This advertorial is sponsored by Shape Memory Medical.

Smart Polymer is a new shape memory polymer technology, available to endovascular specialists, that is incorporated into the Impede embolisation plug family (Shape Memory Medical). Andrew Holden, an interventional radiologist at Auckland City Hospital in Auckland, New Zealand, has been using the product for a number of years and has been involved in some early studies of the technology. Here, he gives his perspectives on the unique properties of the polymer, and details how Smart Polymer devices may be utilised in an interventional radiologist’s practice.
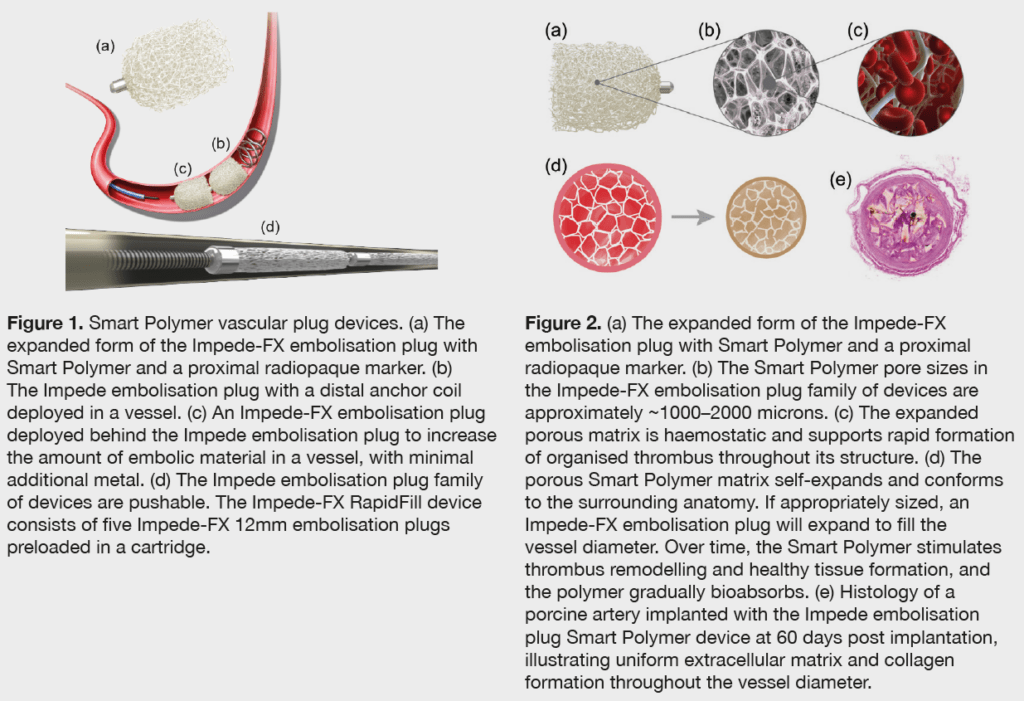
Smart Polymer is a porous scaffold polymer with high surface area. “What that means,” Holden tells Vascular News, “is that blood works its way through the scaffold in which long residence times promote acute thrombosis, so we see acute thrombosis within the scaffold.” What is more important though, he says, is the subsequent healing with mature collagen, followed by resorption of the scaffold. “The ability to produce acute thrombosis then vessel healing and resorption is a really important feature.” In addition, Holden highlights the fact that Shape Memory polymer technology can be used to fill blood spaces without chronic outward force. “At body temperature, the Shape Memory polymer expands to its premanufactured and uncrimped shape, but with a low chronic output force,” he explains.
According to Holden, there are a number of features that differentiate Shape Memory polymer technology from other embolisation devices. “A lot of embolisation devices, be they coils or plugs, do not produce acute occlusion. We may look to occlude a vessel, but find on follow-up that it remains patent, and that is one of the things we looked at very carefully in the first-in-human application for this device in peripheral vessels.”
