This advertorial is sponsored by Bentley.

There are numerous covered stents on the market that are used as bridging stents in fenestrated endovascular aneurysm repair (FEVAR). However, these stents have always been used off-label for such procedures. Now, a physician-initiated study, in cooperation with Bentley, is set to change that with the aim of evaluating the safety and efficacy of the BeGraft peripheral balloon expandable covered stent (Bentley InnoMed GmbH, Hechingen, Germany) used as a bridging stent in FEVAR, with the wider goal of extending its indication in order to offer a proven solution that physicians can rely on.
Eric Verhoeven, professor of vascular surgery at the General Hospital Nuremberg, Paracelsus Medical University in Nuremberg, Germany, is the principal investigator of the study and plans to enrol the first patient soon. In this article, Vascular News speaks to Verhoeven about FEVAR procedures, covering his expert opinion on the technique, key insights into what makes the ideal bridging stent, and what the FEVAR study means for the vascular community and patients alike.
What do you see as the main advantages of a FEVAR procedure over open surgery?
I work in a high-volume centre and in a recent publication based on our experience with FEVAR we had a mortality rate of 0.7%.1 This was a mixed group of patients with regard to surgical risk in which such a low mortality rate would never have been possible with open surgery. In our centre, FEVAR is routine and works very well, as we have the software, the hardware, the team, and the logistics to be able to perform such a complex procedure.
More generally, we offer this procedure when the anatomy is right and when we think there is a benefit for the patient. If you can perform a FEVAR so that the patient can go home three days later and you can stay out of the abdomen—which should not be opened unless it is really necessary—I would recommend it.
What can go wrong in a FEVAR procedure?
We are at the stage where next to nothing can go wrong anymore because we have the right devices. Occasionally, you cannot get into the target vessel, but I must say that this is extremely rare. In fact, I cannot recall one in the last 100 cases.
We have improved our skills over the years, we have better materials, and we have better options, for example with steerable sheaths or indwelling catheters, so it would be extremely rare to not be able to target the vessel and to stent it correctly.
Having said that, what can always occur is a dissection in the target vessel, because of difficult handling, and sometimes a wire perforation. This occurs in perhaps 1–2% of target vessels but demonstrates that these procedures are technically demanding and should be executed by a well-trained team. Overall, the patency rates of the target vessels are extremely high—in the mid-90%.
How essential is it to select the right bridging stent in FEVAR procedures?
Bridging stents are essential in a FEVAR procedure because we need a fairly strong stent that remains open, that can be flared, and that can be moulded. The BeGraft and the Advanta V12 (Getinge) are good examples of such a stent.
What are the specifications of an ideal stent for FEVAR and why are none of the available stents indicated yet?
The ideal bridging stent for FEVAR needs strength, radial force, and must be able to be flared and riveted in the fenestration. Eventually, it would be ideal if bridging stents for FEVAR become softer and smoother in their distal part, so that they could follow the path of the vessel better.
Bentley is the first company that was prepared to move forward by testing their stent grafts with the aim of getting them on-label for FEVAR procedures. While the FEVAR procedure has become standard care and the first option for many patients, it is still an off-label procedure, and that is why we are conducting a study, so that we can get this particular bridging stent graft— the BeGraft—on-label in FEVAR.
What do you like about the Bentley BeGraft stent and could it be considered an ideal bridging stent?
The BeGraft is a second-generation bridging stent and it is something that has been developed and adapted to the needs of physicians over the years. It has been redesigned so that the BeGraft is now stronger, thicker, and better overall. In addition, the BeGraft PLUS can be used for branches, which consists of two layers of expanded polytetrafluoroethylene (ePTFE) and two stents.
Besides the fact that the regular BeGraft has a high radial force, good handling and visibility, and that the flaring works well, the biggest advantage is the fact that it is 6F compatible, which is ideal if you have preloaded fenestrated grafts where you only have a 6F channel. In general, a 6F guiding sheath will track difficult anatomy better than a 7F guiding sheath, which is a huge advantage.
Finally, the BeGraft comes in many lengths and in short intervals, like 27/28mm. In the majority of cases, these are ideal lengths for a bridging stent in FEVAR, not too short and not too long.
Is there an option for the BeGraft PLUS to be used in FEVAR procedures?
You could use the BeGraft PLUS in FEVAR procedures if you had a very calcified vessel for which you need more radial force, which is a key feature of the BeGraft PLUS. This is not needed in 95% of the FEVAR target vessels. You could also use the BeGraft PLUS in FEVAR if you had a longer gap to bridge—like in BEVAR cases.
What do you expect from the FEVAR study?
This FEVAR study, which is approved by the German authorities BfArM, is a physician-initiated trial in cooperation with Bentley and the foundation for cardiovascular research and education (FCRE), and we are very grateful for their support because what we want to achieve is to get on-label approval for the bridging stents. Also involved are seven other investigators who work in experienced centres across Germany: Martin Austermann (St Franziskus-Hospital, Münster), Nikolaos Tsilimparis (Ludwig-Maximilians University Hospital, Munich), Kyriakos Oikonomou (University Hospital Regensburg, Regensburg), Drosos Kotelis (RWTH Aachen University, Aachen), Philipp Geisbüsch (Stuttgart Hospital, Stuttgart), and Johannes Kalder (University of Giessen, Giessen).
We need to include 100 patients and with that we should have data from at least 250 BeGrafts in fenestrations with a follow up of 24 months. We should be able to demonstrate its technical safety, durability, and that it is a favourable choice in FEVAR. The end goal is that the BeGraft will be the first on-label balloon expandable covered stent to be used in fenestrations.
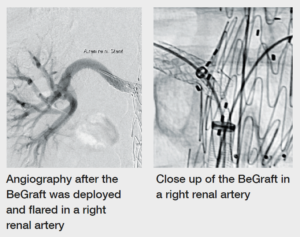
Case images
 References
References
- Verhoeven EL, Katsargyris A, Oikonomou K, et al. Fenestrated endovascular aortic aneurysm repair as a first line treatment option to treat short necked, juxtarenal and suprarenal aneurysms. Eur J Vasc Endovasc Surg 2016;51(6): 536–42.














Very nice to collect real world data ! It is a game changer in your everyday practice . Nevertheless gouvernement decision makers must be advised by very experienced doctors to be well informed about the meaning of those studies to avoid misinterpretation and loose new technologies en techniques for reimbursement !