Medtronic and the University of Texas Health Science Center at Houston (UTHealth) have announced the first patient enrolled in a clinical study assessing the Medtronic Valiant Evo thoracic stent graft system for the minimally invasive repair of descending thoracic aortic aneurysms (TAA). The international, multicentre, prospective study will include 100 patients in the USA and Europe. The news was reported at the 2016 Vascular Annual Meeting of the Society for Vascular Surgery (8–11 June, National Harbor, USA).
Ali Azizzadeh, professor and chief of division of Vascular and Endovascular Surgery in the department of Cardiothoracic and Vascular Surgery at McGovern Medical School at UTHealth, is the US principal investigator for the trial and performed the first procedure on a patient with a descending TAA. Azizzadeh reports that the first patient treated in the investigational device exemption (IDE) trial is an 88-year-old male who is doing well post-treatment.
“The Valiant Evo clinical study has the potential to expand the applicability of thoracic endovascular aortic repair (TEVAR) to a wider spectrum of patients,” said Azizzadeh.
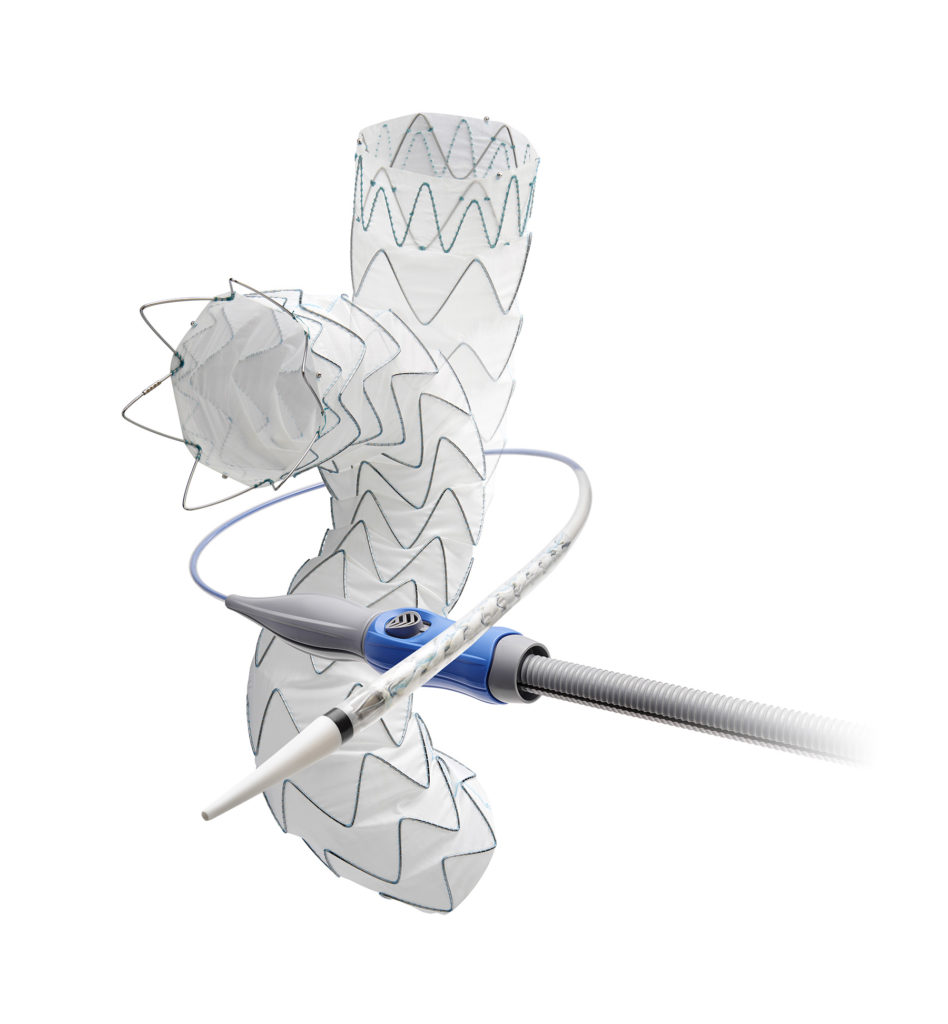
The Valiant Evo system is a lower profile evolution of the Valiant Captivia system, which has treated more than 85,000 patients globally. The Valiant Evo system is designed to deliver a minimally invasive treatment option to a greater number of patients with an 18F and 20F outer diameter profile for most size configurations, proximal closed web and FreeFlo configurations with tip capture, and high stent graft conformability. Additional features include broader tapered offerings and longer lengths for anatomical customization, in addition to an improved ergonomic delivery system. In the USA, the Valiant Evo is an investigational device and not yet approved for commercial use.
“With the Valiant Evo system, we are leveraging a decade of clinical experience and physician collaboration with the Valiant system, with a lower profile device that may help to treat more complex aortic disease,” said Daveen Chopra, vice president and general manager of the Aortic business, which is part of the Aortic & Peripheral Vascular division at Medtronic. “By listening to physicians we believe that this innovation may support greater access to less invasive treatment options for more patients globally.”