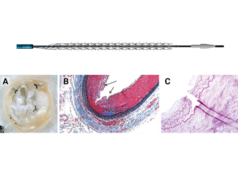
 Cordis has announced positive 24-month results from the Selution SFA Japan trial. The prospective, multicentre, single-arm trial is designed to assess the safety and efficacy of the Selution SLR drug-eluting balloon (DEB) for the endovascular therapy of de novo and non-stented restenotic lesions in the superficial femoral artery (SFA) and the popliteal artery (PA). The findings were presented at the Japan Endovascular Treatment (JET) conference (14–16 June, Fukuoka, Japan).
Cordis has announced positive 24-month results from the Selution SFA Japan trial. The prospective, multicentre, single-arm trial is designed to assess the safety and efficacy of the Selution SLR drug-eluting balloon (DEB) for the endovascular therapy of de novo and non-stented restenotic lesions in the superficial femoral artery (SFA) and the popliteal artery (PA). The findings were presented at the Japan Endovascular Treatment (JET) conference (14–16 June, Fukuoka, Japan).
“The Selution SLR DEB outcomes demonstrate patient benefits that are sustained out to 24 months and can match proven paclitaxel DCB performance in a complex patient population,” said Osamu Iida (Osaka Police Hospital, Osaka, Japan). “These are promising results that build confidence in Limus drug-eluting balloons being a safe and effective option for SFA treatment while avoiding any paclitaxel related concerns.”
The long-term follow-up of Selution SLR DEB confirmed efficient sirolimus drug transfer and retention with safety and efficacy sustained through 24 months in a complex population that included 60.3% diabetics, mean lesion length of 127mm, 17.2% total occlusions, and 47.8% involvement of the popliteal. At 24 months, Selution SLR DEB delivered primary patency of 83.0% and 94.5% freedom from clinically-driven target lesion revascularisation (CD-TLR).
“The Selution SFA Japan trial continues to build confidence in the Selution SLR DEB technology. The Selution SLR DEB delivers some of the highest patency rates amongst SFA paclitaxel DCB studies and differentiates from other Limus-based devices. The ability to achieve durable clinical results in a challenging patient population provides clinicians a solution that will offer value to real world patients” said George Adams, chief medical officer at Cordis. We are excited for the evolution of patient care and to be leading that journey by expanding access to Selution SLR DEB.”
Cordis add that the Selution SFA Japan trial is one of four randomised controlled trials and one real world registry data sets currently underway across coronary, SFA and below-the-knee (BTK) arteries.