Tag: Drug-coated balloon
LUMIFOLLOW registry shows sustained three-year performance of Luminor in femoropopliteal disease
iVascular has revealed the three-year follow-up results from the LUMIFOLLOW registry, a large real-world study evaluating the safety and efficacy of the Luminor drug-coated...
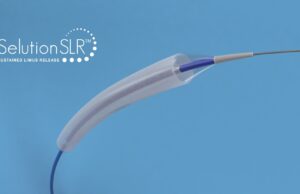
Cordis announces positive 24-month results from Selution SFA Japan trial
Cordis has announced positive 24-month results from the Selution SFA Japan trial. The prospective, multicentre, single-arm trial is designed to assess the safety and...
Enrolment completed in SAVE trial of Selution SLR for AVF treatment
MedAlliance has announced completion of patient enrolment in the SAVE clinical trial with the Selution Sustained Limus Release (SLR) 018 drug-eluting balloon (DEB) for...
Network meta-analysis finds DCB angioplasty “significantly superior” to plain balloon angioplasty...
A recent network meta-analysis shows that, in failing arteriovenous fistulas (AVFs) with outflow stenosis, drug-coated balloon (DCB) angioplasty is “significantly superior” to plain balloon...
Intermittent claudication: EffPac trial confirms benefit and safety of paclitaxel-coated balloon...
In the EffPac trial, designed to compare a drug-coated and an uncoated balloon in the treatment of vascular occlusion in the femoropopliteal region, the...
COMPARE RCT shows similar key outcomes between low- and high-dose drug-coated...
The first head-to-head randomised controlled trial to directly compare two different drug-coated balloons—the lower-dose density (2μg/mm2) Ranger and higher-dose density (3.5μg/mm2) IN.PACT—showed similar results...
Medtronic IN.PACT™ AV DCB, now approved for AV fistula maintenance in...
Watch Andrew Holden (Auckland, New Zealand) present the six-month results of the IN.PACT AV Access trial. The multicentre, prospective, randomised controlled trial shows that treatment...
Pooled analysis of four RCTs finds drug-coated balloons superior to plain...
A pooled analysis of four randomised controlled trials finds drug-coated balloon (DCB) angioplasty superior to plain angioplasty in patients with femoropopliteal artery disease, irrespective...
Lutonix BTK IDE: First Level 1 evidence of positive safety and...
Patrick Geraghty (St Louis, USA) presented new datapoints from the six-month outcomes of the Lutonix BTK IDE trial at the 2018 VEITHsymposium (13–17 November,...
Lutonix 014 DCB gathers first randomised evidence of positive safety and...
Six-month outcomes of the Lutonix BTK IDE trial, a multicentre randomised controlled trial using the Lutonix 014 drug-coated balloon (DCB; BD), has demonstrated that...
First US patient enrolled in ILLUMENATE BTK study of Stellarex 0.014...
The first US patient has been enrolled in the Stellarex ILLUMENATE below-the-knee (BTK) investigational device exemption (IDE) study, led by principal investigators Bill Gray...
Similar patency outcomes in women and men treated with Stellarex DCB
A study presented at VIVA 2018 (5–8 November, Las Vegas, USA) by Maureen Kohi, associate professor of Clinical Radiology, University of California, San Francisco,...
US$60 million raised to fund sirolimus-coated balloon investigational trial
Concept Medical Inc. has approached the FDA for an investigational device exemption (IDE) for their sirolimus-coated balloon (DCB). To support this process, they have...
Terumo gets CE mark for Kanshas drug-coated balloon catheter for lower...
Terumo has announced that it acquired CE mark for the Kanshas drug-coated balloon catheter used in the treatment of lower extremity peripheral arterial disease....
“Fantastic” live case demonstrates potential value of intravascular lithotripsy
In an exciting session at the Charing Cross Symposium (CX; 24-27 April, London, UK), the audience witnessed the demonstration of a novel treatment strategy...
CX 2018: Ulf Teichgräber discusses EffPAC trial 12-month results
Ulf Teichgräber, Jena, Germany, discusses the EffPAC trial 12-month results which were presented for the first time at CX 2018
New CMS reimbursement code “severely underpays” for drug-coated balloons
Drug-coated balloon (DCB) angioplasty devices have been categorised by the US Centers for Medicare and Medicaid Services (CMS) into the same billing code as...
Andrew Holden: Update on the IN.PACT AV Access trial
Andrew Holden spoke to Vascular News at LINC 2018 (Leipzig Interventional Course; 30 January–2 February, Leipzig, Germany) about the new data from the IN.PACT...
Surmodics partners with Abbott to develop their next-generation DCB
Abbott and Surmodics have announced an agreement whereby Abbott will have exclusive worldwide commercialisation rights for Surmodics' SurVeil drug-coated balloon (DCB) to treat the superficial...
