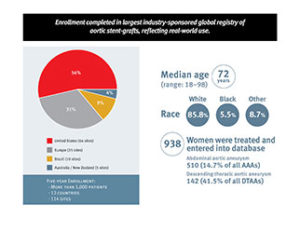
At the 2016 VEITHsymposium (15–19 November, New York, USA), Gore announced that the completion of target enrolment in GREAT (Global registry for endovascular aortic treatment). More than 5,000 patients have been enrolled into what is now, according to a press release, the largest industry-sponsored global registry of aortic stent-grafts. The press release reports that Gore undertook the effort in 2010 to evaluate how their family of aortic devices perform in real-world cases and to continue their commitment to advancing patient care in the treatment of aortic disease.
To ensure the registry is as reflective of real-world use as possible, Gore opened enrolment in the GREAT study to include patients with all pathologies of the aorta. This includes patients who received one or more of the Gore Excluder abdominal aortic aneurysm endoprosthesis, Gore Excluder lliac branch endoprosthesis, or conformable Gore Tag thoracic endoprosthesis.
The registry tracks outcomes during treatment and post-treatment over a 10-year follow-up period. In addition to procedural and clinical outcomes, data collected includes device(s) used, disease state treated, patient demographics, and medical history to measure outcomes in various scenarios. These data will also inform the continual evolvement of the Gore family of aortic stent-grafts for the future. The global lead investigators representing each region include Steven Dubenec, Pierre Galvagni, Ross Milner, Santi Trimarchi, Eric Verhoeven, and Fred Weaver.
Fred Weaver, co-national investigator, comments: “While clinical trials are very necessary to see if a device is appropriate and effective, often the results are not reflective of device use in daily practice. Through GREAT, we can take a look at a large number of patients with a broad set of symptoms and treatments to see how a device performs and make a better determination on proper device use in the future. This registry is truly real-world data.”
The press release notes that GREAT is part of Gore’s dedication to post-market surveillance and monitoring long-term device performance in an effort to improve clinical practice and patient outcomes. A team is dedicated to data analysis stemming from GREAT and ongoing follow-up. Additionally, the team is available to provide specific data upon request in an effort to aid physicians in making more informed treatment decisions for their patients.












