Endospan has announced the initial experience with its custom-made Nexus Duo aortic arch stent graft system. Theodosios Bisdas (Athens Medical Centre, Athens, Greece) presented the multicentre European experience of the first ten patients treated with Nexus Duo at this year’s Leipzig Interventional Course (LINC 2023; 6–9 June, Leipzig, Germany).
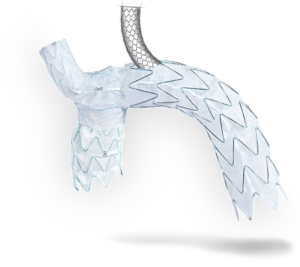
The Nexus Duo is custom-built based on each patient’s anatomy and physician’s preferences. The device is built on the clinically established technology of the Nexus aortic arch stent graft system, which designed to mitigate stroke risks and provide high procedural success.
Bisdas stated: “Ten patients were successfully treated with clinical outcomes in line with Nexus single branch. Aortic arch pathologies often require a tailored solution for each complex anatomy. The Nexus Duo provides many anatomical variations while optimising the treatment.”
Mario Lachat (Hirslanden Klinik Zurich, Zurich, Switzerland) added: “The Nexus Duo’s second branch channel allows transfemoral introduction of all stent grafts necessary for complete endovascular arch repair, with minimal manipulation of supra-aortic vessels. Latter mitigating significantly the stroke risk.
“Our experience with the Nexus in Europe and the US TRIOMPHE IDE [investigational device exemption] study both validate the need for an endovascular aortic arch solution,” says Kevin Mayberry, CEO of Endospan. “The Nexus Duo expands the Nexus platform. The current single branch Nexus is off-the-shelf and available for patients requiring urgent life-saving treatment. The Nexus Duo gives physicians more options for less urgent cases to treat the most complex aortic arch anatomies.”
A press release notes that open surgical aortic arch repair maintains high mortality and morbidity rates. It details that over 120,000 patients in the USA and Europe have thoracic aortic arch disease each year, but only about 25% receive diagnosis or treatment. Patients at high risk for surgery or with complex anatomy often lack treatment options. According to Endospan, the Nexus and Nexus Duo systems provide a minimally invasive alternative, reducing procedure and hospitalisation times.
The company advises that the Nexus aortic arch stent graft system is an investigational device, limited by US law to investigational use. It adds that the Nexus Duo aortic arch stent graft is a custom-made device based on the existing Nexus aortic arch stent graft system, a CE-marked endovascular branch system for the aortic arch.













