According to a study presented at the 2014 European Society for Vascular Surgery (ESVS) meeting (23–25 September, Stockholm, Sweden), a standardised perioperative protocol for immediately diagnosing spinal cord ischaemia after thoracic endovascular aortic aneurysm repair (TEVAR) for the management of thoracoabdominal aortic aneurysms may lead to improved recovery after the complication.
Study presenter Nuno Dias (Vascular Center, Skånes University Hospital, Malmö, Sweden) reported that the incidence of spinal cord ischaemia after the repair of a thoracoabdominal aortic aneurysm remained “significant” (up to 31%) both for patients undergoing open repair and for those undergoing endovascular repair. He added that the complication could have “devastating consequences” for the patient, including reduced quality of life and an increased risk of death during the perioperative period. Therefore, Dias and his fellow investigators aimed to identify the incidence of, and potential risk factors for, spinal cord ischaemia after TEVAR in patients with a thoracoabdominal aortic aneurysm. They also sought to determine the value of using a standardised perioperative protocol for the immediate diagnosis of spinal cord ischaemia after TEVAR.
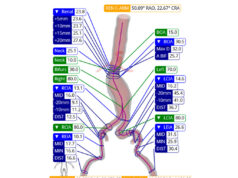
Dias explained that such a protocol has been used at his centre since March 2012 and involves patients undergoing neurological assessment (in the intensive care unit) every hour during the perioperative period after TEVAR. Patients who do not have symptoms of spinal cord ischaemia undergo passive cerebral spinal fluid drainage for 1.5–2 days, are haemodynamically controlled (mean arterial pressure >80mmHg), and their haemoglobin levels are normalised (target >100g/L). In patients who do have symptoms of spinal cord ischaemia, cerebral spinal fluid drainage is maximised (≤15ml over five days), perfusion pressure is increased (mean arterial pressure >90–100mmHg), and the approach to normalise haemoglobin levels is more aggressive (Hb >120g/L). Additionally, CT of the brain and/or spine is performed to exclude other causes of spinal cord ischaemia.
For the study, Dias et al reviewed retrospective data for 71 patients who underwent an elective (51) or acute (21; of whom, seven had a ruptured aneurysm) TEVAR for the management of a thoracoabdominal aortic aneurysm at the investigators’ centre between 2008 and 2013. Of these patients, the majority had a type II (36.1%) or type III (25%) Crawford aneurysm and 38 patients underwent TEVAR after the standardised perioperative protocol had been introduced (after March 2012). Patients received a branched and/or fenestrated endograft.
Overall, 21 patients (31%) developed spinal cord ischaemia after the TEVAR procedure. Dias noted that “surprisingly” there were no significant differences in the incidence of complication between the elective and acute patients (29.4% vs. 35%, respectively; p=0.776). However, he added that—according to a multivariate analysis—patients with a Crawford type II aneurysm (odds ratio 0.117; p=0.009) or those who received increased contrast volume during the TEVAR procedure (odds ratio 7.5; p=0.001) had an increased risk of developing spinal cord ischaemia. Furthermore, Dias commented that the incidence of the condition: “[it] seems to decrease with increasing experience” because it was reduced in patients who underwent TEVAR after 2012 (ie. when the operators would have had more experience) but not to a significant extent.
Thirteen patients in the group (33 overall) that underwent TEVAR prior to the introduction of the standardised spinal cord ischaemia perioperative protocol developed the complication compared with nine in the group (38 overall) that underwent TEVAR after its introduction (a non-significant difference). Whereas seven patients in the without-protocol group had no signs of recovery, all nine patients in the with-protocol group showed signs of recovery (five partial and four complete)—and this difference was significant (p=0.027). Dias stated: “More than 50% of patients in the with-protocol group became ambulatory and four of these were ambulatory without assistance but the vast majority of the without-protocol group did not become ambulatory. This did not lead to a significant difference between the groups but it is quite significant [regarding the protocol’s potential effect] in terms of quality of life.”
He concluded: “Recovery looks like it is enhanced quite significantly by an intensive protocol that is aiming at the immediate diagnosis of spinal cord ischaemia.”