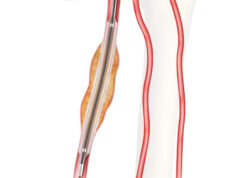
Cook Medical has launched the Zilver PTX drug-eluting peripheral stent thumbwheel delivery system in France, with the French Health Ministry now having approved reimbursement for the system.
A Cook Medical press release states that the rotating thumbwheel system, which is available for purchase in approved markets across Europe, provides deployment for the world’s first drug-eluting stent for the superficial femoral artery (SFA).
The thumbwheel design features include an ergonomic handle that facilitates single-handed deployment and is available with stents up to 120mm in length.
Zilver PTX received its initial CE mark in 2009 and US Food and Drug Administration approval in 2012. Five-year data have shown that the Zilver PTX reduces restenosis and reinterventions when compared to Zilver bare-metal stents, and that it inhibits neointimal hyperplasia while paclitaxel helps maintain patency by working to prevent restenosis.