The US Food and Drug Administration (FDA) has granted Breakthrough Device Designation to Medtronic for its novel Valiant thoracoabdominal aneurysm (TAAA) stent graft. A press release reports that the device is designed to offer an off-the-shelf solution for complex endovascular aneurysm repair (EVAR) procedures, adding that the Valiant TAAA has a “size matrix to enable broad patient applicability for one of vascular surgery’s most difficult pathologies”.
According to the press release, the aim of FDA Breakthrough Device Designation programme is to help patients “receive more timely access to breakthrough technologies that have the potential to provide more effective treatment or diagnosis for life-threatening or irreversibly debilitating diseases or conditions”. Specifically, with the programme, the FDA will provide Medtronic with priority review and interactive communication regarding device development and clinical trial protocols through to commercialisation decisions. Murray Shames (Division of Vascular and Endovascular Surgery, University of South Florida College of Medicine, Tampa, USA), an investigator for Valiant TAAA, comments: “Breakthrough designation from the FDA means that we will be able to deliver this much needed treatment to patients sooner than expected. With an open surgery mortality rate of 25%, it is critical that we deliver for this unmet patient need. Physicians and industry must continue to innovate and provide hope for those with challenging disease states.”
The press release reports that, at present, the Valiant TAAA is being evaluated in five physician-sponsored investigation device exemption (IDE) trials for the management of thoracoabdominal aneurysms. Furthermore, the Valiant system was developed in collaboration with Patrick Kelly (Sanford Health Commercialization, Sioux Falls, USA)—a vascular surgeon and inventor who specialises in complex vascular disease. John Farquhar, vice president and general manager of the Aortic business, which is part of the Cardiac and Vascular Group at Medtronic, says: “In addition to a high mortality rate for open surgical repair, physicians do not have good options when it comes to treatment for failed endografts. The FDA’s breakthrough designation for Valiant TAAA and our collaboration with leading physicians is an example of our willingness to take a courageous approach and challenge the limitations of current treatment options. This is about going further, together to improve patient outcomes.”
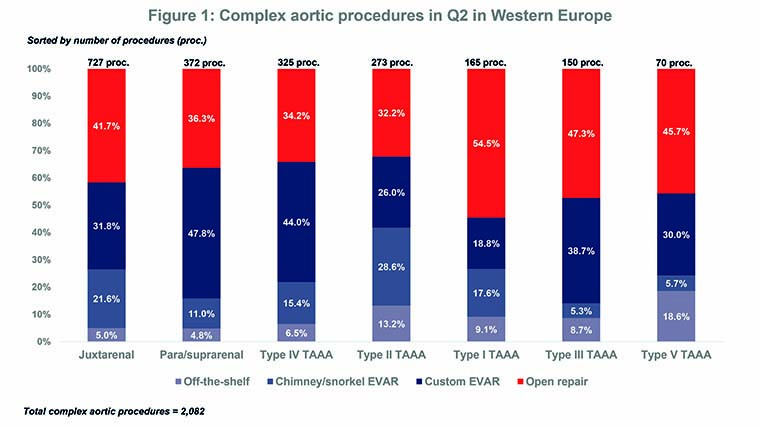
Complex open surgery is the standard of care for thoracoabdominal aneurysms but, according to the press release, it is associated with high morbidity and mortality. Furthermore, 40% of patients are not considered candidates for surgical treatment. Of note, the BIBA MedTech Aortic Segmentation Monitor shows that the endovascular approach was the most common method for treating complex aneurysms—e.g. thoracoabdominal aneurysms or juxtarenal aneurysms—in Western Europe in Q2: 60.1% vs. 39.9% for open surgery.
The Monitor also indicates that EVAR with a custom-made device(s) is typically the predominant endovascular approach for such complex aneurysms while EVAR with an off-the-shelf device (similar to the Valiant TAAA) tends to be the least used approach. Figure 1 shows the breakdown of complex aortic procedures by aortic segment and procedure type. Medtronic had the second highest share (20.2%) of the Q2 complex EVAR market but, with its Zenith range, Cook Medical was the market leader (47.9%).
Expanding the Valiant TEVAR range
As well as expanding its complex EVAR range, Medtronic also plans to add to its range of device for thoracic endovascular aortic (TEVAR) procedures. The Valiant Navion Left Subclavian Artery (LSA) stent graft received FDA Breakthrough Device Designation in May this year and, the press release explains, its aim is “to allow for more timely delivery of treatment options for patients in left subclavian artery coverage during TEVAR”.
The Valiant Navion LSA is based on the investigational Valiant Mona LSA device and the Valiant Navion stent (which received FDA approval last year). Like Valiant Navion, the Valiant Navion LSA has a low-profile design but also has a branch cuff that is intended to replicate the natural anatomy of the LSA to maximise seal and patency.
“About 40% of thoracic aortic aneurysms in branch vessels involve coverage of the LSA. With no currently available off-the-shelf solutions, surgical bypass remains the standard of care for these patients. By leveraging our low-profile, Valiant Navion platform, we hope Valiant Navion LSA will expand endovascular treatment options to those in need. It is a matter of putting patients first and delivering innovation where it matters most,” comments Farquhar.

Source: BIBA MedTech Aortic Segmentation Monitor
Data from the Valiant EVO global clinical trial, which was published in the Journal of Vascular Surgery earlier this year, show that only 2.3% of patients who received a Valiant Navion device for the management of descending thoracic aneurysms had a major device effect at 30 days. Furthermore, no access or deployment failures were reported, the rate of freedom from all-cause mortality (at 30 days) was 97.7%, and the rate of endoleaks was 2.5% (one type Ia and one type II). The investigators Ali Azizzadeh (Cedars-Sinai Medical Center, Los Angeles, USA) conclude: “Access/deployment failures, major device effects, and endoleak were rare in the first 30 days of the Valiant Evo clinical trial. The Valiant Navion thoracic stent graft system has shown encouraging 30-day results in this challenging cohort and trial patients will continue to be followed through five years.”
The Aortic Segmentation Monitor shows that 31.8% of non-valvular aortic procedures (21,213 overall) performed in Q2 were for the thoracic aorta. Furthermore, of these thoracic procedures, 37% were with an endovascular approach vs. 4% for a hybrid procedure vs. 59% for open procedures. In contrast to the complex EVAR market, Medtronic was the leader of the thoracic endovascular market (36%). see Figure 2.
This article is part of a series of BIBA Briefings columns in Vascular News. For previous columns, click here.
BIBA Briefings Insights reports give in-depth analysis of the latest market intelligence from BIBA MedTech Insights. They also review that the latest technology news and pipeline developments.
For editorial enquiries, please contact Dawn Powell: [email protected]
For sales enquiries (including BIBA MedTech Insights), please contact Merveille Anderson:[email protected]













