
An industrially produced stent graft for abdominal aortic aneurysm repair was first approved by the Japanese Ministry of Health, Labour and Welfare (MHLW) in Japan in July 2006, followed by a device for thoracic aortic aneurysm repair the next year.
The Japanese Society for Vascular Surgery (JSVS) acknowledged the necessity of prescribing certain standards for facilities and physicians involved in the usage of stent grafts for the purpose of safe and effective introduction of the new technology. The JSVS concluded that joint standards should be developed by a group of relevant academic associations, such as those related to vascular surgery, cardiology, interventional radiology, and endovascular therapy. It then requested the Japanese College of Angiology (JCA—a comprehensive academic association covering various relevant fields) to take charge of the preparation and management of the practice standards. In response to the JCA proposal, a committee to prepare the practice standards for stent grafting, consisting of representative members recommended by the 10 academic associations, came into being.
In relation to reimbursement for the stent graft system, the MHLW added a notice that the use of the device should be in compliance with the practice standards as a requirement related to the payments for medical materials. To achieve safe and high quality endovascular aneurysm repair (EVAR) and thoracic endovascular aneurysm repair (TEVAR), the Japanese Committee for Stentgraft Management (JACSM) was founded to cope with compliance of the practice standards of aortic stent grafting and promote follow-up surveys of all cases.
The JACSM was established with the aim of ensuring the safe and proper reach of commercial stent grafts following their regulatory approval. JACSM comprises 10 associations related to endovascular treatment. Based on the practice standards developed by JACSM, the status of practising institutions, practising surgeons, and supervising surgeons were determined by JACSM and the all results of follow-up surveys were registered.
After nine years, more than half of abdominal aortic aneurysms were treated with one of the five types of the devices (see graph). By August 2015, 534 institutions had met the practice standards. The number of practising surgeons has reached 1,192, and the number of supervising surgeons has reached 700. Practising institutions are obligated to report treatment results for individual cases in a case-registry system via the internet. There were 46,692 registered cases by August 2015.
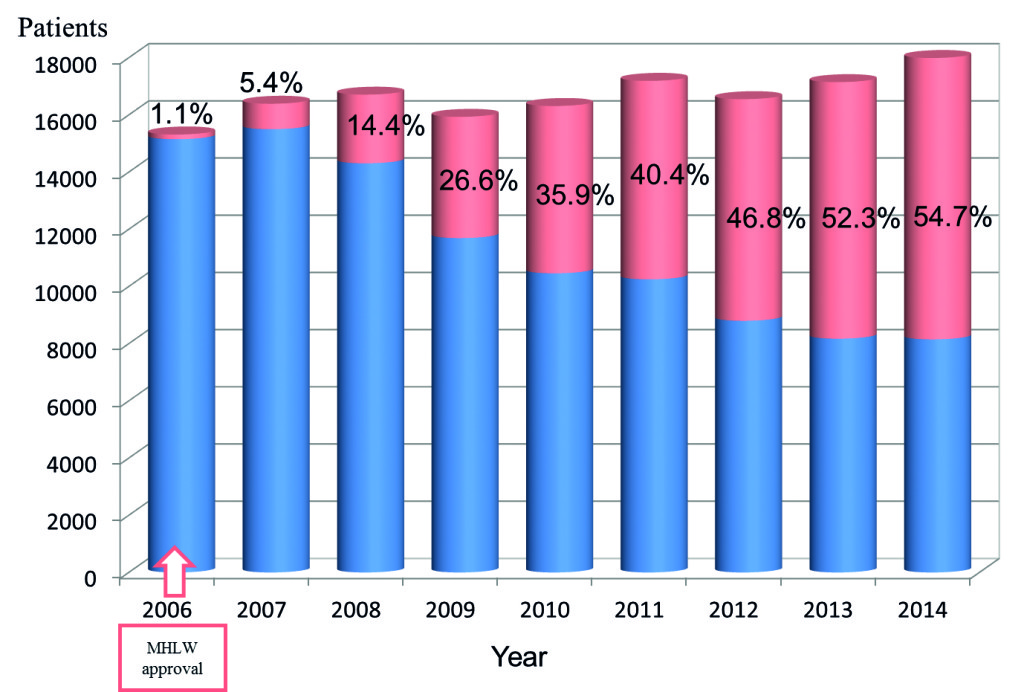
We are now reporting the outcomes of the 3,250 cases registered during the two years following approval of commercial stent grafts, from July 2006 to June 2008. During the same period, among the number of applications of corporate stents nationwide, EVAR was performed in 3,322 cases. Thus, the patient registration rate was 97%. The subjects included 3,209 of these patients for whom all data until hospital discharge were recorded (input rate 96%).
The patients included 2,762 males and 447 females, with a mean age of 75.5 years. The mean aneurysm diameter was 51.7mm. As for comorbidities, hypertension was 63.8% and coronary artery disease was 29.8%. The postoperative complications at the time of discharge are as follows: 19 cases (0.6%) of hospital mortality, 35 cases (1.1%) of type I endoleaks, 395 cases (12.7%) of type II, 25 cases (0.8%) of type III, two cases (0.1%) of type IV, and five cases (0.3%) that were unclear. There were no cases of migration at the time of discharge.
These excellent outcomes from the follow-up survey indicate the validity of the practice standards managed by JACSM.
Kimihiro Komori is at the Division of Vascular Surgery, Department of Surgery, Nagoya University Graduates School of Medicine, Nagoya, Japan, and is the chairman of the Japanese Committee for Stentgraft Management













