Data from the LOCOMOTIVE trial—a trial investigating the VascuFlex Multi-LOC multiple stent delivery system (MSDS; B Braun)—revealed that the strategy was safe and effective for focal stenting in long femoropopliteal lesions. The system achieved primary patency of 90.7% (n=68/75) and a 5.3% (n=4/75) all-cause target lesion revascularisation (TLR) rate of at six months. The data were published in VASA (Amendt K et al. Vasa. 2017 Aug 31:1-10).
LOCOMOTIVE is studying the MSDS strategy in de novo and restenotic lesions, with the exception of in-stent restenosis (ISR) or restenosis following drug-coated balloon (DCB) angioplasty.
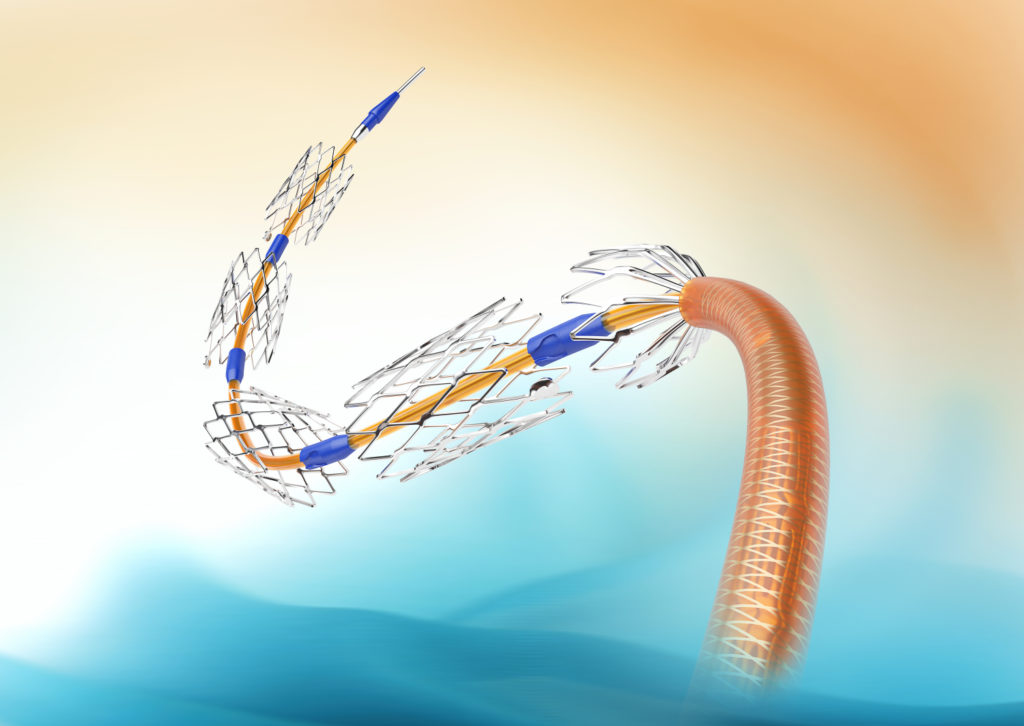
MSDS was indicated to treat flow limiting dissections or significant recoil (persisting stenosis >30%) after conventional balloon angioplasty and/or DCB dilatations in the superficial femoral artery and/or the popliteal segments (P1–P3).
The first-in-man LOCOMOTIVE study (ClinicalTrials.gov Identifier: NCT02531230) included 75 patients (mean age 72.9±9.2 years) in several German centres. The primary endpoint was defined as the all-cause TLR at six months. Femoropopliteal lesions were prepared with uncoated and/or paclitaxel-coated peripheral balloon catheters. When flow limiting dissections, elastic recoil or recoil due to calcification required stenting, up to six short stents per delivery device—each 13 mm in length—were implanted. Sonographic follow-ups and clinical assessments were scheduled at six months post-procedure.
The majority of the 176 individually-treated lesions were in the superficial femoral artery (76.2%, n=134/176) while the rate of TASC C/D lesions was 51.1% (n=90/176). The total lesion length was 14.5±9cm with reference vessel diameters of 5.6±0.7mm. Overall, 47±18% of lesion lengths could be saved with stenting. At six months, the primary patency rate was 90.7% (n=68/75) and the all-cause TLR rate was at 5.3% (n=4/75) in the overall cohort.
“The first clinical experience at six months suggests that the MSDS strategy was safe and effective in patients undergoing femoropopliteal revascularisations with TLR rates in critical and non-critical limb ischaemia patients of between 5% and 6% at six months,” write the authors, led by Klaus Amendt (Mannheim, Germany). “Moreover, the overall primary patency at six months was over 90% in morphologically challenging lesions and the procedural success rate to release the individual stent segments was 100%. Overall, almost half of the lesion lengths could be saved from stenting as compared to the full metal jacket strategy.”
The LOCOMOTIVE EXTENDED (ClinicalTrials.gov Identifier: NCT02900274) trial will confirm these early promising results in a larger patient cohort (>200 patients) in Europe and Asia.













