Tag: ReFlow Medical
Reflow Medical announces 12-month results from DEEPER REVEAL trial
Reflow Medical has announced that 12-month results from the DEEPER REVEAL clinical trial were presented for the first time at the Society of Interventional...
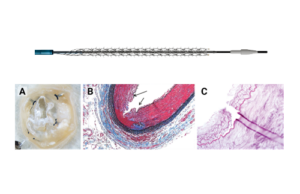
Spur RST: a novel approach to address limitations in BTK CLTI
This advertorial, sponsored by Reflow Medical, is only available in selected countries and geographies.
Addressing both recoil and impaired drug uptake remains central to...
US FDA grants de novo clearance for Reflow Medical’s Spur peripheral...
Reflow Medical recently announced that the US Food and Drug Administration (FDA) has granted de novo clearance for the company’s Spur peripheral retrievable stent...
Reflow Medical expands global reach with new European subsidiary
Reflow Medical has announced the opening of its European subsidiary in Landsberg am Lech, Germany. This strategic expansion strengthens the company’s international presence and...
Reflow Medical completes enrolment in the DEEPER REVEAL clinical trial
Reflow Medical has announced completion of enrolment in the DEEPER REVEAL clinical trial to evaluate the Reflow Spur stent.
The company notes in a...
Reflow Medical receives CE mark for Bare Temporary Spur stent system
Reflow Medical recently announced it has received CE mark certification in the European Union for the Bare Temporary Spur stent system. The device is...
Reflow Medical introduces Spex LP shapeable reinforced support catheter
Reflow Medical introduces the low-profile Spex LP 0.014 and 0.018-inch reinforced support catheters.
According to the company, the Spex LP is designed with a low-profile...
Reflow Medical receives approval in Japan for the Wingman catheter
Reflow Medical has announced that Japan’s Pharmaceuticals and Medical Devices Agency (PMDA) has approved the Wingman chronic total occlusion (CTO) catheter. Reflow Medical has...
FDA grants breakthrough device designation to Reflow Medical’s Temporary Spur stent...
Reflow Medical announces that the Temporary Spur stent system, a novel retrievable stent technology intended for the treatment of below-the-knee (BTK) peripheral arterial disease,...
VIVA 2019: Temporary Spur stent system appears to be feasible and...
Today at the 2019 Vascular Interventional Advances conference (VIVA; 4–7 November, Las Vegas, USA), Thomas Zeller (Bad Krozingen, Germany) gave an update on the...
Reflow Medical completes enrolment in the Wing-IT IDE CTO clinical trial
Reflow Medical has announced the completion of enrolment in its Wing-IT investigational device exemption (IDE) trial; a prospective, multicentre, nonrandomised study evaluating the ability...
DEEPER first-in-human results presented at VIVA
The first-in-human data from the DEEPER trial were presented at VIVA 2018 (5–8 November, Las Vegas, USA) by John Laird, Adventist Heart & Vascular...
Enrolment commences for Wing-IT IDE clinical trial of Wingman catheter
The first patients have been enrolled in a prospective, multicentre, non-randomised study intended to evaluate the ability of the Reflow Wingman Catheter (Reflow Medical)...