PQ Bypass has announced enrolment of the final subject in the company’s DETOUR II investigational device exemption (IDE) clinical trial. This milestone occurs only a month after the Detour system entered the US Food and Drug Administration (FDA)’s breakthrough device programme.
DETOUR II is led by national co-principal investigators Sean Lyden (Cleveland Clinic, Cleveland, USA) and Jihad Mustapha (Advanced Cardiac and Vascular Centers, Grand Rapids, USA). Both national co-PIs receive compensation for their duties in this role.
“The speed with which we were able to enroll DETOUR2 in 2020 speaks to the large patient population that exists with long-segment femoropopliteal disease that has sub-optimal endovascular options,” says Lyden.
“If percutaneous fem-pop bypass is shown to be safe and effective, similar to the outcomes demonstrated in DETOUR1 study, it could be a game changer for the way we treat complex, long-segment SFA [superficial femoral artery] disease today,” continues Mustapha.
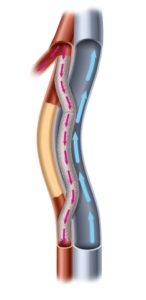
DETOUR II is a prospective, multicentre trial evaluating the Detour system for percutaneous femoral-popliteal bypass in patients with extremely long, complex lesions in the SFA. The study enrolled 202 patients in 36 sites in the USA and Europe, and is assessing freedom from major adverse events (MAE) within 30 days of the index procedure as the primary safety endpoint. The primary effectiveness is primary patency at 12 months.
“After finishing enrolment in DETOUR II and achieving the breakthrough device designation, PQ Bypass is on track to deliver results from this study much earlier than what we originally expected,” says Heather Simonsen, president of PQ Bypass. “We would like to thank the DETOUR II investigational sites for their ongoing contribution to this important research.”