Laminate Medical Technologies (Laminate) has announced the completion enrolment of the VALUE study for the VasQ External Support. The postmarket study enrolled 80 patients (50 upper arm and 30 forearm fistulas) for sites across Germany, France, Spain, and the UK and will be followed for one year. The study is the first prospective study to assess the use of the device in the forearm. The forearm fistula carries a higher risk of primary failure relative to upper arm fistulas but is preferred by surgeons as it preserves more options for the patient’s future access needs.
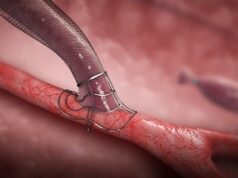
VasQ is intended to promote usability of both forearm and upper arm fistulas by providing an external support to optimise the haemodynamics and reinforce against the excessive mechanical stress experienced by the surgically created connection of the vein to the artery. The clinical benefits of VasQ has been demonstrated over the standard of care for upper arm fistulas in a randomised controlled study recently published in the American Journal of Kidney Disease. For the VALUE study, an interim analysis of the first 63 patients enrolled was released this fall, which reported similarly beneficial clinical outcomes for both upper arm and forearm fistulas.
Gaspar Mestres of the Cardiovascular Institute Hospital Clinic, Barcelona, Spain stated, “VasQ has become an essential part of my clinical practice to give my patients’ fistulas a greater chance for success. Increasing usability and minimising reinterventions with VasQ provides tremendous benefit to my patients’ health while on haemodialysis. I am excited to see the longer-term benefits of the device for both forearm and upper arm fistulas validated for clinics across Europe in VALUE.”
“We are grateful to our principle investigators for their participation in VALUE,” said Laminate CEO Tammy Gilon. “The expansion of our clinical evidence to support the benefits of VasQ in forearm patients is critical to our company goal of reducing fistula primary failure rates globally.”