First look at the interim LUTONIX DCB AV registry data
Vascular News interviews Panos Kitrou, Patras University Hospital, Rio, Greece, on the interim data from the LUTONIX DCB AV registry trial. The data were presented...
First-in-man SELUTION study achieves primary endpoint
Primary endpoint data from the first-in-man study of SELUTION, Med Alliance’s novel sirolimus-coated balloon, were presented at the Leipzig Interventional Course (LINC; 29 January–2...
LINC 2018 data highlight IN.PACT Admiral as safe and effective treatment...
Medtronic added to its body of clinical evidence supporting the IN.PACT Admiral drug-coated balloon with new presentations that demonstrated durable and consistent clinical outcomes...
“Promising” 12-month outcomes achieved with the Chocolate Touch drug-coated balloon
The final results of the ENDURE study, investigating the efficacy of the Chocolate Touch drug-coated balloon (QT Vascular), show “promising evidence of the drug...
BD announces completion of enrolment in Lutonix 014 DCB below-the-knee trial
Enrolment is complete in the Lutonix below-the-knee trial with BD planning to submit a pre-market approval application to the US Food and Drug Administration...
First-in-man study of Med Alliance’s Selution sirolimus-coated balloon completes enrolment
Med Alliance has announced the on-schedule completion of patient enrolment in the first-in-man (FIM) study of Selution, the company’s sirolimus-coated balloon. Fifty patients have...
Positive results for Boston Scientific Ranger paclitaxel-coated PTA balloon catheter
Positive results from the RANGER SFA trial for Boston Scientific’s Ranger paclitaxel-coated percutaneous transluminal angioplasty (PTA) balloon catheter have been presented at the 2017...
Medtronic launches European below-the-knee clinical study of drug-coated balloon
Medtronic has launched the IN.PACT BTK study to evaluate the effectiveness of using a drug-coated balloon in patients with below-the-knee (BTK) peripheral artery disease.
This...
Lutonix AV results provide tailwind for drug-coated balloons in dysfunctional arteriovenous...
Scott Trerotola presented the first release of eight-month data from the Lutonix AV trial at the Leipzig Interventional Course (LINC; 24–27 January, Leipzig, Germany)...
CONSEQUENT trial 12-month data support drug-coated balloons for long lesions
Twelve-month results of the CONSEQUENT trial have shown lower late lumen loss, lower rates of 12-month target lesion revascularisation and longer walking distance increases...
US FDA grants Medtronic investigational device exemption for IN.PACT Admiral AV...
Medtronic has received an investigational device exemption (IDE) from the US Food and Drug Administration (FDA) to initiate a study of the IN.PACT Admiral...
Results from Asia and Belgium demonstrate consistent results from Medtronic IN.PACT...
New data presented at the 2017 Leipzig Interventional Course (LINC) have demonstrated strong efficacy and consistent outcomes for Medtronic’s IN.PACT Admiral drug-coated balloon across...
Encouraging results for the Biotronik Passeo-18 Lux drug-coated balloon and Pulsar-18...
Interim data presented at the 2017 Leipzig Interventional Course (LINC; 24-27 January, Leipzig, Germany) has demonstrated encouraging results for Biotronik dedicated lower limb intervention portfolio.
“Currently...
US FDA grants QT Vascular full IDE approval for pivotal trial...
The US Food and Drug Administration (FDA) has granted QT Vascular full Investigational Device Exemption (IDE) approval to begin enrolling patients in the pivotal...
Stellarex ILLUMENATE 12-month pivotal results are “top-tier” in complex patients
Spectranetics has announced that the final 12-month results of the Stellarex drug-coated balloon ILLUMENATE pivotal trial have been presented at the Transcatheter Cardiovascular Therapeutics...
Spectranetics files FDA pre-market approval application for the Stellarex drug-coated balloon
Spectranetics has submitted its pre-market approval application for the Stellarex drug-coated angioplasty balloon to the US Food and Drug Administration (FDA). Stellarex is designed to restore...
Lutonix Global Real-World Registry 24-month outcomes presented at VIVA 2016
Bard has announced the presentation of the final 24-month results from the Lutonix Global Real-World Registry at the Vascular Intervention Advances (VIVA) 2016 meeting...
US FDA approves first drug-coated balloon for treatment of in-stent restenosis
The US Food and Drug Administration (FDA) has approved Medtronic’s IN.PACT Admiral drug-coated balloon as a treatment for in-stent restenosis in patients with peripheral...
UK NICE publishes Medtech Innovation Briefing on Lutonix
The UK National Institute for Health and Care Excellence (NICE) has developed a Medtech Innovation Briefing (MIB) on the Lutonix drug-coated balloon (DCB) for...
Medtronic receives FDA approval for IN.PACT Admiral DCB 150mm lengths
Medtronic has received US Food and Drug Administration (FDA) approval for the IN.PACT Admiral drug-coated balloon (DCB) in longer, 150mm lengths. The 150mm length...