Tag: critical limb ischaemia
ESRD strongly associated with adverse limb events in new study
A new investigation into the association between end-stage renal disease (ESRD) and major adverse limb events (MALEs) has found that not only is it...
First-in-man use of the Tractus Crossing Support Catheter with its Jigsaw...
Tractus Vascular recently announced the first-in-man use of the Tractus Crossing Support Catheter (Tractus CSC). The Tractus CSC represents a highly novel approach to...
The CLI Global Society announces the Journal of Critical Limb Ischemia
The CLI Global Society is launching the first peer-reviewed academic journal focusing on interventional techniques pertaining to critical limb ischemia (CLI). The new journal,...
The CLI Global Society announces the Journal of Critical Limb Ischemia
The CLI Global Society is launching the first peer-reviewed academic journal focusing on interventional techniques pertaining to critical limb ischemia (CLI). The new journal,...
Rexgenero acquires the key technology and programme assets of aratinga.bio SAS...
Rexgenero, a regenerative medicine company developing advanced cell therapies to treat chronic limb-threatening ischaemia (CLTI), has announced the acquisition of all the key assets...
First US commercial use of Tack Endovascular System (4F) in BTK...
Intact Vascular has announced the first commercial use of its Tack Endovascular System (4F) in multiple sites across the USA. Notably the first FDA-approved...
First US commercial use of Tack Endovascular System in BTK arteries...
Intact Vascular has announced the first commercial use of its Tack Endovascular System (4F) in multiple sites across the USA. Notably the first FDA-approved...
Patient enrolment completed in MedAlliance PRESTIGE BTK clinical trial
MedAlliance has announced completion of patient enrolment in the PRESTIGE below-the-knee (BTK) clinical trial. The objective of this clinical investigation is to evaluate the...
PROMISE I trial affirms potential of LimFlow system for treatment of...
Initial findings of an early feasibility trial have demonstrated that the LimFlow System does represent a safe and reproducible method of treating patients with...
Full revascularisation “may not be the best solution” for CLI patients
Aiming for full revascularisation “may not be the best solution” for treating patients with critical limb ischaemia (CLI) according to Thomas Zeller (Bad Krozingen,...
More focus needed on best medical therapy and perfusion imaging for...
Curiously, the mortality risk for patients with critical limb ischaemia (CLI) has not changed over the last few decades, despite advances in cardiovascular medicine...
CLI: “Know the patient, know the disease and know what you...
Bruce Gray (Greenville, USA) talks to Vascular News after winning the LIVE (Leader In EndoVascular Education) award at VIVA 2019 (4–7 November, Las Vegas,...
ESVS 2019: HT-CLI trial to definitively test if cell technology can...
Bijan Modarai (London, UK) talks to BLearning Peripheral at ESVS 2019 (European Society for Vascular Surgery; 24–27 September; Hamburg, Germany) about the HT-CLI trial, which will look at the...
First patient enrolled in Cook Regentec’s clinical trial of investigational therapy...
Cook Regentec has announced enrolment of the first patient in an international clinical trial evaluating the HemaTrate Blood Filtration System to treat patients with...
Intraoperative oxygen monitoring predicts wound healing in critical limb ischaemia patients...
Preliminary findings from an initial study with 25 patients have shown that measuring the changes in oxygen concentration at baseline, during, and after endovascular...
The reactive and repetitive multidisciplinary CLI team
It is imperative that every medical institution has an interdisciplinary CLI team, argues Jihad Mustapha. Here, he exhorts the benefits of working in such...
Tack Endovascular System launches in the EU with first commercial use...
The first commercial use of the Tack Endovascular System (Intact Vascular) has taken place in multiple hospitals within Germany. A novel therapy for dissection...
First European patients treated with Peripheral Orbital atherectomy system
The first patients in Germany have been treated with the Stealth 360 Peripheral Orbital Atherectomy System (OAS; Cardiovascular Systems Inc). The German cases represent...
Lutonix 014 DCB gathers first randomised evidence of positive safety and...
