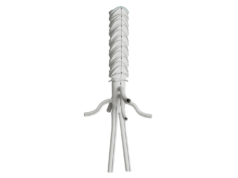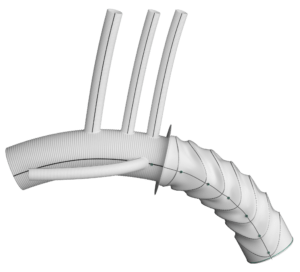
Terumo Aortic today announced that the US Centers for Medicare and Medicaid Services (CMS) has granted approval of a new technology add-on payment (NTAP) for Terumo Aortic’s Thoraflex Hybrid device under the inpatient prospective payment system (IPPS).
Thoraflex Hybrid is the first of its kind device used in frozen elephant trunk (FET) repair in the USA, according to Terumo Aortic. It was granted Breakthrough Device designation by the US Food and Drug Administration (FDA) in 2020 followed by FDA approval for commercial sale in the USA earlier this year. This hybrid device allows patients with suitably limited disease to be treated in a single stage procedure rather than two procedures which has previously been the conventional pathway in the USA for this group of patients.
The add-on payment will allow hospitals to be reimbursed for the incremental costs relating to the implantation of the Thoraflex Hybrid FET device to support the treatment of patients with complex aortic arch disease—this is in addition to the diagnosis related group (DRG) reimbursement.
The add-on payment is effective in CMS’ FY23 fiscal year, starting on 1 October 2022 and has been assigned the maximum new technology add-on payment—65% of the average cost of the technology, for a case involving the use of the Thoraflex Hybrid device.
Jeffrey Mifek, global vice president of clinical and medical affairs for Terumo Aortic commented: “We are delighted to have received this approval from CMS and it represents an important milestone for Terumo Aortic. The add-on payment provides further evidence that the Thoraflex Hybrid device significantly improves overall patient outcomes, compared to other available treatments.”
A company press release notes that Thoraflex Hybrid received CE mark approval in 2012 with more than 13,000 devices sold commercially around the world over the past 10 years.