A recent study revealed that endovascular repair of complex aortic aneurysms using fenestrated and branched endografts provides a safe and effective alternative to open surgical repair, with the lowest ever reported mortality rate in this patient population. This is according to researchers at The University of Texas Health Science Center at Houston (UTHealth Houston, Houston, USA) and Memorial Hermann Heart and Vascular Institute Aortic Center (Houston, USA).
The study, “Midterm outcomes of a prospective, nonrandomised study to evaluate endovascular repair of complex aortic aneurysms using fenestrated-branched endografts,” led by senior author Gustavo S Oderich (McGovern Medical School at UTHealth Houston), was published in the September 2021 Annals of Surgery.
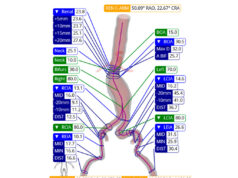
The study focused on treatment of complex aortic aneurysms, including pararenal and thoracoabdominal aortic aneurysms, which affect the aorta and the branches to the kidneys and intestines. These aneurysms are associated with high risk of rupture and death. While most aortic aneurysms are not associated with symptoms, if undiagnosed, progressive enlargement can lead to rupture with a high fatality rate.
The researchers reported that endovascular repair using fenestrated and branched endografts was associated with a 1% risk of mortality at 30 days, which compares to a mortality of 21% in a recent study of over 14,000 patients treated by open surgical repair in the USA. The improvement in mortality also compares favourably to mortality rates of open repair in large aortic centres, which range between 9–17%.
“When we began the study, we had no idea the rate of mortality would be so much lower than the historical results of open surgical repair,” Oderich said.
The study, which included 430 patients with an average age of 74, also showed better outcomes regarding major adverse events, including paralysis and paraplegia caused by spinal cord injury. Spinal cord injury affected 6% of patients enrolled in the fenestrated or branched endovascular aneurysm repair (F/BEVAR) study, which is similar to contemporary open surgical studies in which the rate of spinal cord injuries reported was 5–10%.
This research is supported by Cook Medical Inc and GE Healthcare, which provided funding for research co-ordinator support. The study is registered on ClinicalTrials.gov (study IDs NCT01937949 and NCT02089607).