Silk Road Medical has received US$47 million in new funding led by new investors Norwest Venture Partners and funds managed by Janus Capital Management LLC. Existing investors Warburg Pincus, The Vertical Group, and CRG also participated in the round. The new financing will be used to support the company’s rapid commercial growth of its proprietary ENROUTE family of products for transcarotid artery revascularisation (TCAR) procedures.
“Our syndicate of investors speaks to the global standard of care potential for TCAR and our commercial success to date. With this financing we will continue to build our commercial footprint and momentum in the USA and enter new markets abroad,” commented Erica Rogers, chief executive officer, Silk Road Medical. “We are delighted to have such strong financial support as we work to solve unmet needs through innovation, including other transcarotid applications of our intellectual property in acute ischaemic stroke and aortic valve disease.”
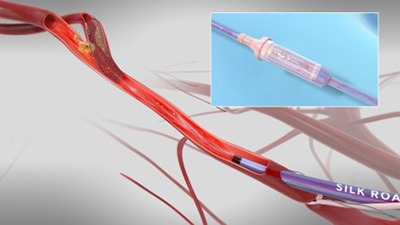
TCAR is a clinically proven procedure combining surgical principles of neuroprotection with minimally invasive endovascular techniques to treat blockages in the carotid artery at risk of causing a stroke. Silk Road’s ENROUTE neuroprotection system allows the physician to directly access the common carotid artery in the neck and initiate high rate temporary blood flow reversal to protect the brain from stroke while delivering and implanting Silk Road’s ENROUTE transcarotid stent. The ENROUTE system and stent are currently the only two devices cleared by the US Food and Drug Administration (FDA), approved and indicated for use in transcarotid procedures. To date, more than 2,000 TCAR procedures have been performed worldwide.
As part of the financing, Robert Mittendorff, a partner at Norwest Venture Partners, will join the company’s board of directors. Robert is a board certified emergency physician and former operating executive in the cardiovascular device industry, where he had the privilege to partner with leading vascular surgeons around the world in product development capacities.
“The team at Silk Road has created a category-defining medical procedure that has the potential to transform the treatment of carotid artery disease, which is a major cause of stroke,” said Mittendorff. “TCAR is an elegant, patient-friendly approach that has really resonated with physician leaders in the field. I have followed the company and its progress for years and have been pleased to see their rapid FDA approvals and favourable reimbursement decisions. I look forward to joining the board as we work to improve the treatment of this disease.”
“TCAR represents one of the next potential multibillion dollar medical device markets, and Silk Road is eponymous with the category with a sustainable advantage,” said Ethan Lovell of Janus Henderson Investors. “With 13 million ischaemic strokes occurring annually and carotid artery disease the culprit in up to one third of cases, the opportunity for TCAR is tremendous.”












