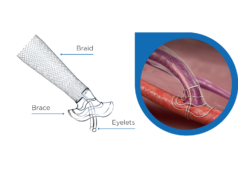
TVA Medical’s everlinQ 4 endovascular arteriovenous fistula (AVF) system has received CE mark in the European Union. The technology uses a 4F catheter system with enhanced visual indicators to create haemodialysis access using an endovascular technique without open surgery.
In a prospective, single-arm, single-centre study evaluating the everlinQ 4 endoAVF system, 97% of endovascular AVF procedures were successful, and fistula maturation was achieved in 83% of patients.
Tobias Steinke, chief of Vascular and Endovascular Surgery, Schön Klinik, Düsseldorf, Germany, performed the first case in Europe using the everlinQ 4 system. “The new system with smaller profile catheters and radiopaque visual indicators was extremely easy to use,” he said. “This new approach streamlines the procedure for providing patients a functional AV fistula without open surgery.”
“We are pleased to achieve this next regulatory milestone that broadens availability of the everlinQ endoAVF system for patients,” said Adam L Berman, president and CEO of TVA Medical. “We are now offering the 4F catheter system in Europe, as well as in the everlinQ endoAVF EU Study, which is gathering additional clinical data to support future clinician education, patient access, and reimbursement of the everlinQ endoAVF System.”