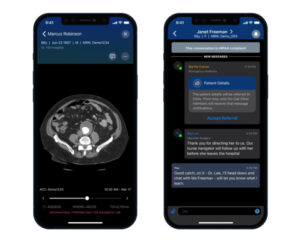
Viz.ai recently announced it has received US Food and Drug Administration (FDA) 510(k) clearance for its algorithm intended to detect suspected abdominal aortic aneurysm (AAA). Viz AAA is the first FDA-cleared AI-powered solution for the detection and triage of suspected AAA, the company claims.
“There is an estimated 1.1 million Americans living with an AAA. The majority of these patients are asymptomatic and many are unaware of their disease until a rupture occurs. This is a catastrophic medical emergency, resulting in over 10,000 deaths each year,” said Philip Batista (Cooper University Health Care, Camden, USA). “This algorithm is a powerful new tool for healthcare professionals to more readily identify and capture individuals with AAA and, importantly, automatically refer those at imminent risk for rupture.”
“I look forward to the new aortic platform here at TriHealth, as its implementation will allow for enhanced identification, triage and team member communication, resulting in the best possible care for our aortic patients,” said Patrick Muck (TriHealth Heart Institute, Cincinnati, USA). “We have been using the Viz software for the detection of suspected pulmonary embolism over the last several months and have seen improvements in patient care across our institution.”
Viz AAA uses artificial intelligence (AI) to automatically search for the presence of a AAA from computed tomography angiography (CTA) from any scanner in a hospital network, a press release details. The new AI algorithm and clinical workflow solution will be a part of the Viz Aortic Module, an AI solution designed to accelerate treatment decisions for all aortic pathology.
Viz.ai notes that the software can be integrated into existing hospital workflows, allowing physicians to quickly and easily identify patients who may have a suspected AAA and take appropriate measures to prevent a rupture.













