This advertorial is sponsored by Gore.
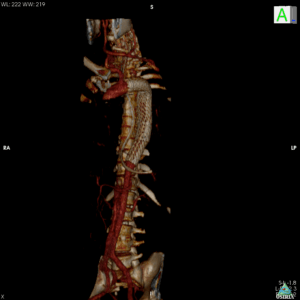
During a satellite symposia at the 2023 Charing Cross (CX) International Symposium (25–27 April, London, UK), speakers addressed the “key factor” of conformability in the endovascular repair of multiple vascular pathologies.
First to address the audience, Michele Antonello (Padua, Italy) argued that true conformability can reduce the risk of distal sine in the endovascular repair of type B aortic dissection (TBAD). Speaking to Vascular News ahead of CX, Antonello remarked that conformability is a “key factor” in device selection, and especially so in relation to TBAD as the aorta is more “fragile” in this pathology. “Theoretically,” the presenter explained, “the less you try to modify the anatomy of the aorta, the less the endograft will shock the adventitia.”
Antonello explained that the GORE® TAG® Conformable Thoracic Stent Graft with ACTIVE CONTROL System “keeps the curvature of the arch of the aorta” and stays in the position to which it is moved. This has the effect of reducing the possibility of trauma to the aortic wall, Antonello noted. In addition, Antonello detailed that there is a theoretically lower risk of a new dissection created by the TAG Conformable Device than with a standard device.
Data from the GREAT Registry of a real-world population have shown that the TAG Conformable Device has a low distal sine rate of 4% in chronic TBAD aortic dissection patients, Antonello communicated with the audience at CX. The results from this study are “particularly good,” he shared with this newspaper, citing low incidence of three events: neurological complications, new endoleaks and new dissections created by the endograft. “I think [the TAG Conformable Device] is the only endograft that has such robust data coming from a real-life registry.”
Later in the symposium, Jan Heyligers (Tilburg, Netherlands) addressed the question ‘Does conformability have an impact on the outcomes of endovascular repair of iliac limb performance?’ In an interview with Vascular News, Heyligers remarked that conformability is important in treating iliac pathology since the flow and curvature of the iliacs are challenging. “We are aiming to have a device that is conformable to the anatomy.”
According to Heyligers, there are differences in conformability in the iliac branch devices (IBDs) available on the market. “There are some devices that are stiffer than others,” he explained, noting that the GORE® EXCLUDER® Iliac Branch Endoprosthesis (IBE) seems to be the most conformable to the actual anatomy, based on his clinical experience with the device.
Heyligers detailed that he and his team have researched the Gore endograft in tortuous anatomy. He shared, for example, that in one study they implanted 17 IBEs and conducted electrocardiography (ECG)-gated computed tomography (CT) analysis pre-and postoperatively. “We have learned that after implantation of an IBE there is a significant increase in motion,” he reported.
“In general, the more straightforward your anatomy is, the more probable it is that your device will endure,” Heyligers remarked. Then again, he noted that the more adapted the system is to the anatomy, the more likely it is to translate into good outcomes, irrespective of the anatomy. The IBE is a “proven solution” in this regard, he said in summary.
Giovanni Pratesi (Genoa, Italy) also spoke during the session, sharing his insights on the impact of conformability on the outcomes of endovascular repair of abdominal aortic aneurysm (AAA) with angulated infrarenal necks.
In endovascular aneurysm repair (EVAR), Pratesi told Vascular News, conformability is “intrinsic” to how a stent graft adapts to a patient’s anatomy and can improve procedural outcomes.
During the symposium, Pratesi discussed three-year follow-up data from the European multicentre EXCeL registry on the GORE® EXCLUDER® Conformable AAA Endoprosthesis with ACTIVE CONTROL System, noting how it adds to the long-term evidence from numerous trials and registries worldwide that support the efficacy of all Gore conformable devices.
The indication to treat challenging anatomies within the Instructions for Use (IFU) is “undoubtedly” an advantage of the GORE EXCLUDER Conformable Device, Pratesi remarked. In particular, he explained, the device allows for the treatment of short as well as severely angulated necks, offering the capability to treat anatomy previously considered unsuitable for standard EVAR.












