Tag: BIBA Briefings
BIBA Briefings: Critical limb-threatening ischaemia interventions buck trend for emergency only...
A BIBA MedTech Insights survey (conducted May 2020) indicates a trend towards centres only performing emergency percutaneous peripheral procedures during the COVID-19 pandemic. However,...
BIBA Briefings: Critical limb ischaemia interventions buck trend for emergency only...
A BIBA MedTech Insights survey (conducted May 2020) indicates a trend towards centres only performing emergency percutaneous peripheral procedures during the COVID-19 pandemic. However,...
BIBA Briefings: Survey indicates that elective aortic procedures could return to...
A new survey, conducted in May 2020, shows that about 65% of physicians in Western Europe believe their centres could be performing their normal...
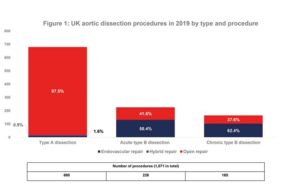
BIBA Briefings: Award for film raising awareness of aortic dissection
A short film (“Today is a Good Day”), which showcases the work of Aortic Dissection Awareness UK & Ireland, has won the Rare Disease Film Festival’s Peoples’...
BIBA Briefings: Medtronic receives US FDA Breakthrough Device Designation for complex...
The US Food and Drug Administration (FDA) has granted Breakthrough Device Designation to Medtronic for its novel Valiant thoracoabdominal aneurysm (TAAA) stent graft. A...
TCT 2019: System for managing deep vein reflux wins Shark Tank...
Source: CRF
At the 2019 Transcatheter Cardiovascular Therapeutics (TCT) meeting (25–29 September, San Francisco, USA), InterVene won the TCT-Shark Tank Innovation Competition for its novel endovenous system (BlueLeaf)...
TCT 2019: System for managing deep vein reflux wins Shark Tank...
At the 2019 Transcatheter Cardiovascular Therapeutics (TCT) meeting (25–29 September, San Francisco, USA), InterVene won the TCT-Shark Tank Innovation Competition for its novel endovenous system (BlueLeaf) for...
Private equity firm invests in UK centre performing EVLA for varicose...
A private equity firm, Palatine (Manchester, UK), has announced that it has made a “significant investment” into a UK centre—Veincentre—that provides endovenous laser ablation...
CE mark reinstated for Nellix endovascular aneurysm sealing system
Endologix has announced that the CE mark for the Nellix endovascular aneurysm sealing (EVAS) system has been reinstated. The CE mark for the system...