Current Intact Vascular investors—New Enterprise Associates, Quaker Partners and HIG BioHealth Partners—have exercised their right to invest additional capital as part of the company’s Series B financing, bringing the total raised to US$46m after the financing initially closed in May 2015. The funds will be used to advance the company’s clinical development programme, encompassing both above-the-knee and below-the-knee clinical trials, as well as product development initiatives aimed at expanding the utility of the Tack endovascular system for the treatment of peripheral artery disease.
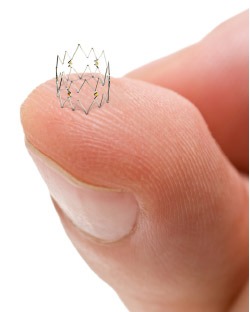
The Tack endovascular system is an endovascular technology used to repair arterial dissections following percutaneous transluminal angioplasty. Intact Vascular says that the Tack system permits “highly targeted repair of diseased arteries, while minimising the metal left behind and inflammation of the artery wall, both of which are significant problems associated with the current method of dissection repair, stenting.”
Intact Vascular is currently sponsoring three large, international clinical trials: Tack Optimized Balloon Angioplasty II (TOBA II), TOBA II BTK, and TOBA III. TOBA II and TOBA III are investigating the safety and efficacy of the Tack system in the treatment of above-the-knee peripheral artery disease in combination with both percutaneous transluminal angioplasty and drug-coated angioplasty balloons. TOBA II BTK is focused on use of the Tack system in conjunction with angioplasty to treat below-the-knee peripheral artery disease (also referred to as critical limb ischaemia). The TOBA II BTK trial is the first pivotal trial approved by the US Food and Drug Administration (FDA) for the treatment of critical limb ischaemia with a vascular implant below the knee.
“We welcome the additional investment from our investors and greatly appreciate their vote of confidence in us,” said Bruce Shook, Intact Vascular’s president and chief executive officer. “We are well positioned to use this new investment to rapidly advance our clinical and product development initiatives.”
“The team at Intact Vascular has made substantial progress since our initial investment,” stated Justin Klein, partner at New Enterprise Associates. “Intact is poised to bring a truly innovative endovascular technology to the peripheral vascular market place, and we are pleased to be a part of that important effort.”