Six-month outcomes of the Lutonix BTK IDE trial, a multicentre randomised controlled trial using the Lutonix 014 drug-coated balloon (DCB; BD), has demonstrated that...
First US implants of MicroStent for peripheral arterial disease take place
MicroStent (Micro Medical Solutions), a vascular stent designed to reduce below-the-knee amputations caused by critical limb ischaemia (CLI) resulting from peripheral arterial disease (PAD)...
Intraoperative oxygen monitoring predicts wound healing in critical limb ischaemia patients...
Preliminary findings from an initial study with 25 patients have shown that measuring the changes in oxygen concentration at baseline, during, and after endovascular...
BD announces completion of enrolment in Lutonix 014 DCB below-the-knee trial
Enrolment is complete in the Lutonix below-the-knee trial with BD planning to submit a pre-market approval application to the US Food and Drug Administration...
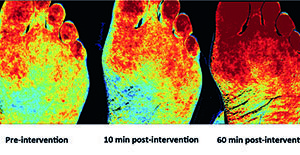
Hyperspectral imaging for assessing regional foot perfusion
There has been significant progress in limb salvage in patients with peripheral artery disease (PAD) and critical limb ischaemia (CLI) over the past two...
Positive results from pilot study of LimFlow Percutaneous Deep Vein Arterialization...
LimFlow announced on 1 August 2017 publication of positive results from the pilot study of the LimFlow Percutaneous Deep Vein Arterialization System (pDVA) in...
The search for modifiable risk factors to improve bypass results is...
A large study of the outcomes of 3,033 patients undergoing lower extremity bypass has reported a 10.6% infection rate and identified procedural risk factors....
Eyes may hold clues to future narrowing of leg vessels
Changes in tiny blood vessels of the eye may predict a higher risk of later narrowing in the large blood vessels in the legs,...
Medtronic launches European below-the-knee clinical study of drug-coated balloon
Medtronic has launched the IN.PACT BTK study to evaluate the effectiveness of using a drug-coated balloon in patients with below-the-knee (BTK) peripheral artery disease.
This...
Micro Medical Solutions receives CE mark for MicroStent
Micro Medical Solutions (MMS) has received CE mark approval for MicroStent, allowing the company to market this technology in Europe. MicroStent is a vascular...
TOBA II BTK clinical trial commences enrolment with first implant of...
Intact Vascular’s TOBA II BTK (Tack optimised balloon angioplasty II below the knee) clinical trial has commenced enrolment, with the first patient treated by Joseph Cardenas at Yuma Regional Hospital and...
BEST-CLI trial enrolment nears halfway mark
As the second year of enrolment draws to a close, the Best Endovascular vs. Best Surgical Therapy in Patients with Critical Limb Ischemia (BEST-CLI)...
Tack endovascular system secures CE mark for repair of dissections following...
Intact Vascular has attained the CE mark for the Tack endovascular system for the repair of arterial dissections following percutaneous transluminal angioplasty (PTA) below...
Intact Vascular closes on additional funding to advance development of Tack...
Current Intact Vascular investors—New Enterprise Associates, Quaker Partners and HIG BioHealth Partners—have exercised their right to invest additional capital as part of the company’s...
Spectranetics receives CE mark for Stellarex 0.014 drug-coated balloon
Spectranetics’ Stellarex 0.014” drug-coated angioplasty balloon has received the CE mark.
The Stellarex 0.014” device is designed to treat small vessels, below-the-knee disease, and challenging...
FDA approves TOBA II BTK pivotal IDE clinical study
The US Food and Drug Administration (FDA) has granted staged approval for Intact Vascular’s investigational device exemption (IDE) application to begin its TOBA II BTK...
Stentys expands benefits of drug-eluting, self-expanding stent to lower limb arteries
Clinical trial found Stentys treatment prevented amputation in 99% of cases.
The IN.PACT DEEP trial: Putting the results into context
By Krishna Rocha-Singh
How can we account for the lack of treatment effect of the IN.PACT Amphirion below-the-knee drug-eluting balloon (Medtronic) versus standard percutaneous transluminal...