Tag: VasQ
Laminate Medical announces first implantation of VasQ AVF device
Laminate Medical has announced the first implantation of their VasQ device in the USA, performed by Ari Kramer (Spartanburg Regional Hospital, Spartanburg, USA). The...
Laminate Medical announces FDA clearance for VasQ AVF creation device
Laminate Medical Technologies has announced their flagship device, the VasQ External Vascular Support, has been cleared by the US Food and Drug Administration (FDA)...
VasQ external support comparative US study results presented at VASA 2022
A Laminate Medical Technologies press release reports that the US VasQ external support comparative study results were presented this month for the first time...
Largest real-world experience to date with VasQ device indicates long-term benefits...
The VasQ external support device (Laminate Medical) has demonstrated long-term benefits for radiocephalic (forearm) arteriovenous fistula (AVF) creation in a retrospective analysis of 150...
VasQ external support awarded NUB Status 1 reimbursement renewal for 2021...
The German Institute for Hospital Remuneration (InEK) has renewed the new examination and treatment methods (NUB) Status 1 designation of VasQ external support (Laminate...
VasQ device improves AVF creation in haemodialysis patients
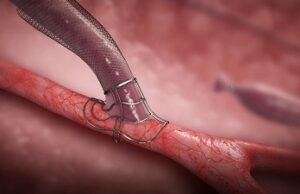
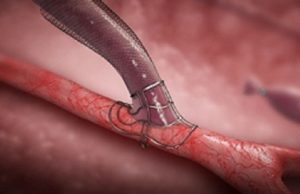
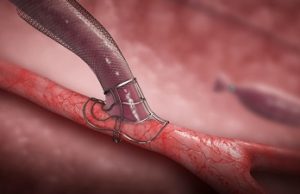
VasQ, a high haemocompatibility biosynthetic vascular device from Laminate Medical Technologies, could be protective against the haemodynamic modifications that occur during arteriovenous fistulae (AVF)...
VasQ external support awarded breakthrough device designation by the FDA
The US Food and Drug Administration (FDA) has designated Laminate Medical’s VasQ external support for the creation of arteriovenous fistulas (AVF) in haemodialysis patients...
Published real-world experience demonstrates VasQ External Support improves outcomes when adopted...
Robert Shahverdyan, head of the Vascular Access Center at Asklepios Klinik Barmbek, Hamburg, Germany, has recently published a retrospective analysis of his first 32...
Enrolment of the VasQ external support US pivotal study now complete
Laminate Medical Technologies has announced the completion of enrolment into the VasQ external support US pivotal study. The study was conducted at 17 sites...
Enrolment complete for VALUE, the prospective EU VasQ external support postmarket...
Laminate Medical Technologies (Laminate) has announced the completion enrolment of the VALUE study for the VasQ External Support. The postmarket study enrolled 80 patients...
VasQ External Support demonstrates significant improvement in fistula usability
Laminate Medical Technologies (Laminate) has announced that the American Journal of Kidney Disease has published the first multicentre, randomised controlled study of the VasQ...
Laminate Medical Technologies announces enrolment of first forearm patients in pivotal...
Laminate Medical Technologies has announced the enrolment of their first forearm fistula patients in their US pivotal trial of the VasQ device. VasQ is...
Laminate Medical Technologies announces first forearm fistula cases in Germany
Laminate Medical Technologies (Laminate), a privately-held start-up developing the VasQ implanted blood vessel external support device for patients requiring arteriovenous (AV) fistula as vascular access...
Laminate Medical Technologies announces first forearm fistula cases in Germany
Laminate Medical Technologies (Laminate), a privately-held start-up developing the VasQ implanted blood vessel external support device for patients requiring arteriovenous (AV) fistula as vascular access...
Laminate Medical Technologies announces first forearm fistula cases in Germany
Laminate Medical Technologies (Laminate), a privately-held start-up developing the VasQ implanted blood vessel external support device for patients requiring arteriovenous (AV) fistula as vascular access...
First patients enrolled in US pivotal trial of the VasQ device
The first patients have been enrolled in the US FDA pivotal study of VasQ (Laminate Medical Technologies), an implanted blood vessel external support device...
First four Spanish VasQ cases completed
Laminate Medical has announced the success of the first four Spanish clinical cases using the VasQ device.
The cases were performed by Gaspar Mestres at...
Laminate receives IDE approval from FDA to initiate study of the...
Laminate Medical Technologies, an Israeli biomedical start-up developing VasQ, an implanted blood vessel external support device for patients requiring arteriovenous fistula as vascular access for...
Laminate Medical Technologies completes US$8m Series B financing round
Laminate Medical Technologies has announced the close of a US$8m Series B financing round. This round of funding will help finance continued development and...
VasQ series at Berlin’s Charité University Hospital produces “exceptionally promising results”
As part of the main scientific programme at Dreiländertagung 2016 (4–8 October, Bern Switzerland), Peter Olschewski has reported “exceptionally promising results” from the first...