Turning to the issue of landing zones for devices, Holden points out that the Shape Memory polymer stands out in this arena: “If, for example, a vessel is irregular or tapers, in some of the devices, landing is difficult, but Shape Memory by its ability to conform to the vessel is obviously well suited.” He also underlines the fact that, in peripheral applications, Shape Memory technology has a localisation coil to prevent migration of devices.
Holden details that his clinical experience with the technology has been encouraging so far. About five years ago, he and colleagues conducted the first-in-human trial with the Impede plug, and since then they have recorded accurate deployment, acute thrombosis, and no migration with the technology. In addition, they report having seen no evidence of recanalisation on follow-up imaging. “Our experience to date has left us feeling positive about the Impede’s role as an embolic,” Holden remarks. Holden adds that, in the last 12 months, he has been involved in the AAA-SHAPE first-in-human study, using the Impede device as an aneurysm sac filling technology to promote sac regression and prevent endoleaks and reintervention.
Considering the main indications for the Shape Memory polymer, Holden summarises: “Accurate deployment, conformability, acute occlusion, and a lack of recanalisation—all of those features mean that this technology lends itself ideally as a peripheral vascular embolic device, as well as its obvious interesting aneurysm sac filling application.”
Holden believes that an ideal first case in the peripheral vessels could be an aortic branch artery embolisation. “Although we can put standard plugs in these vessels, we know that we see an incidence of recanalisation or incomplete occlusion, and we rely on the fact that, on follow-up, we will see these plugs are actually occluding vessels.” With the Shape Memory technology, “we do get acute occlusions,” Holden communicates, “so we are confident that we will not see recanalisation”.
On the aneurysm side, Holden advises starting out on aneurysms with big blood volumes and large aortic side branches—the cases that are “most concerning”.
Holden details that the shape memory polymer has performed well in aneursym sac filling trial cases in his practice. “We have had cases where patients have had large aortic side branches and to date we have not had to do side branch embolisation as an ancillary procedure,” he recalls.
Taking into account his own clinical experience, early data, and planned future studies, Holden expresses a cautious optimism about this emerging technology. “It is being internationally recognised that while endovascular aneurysm repair (EVAR) is certainly minimally invasive, it has left us with significant problems. It is not the final product. The biggest issues I think are failure to protect patients from aneurysm sac progression or ongoing growth, but also failure to protect them from reintervention. I have believed for a long time that active sac management is something that is missing with EVAR, and I think this technology is very promising. However, I would stress that the results, while being positive in our first-in-human trials, are very early results, and we do need to see a larger body of results and bigger studies. Fortunately, those are being planned, so that is very exciting.”

Disclaimers: Andrew Holden is a consultant to Shape Memory Medical and principal investigator of the AAA-SHAPE_NZ trial.
Future device applications, prospective studies involving investigational devices, and investigational uses of approved devices are discussed in this interview. The content contains information about AAA-SHAPE, Shape Memory Medical’s prospective investigational studies of the Impede-FX embolisation plug and the Impede-FX RapidFill when used for prophylactic abdominal aortic aneurysm (AAA) sac filling during elective endovascular aneurysm repair (EVAR).
For more information about the AAA-SHAPE Studies, please visit https:/ / clinicaltrials.gov/ct2/home
NCT04227054 and NCT04751578
Not all devices discussed in this interview are available in all regions.
In countries recognising CE marking, the Impede Embolisation Plug, the Impede-FX embolisation plug, and Impede-FX RapidFill are indicated to obstruct or reduce the rate of blood flow in the peripheral vasculature.
In the USA, the Impede embolisation plug is indicated to obstruct or reduce the rate of blood flow in the peripheral vasculature and the Impede-FX embolisation plug is indicated for use with the Imped e embolisation plug to obstruct or reduce the rate of blood flow in the peripheral vasculature.
The Impede-FX RapidFill device is not approved for sale in the USA.