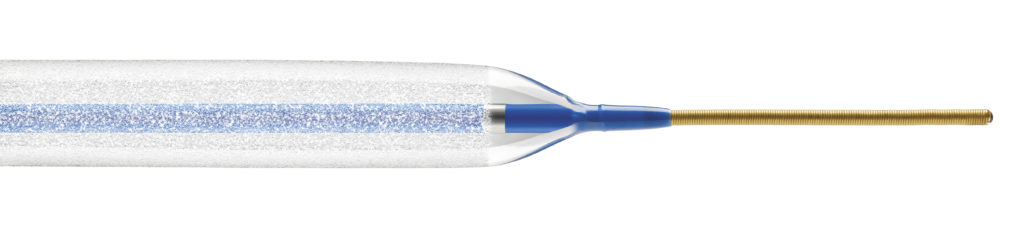
Spectranetics has submitted its pre-market approval application for the Stellarex drug-coated angioplasty balloon to the US Food and Drug Administration (FDA). Stellarex is designed to restore and maintain blood flow to the superficial femoral and popliteal arteries in patients with peripheral artery disease.
“The Stellarex drug-coated balloon has been studied extensively in two randomised controlled trials as part of the rigorous ILLUMENATE series that enrolled more than 1,100 patients,” said Amanda Johnson, vice president of Regulatory and Medical Affairs. “Key opinion leaders have called the results reported to date ‘remarkable,’ especially in light of Stellarex’s low drug concentration. This pre-market approval application marks a unique and important milestone in the future landscape of treating patients with peripheral artery disease.”
President and chief executive officer Scott Drake said, “Stellarex has been called a no-compromise solution with its low drug dosage and top-tier efficacy. The pre-market approval application embodies our commitment to leading the way in clinical science by providing proven treatment algorithms backed by robust clinical evidence. We are preparing diligently for our launch into the US market, which we anticipate in the second half of 2017, and are looking forward to improving patient care with this next-generation device.”
Spectranetics launched Stellarex in Europe in January 2015. Stellarex uses EnduraCoat technology, a durable, uniform coating designed to prevent drug loss during transit and facilitate controlled, efficient drug delivery to the treatment site. It is not for sale in the USA.













