The German Institute for Hospital Remuneration (InEK) has renewed the new examination and treatment methods (NUB) Status 1 designation of VasQ external support (Laminate Medical Technologies) for 2021.
NUB Status 1 provides supplemental reimbursement for innovative medical devices that have the potential to improve the standard of care for patients. VasQ external support first received NUB Status 1 in 2017 with 35 participating hospitals and has since grown to 321 participating hospitals to date, which represents the second most among devices that serve the dialysis patient population.
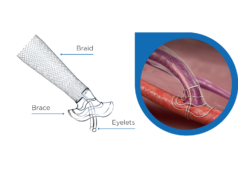
The VasQ external support is implanted around the anastomosis during the creation of an arteriovenous fistula (AVF) to promote more usability without the need for the frequent additional procedures typically required for standard AVFs. AVFs created with the VasQ external support have reported in publications and presentations usability rates as high as 86% or better by six months in seven clinical studies. Growing clinical evidence in support of VasQ as well as the building momentum of usage in InEK calculating hospitals are critical components to support a permanent diagnosis-related group (DRG) code for reimbursement.
“My practice relies on VasQ as our standard of care for creating almost all of our forearm and upper arm surgical fistulas,” stated Robert Shahverdyan, head of Vascular Access Center Hamburg at Asklepios Klinik Barmbek (Hamburg, Germany). “The device has improved my fistula outcomes for my patients, and I am grateful for the NUB designation so my work with VasQ can continue with sufficient reimbursement to cover the additional costs of the device.”
The device is currently being implanted across Europe under a CE mark and has also completed enrolment in an investigational device exemption (IDE) pivotal clinical study to be evaluated by the US Food and Drug Administration (FDA) through the de novo pathway for use in the USA. The FDA has already awarded the device a breakthrough designation based on the breadth of clinical evidence collected so far for VasQ that consistently demonstrates improvement over the standard of care for AVF creation. The designation provides VasQ with the substantiation of clinical improvement required by the Centers for Medicare and Medicaid Services for providing additional reimbursement for new technologies once cleared for the US market.