siemens Healthineers roadmap function, which creates subtracted angiography images for easier navigation of the patient’s vascular system during subsequent fluoroscopy, allowing dose reduction during the fluoro mode.
Additionally, the Artis icono platform significantly enhances ease of use through Case Flows, which provide personalised workflow plans to optimise imaging parameters and system positions, in addition to displaying layouts for the entire procedure. Case Flows adapt to user needs, providing flexibility when executing certain sequences. Users can therefore standardise procedures across multiple Artis icono labs for improved outcomes and more consistent documentation.