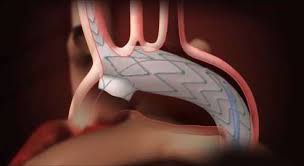
Mario Lachat (University of Zürich, Zürich, Switzerland) presented mid-term results of an open-label, non-randomised single-arm investigational clinical study of the Nexus (EndoSpan) aortic arch stent graft system in 25 patients. Speaking at Controversies & Updates in Vascular Surgery (CACVS; 7–9 February, Paris, France), Lachat reported that the early data were “very encouraging”, and disclosed that the company expects CE approval for the device in the first quarter of 2019.
“Nexus is a relatively simple arch procedure,” Lachat said, noting that “ease of implementation will allow for greater physician utility.” In 25 patients, successful access, deployment and procedural survival was attained in 100% of cases. Mean endoluminal repair time was 68 minutes, with a total mean procedural time of 186 minutes. At 30 days, two deaths were reported (8%; one ventricular fibrillation, one cardiac arrest) and two non-disabling strokes (8%). One further death (4%; cardiovascular accident) was reported in follow-up >30 days.
Highlighting the 100% immediate technical success rate, Lachat noted the study demonstrated the “excellent safety profile” of the device, with no aneurysm-related deaths.
Lachat concluded that longer-term data are needed to further study performance of the Nexus aortic arch stent graft system.













