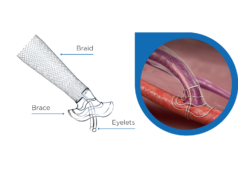
As part of the main scientific programme at Dreiländertagung 2016 (4–8 October, Bern Switzerland), Peter Olschewski has reported “exceptionally promising results” from the first six VasQ (Laminate Medical Technologies) cases performed in a single-centre experience in Charité University Hospital, Berlin, Germany. VasQ is an external support device for arteriovenous fistulae (AVF) implanted during surgical fistula creation.
Olschewski told Dreiländertagung delegates that a surgical brachiocephalic AVF is “the first choice if there is no option for a distal AVF in the forearm. All patients treated with VasQ did fit this criterion.”
Olschewski said that previously-published literature has reported up to a 38% failure rate in AVF surgery, meaning that there is a need to improve these outcomes. He presented data on six patients treated with the VasQ and an average follow-up of 80 days post-implantation. Olschewski explained that all six patients were able to receive haemodialysis (five were already on dialysis and one was still pre-emptive but eligible for dialysis). He also confirmed that clinical results of the cases reflect the “compelling” results of Laminate Medical´s VasQ trial, published by Eric Chemla (St George’s Hospital, London, UK) in the Journal of Vascular Access earlier this year. Furthermore, he highlighted the “great advantage” of using VasQ without changing anything in the standard AVF procedure.
To support the need for additional multicentre clinical data, which Olschewski alluded to in his lecture, Laminate Medical is launching a multinational, clinical programme to collect data in both Europe and the USA on VasQ´s performance in dialysis access surgeries.