 Remote monitoring of bypass flow using an implantable piezoelectric sensor, which can also be placed on stents, has shown successful and reliable transmission of signal over the first 30 days of an in vivo study. The experiment, which used the Graftworx system in an ovine carotid artery bypass model, raises the possibility of remote monitoring of vascular flow using wireless technology.
Remote monitoring of bypass flow using an implantable piezoelectric sensor, which can also be placed on stents, has shown successful and reliable transmission of signal over the first 30 days of an in vivo study. The experiment, which used the Graftworx system in an ovine carotid artery bypass model, raises the possibility of remote monitoring of vascular flow using wireless technology.
“We all know that when bypass grafts fail, we see an increase in the severity of symptoms, and claudication can convert into limb-threatening ischaemia,” said Richard Neville, Inova Health System, Falls Church, USA, during his presentation at the 2016 VEITHsymposium (15–19 November, New York, USA). Neville continued, “If a graft does become occluded, 75% of cases must be managed with open surgical techniques and reoperations are often more complex. By detecting a failing, or haemodynamically compromised graft, 75% of patients can be treated with endovascular techniques, hopefully preventing the more severe symptoms.”
Several methods are currently used to detect failing grafts. Clinical signs include return of symptoms or change in pulse exam or ankle brachial index, although these methods are episodic and often miss critical lesions. Duplex ultrasound has been used to identify failing grafts using B mode images and velocity measurements; when peak systolic velocity is greater than 300cm, velocity ratio is higher than 3 or the flow velocity in the graft is less than 40cm. In most practices, current standard clinical protocol is to conduct follow-up at three, six and 12 months post-procedure, and annually thereafter.
Neville detailed an early concept—tested in 1990 by Gupta et al—of a piezoelectric film (polyvinyl fluoride) sensor, attached to 6mm of PTFE, to monitor flow in a vascular graft. The concept was tested for iliofemoral bypasses in dogs, and achieved good correlation between sensor and flow meter. However, this study ran into problems with data transmission through skin, material and function deterioration over time and graft function.
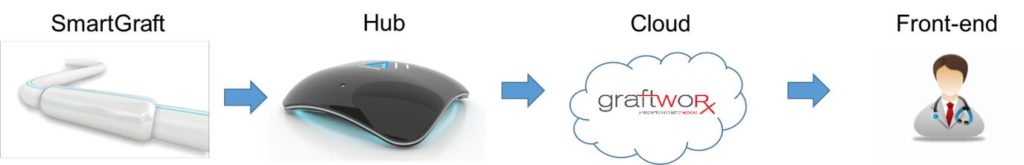
Inspired by this early concept, Neville and colleagues set out to overcome its initial problems using modern technology. In this case, the sensor was a piezoelectric sensor on a PTFE graft—a “SmartGraft”—which would convert mechanical flow forces to electrical transduction. A proprietary numerical parameter was then assigned based on a proprietary formula using phono-angiography frequency spectrum. Microchip Bluetooth technology was used for the wireless transmission of data, powered by a micro-battery. An external data processor was used for data analysis and reporting.

Initially, the concept was trialled in in vitro experiments to test the ability of the microsensor system to quantify and wirelessly transmit a flow signal with variable flow rates and record degrees of stenosis (Neville et al, J Vasc Surg 2016). Attached to the surface of ePTFE grafts, Neville reported that the system was “successful, with the device showing that it could quantify stenosis, its location, and its degree.”
The first in vivo experiment was designed to assess the system’s ability to transmit a signal per viable tissue under physiologic conditions, using an ovine carotid artery bypass model. At 30 days, an arteriogram of the sensor graft was conducted to compare with direct ultrasound flowmeter and a histopathology of graft sensor segment.
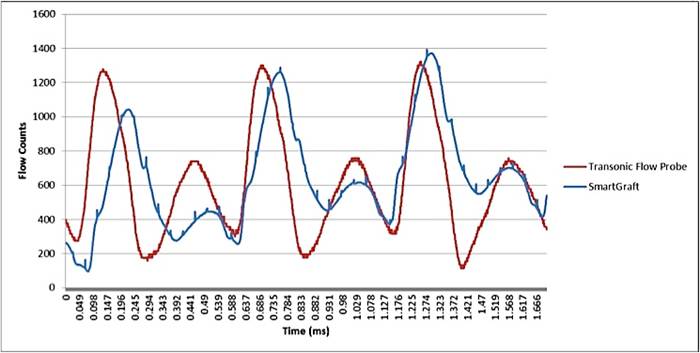
The system achieved “successful and reliable transmission of signal over 30 days,” Neville said, and all grafts remained patent. The histology found that the sensor achieved good apposition to PTFE, there was “minimal” tissue reaction adjacent to the sensor, and that there was a fibrous capsule with mild lymphohistiocytic inflammation. Measurement of flow was also found to be comparable to direct transonic flowmeter.
This success encouraged Neville and colleagues to pursue an advanced in vivo experiment, in which a self-monitoring arteriovenous graft was implanted in an ovine fistula model (carotid artery to jugular vein), transmitting sensor data to a hub three times a day, which was then uploaded to the cloud before being transferred to the remote monitoring system. The study outcomes were the success of automated monitoring of the graft function per tissue over time and the detection of stenosis and thrombosis. “Over three weeks, we could see the progression of stenosis from day one all the way to day seven. We could also see the occlusion, and the system detected an abnormality at eight hours and thrombosis at 24 hours.”
 “We believe that we have shown that remote monitoring of vascular flow data using wireless technology is feasible,” Neville told the audience. “Moving forward, we want to improve and expand data transmission to new areas,” he said. The first-in-man clinical trial for the system is planned for later this year, with an external remote monitoring device for arteriovenous fistulae. Neville and colleagues are also now researching the implementation of these systems on stent technology, he concluded.
“We believe that we have shown that remote monitoring of vascular flow data using wireless technology is feasible,” Neville told the audience. “Moving forward, we want to improve and expand data transmission to new areas,” he said. The first-in-man clinical trial for the system is planned for later this year, with an external remote monitoring device for arteriovenous fistulae. Neville and colleagues are also now researching the implementation of these systems on stent technology, he concluded.












