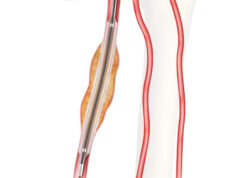
 As announced at the Society for Vascular Surgery (SVS) Vascular Annual Meeting (VAM 2023; 14–17 June, National Harbor, USA), the Advance Serenity hydrophilic percutaneous transluminal angioplasty (PTA) balloon catheter product line is now available with even more options. Interventionists who perform peripheral intervention procedures in the USA and Canada now have access to the Advance Serenity product for below-the-knee and above-the-knee procedures to treat patients with peripheral artery disease (PAD).
As announced at the Society for Vascular Surgery (SVS) Vascular Annual Meeting (VAM 2023; 14–17 June, National Harbor, USA), the Advance Serenity hydrophilic percutaneous transluminal angioplasty (PTA) balloon catheter product line is now available with even more options. Interventionists who perform peripheral intervention procedures in the USA and Canada now have access to the Advance Serenity product for below-the-knee and above-the-knee procedures to treat patients with peripheral artery disease (PAD).
Now, Advance Serenity is available to help physicians treat complex lesions with more balloon sizes. Advance Serenity is still made with its laser-formed, low-profile entry tip. The hydrophilic coating provides lubricity to navigate through diseased vessels with reduced friction. The Advance Serenity 18 is now larger, with balloon diameters from 5mm to 10mm, and is compatible with 4, 4.5, 5 and 5.5Fr sheaths to help physicians treat complex lesions with options to include larger-vessel PAD. The Advance Serenity 14 and Serenity 18 balloons are still available in 4Fr sheath compatibility and 2mm to 4mm balloon diameters. All Advance Serenity products are available in both the USA and Canada and will be available in Europe in the coming months.
“After looking at the needs of patients with this condition, we built on our previous innovation and are excited to expand the sizes, uses and availability of this product. The Serenity balloon complements our balloon portfolio for interventionists who are treating patients with complex lesions,” said Alec Cerchiari, director of product management for Cook’s PAD and Venous Specialty.
The catheter is manufactured by Surmodics and distributed by Cook Medical. You can learn more about the Serenity product here.