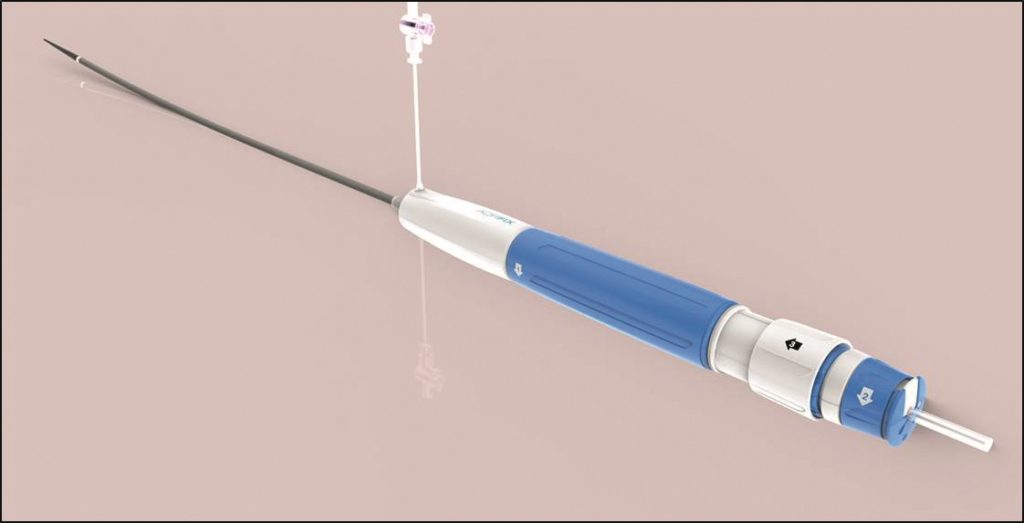
Lombard Medical has received CE mark approval for its IntelliFlex Low Profile (LP) delivery system for the Aorfix endovascular stent graft. This new system will be used for the delivery of the Aorfix endovascular stent graft, the only abdominal aortic aneurysm stent graft with global approval to treat aortic neck angulation up to 90 degrees. The company plans to begin shipments in July to its direct markets in the UK and Germany, and expects a full commercial launch in the other European markets later in the year.
The IntelliFlex LP delivery system features a low profile, intuitive, compact and ergonomic design that enhances the precision and control of Aorfix deployment and placement. The system also incorporates an integrated exchange sheath to facilitate the delivery of ancillary devices during the endovascular procedure. This feature avoids the need for catheter exchanges, potentially reducing procedure time, blood loss and vessel trauma. Andrew Holden, director of Interventional Services and associate professor of radiology at Auckland University School of Medicine, and Andrew Hill, of Auckland City Hospital in New Zealand completed a successful first-in-man use of the IntelliFlex LP to implant an Aorfix endovascular stent graft in November 2015.
“The first-in-man procedure with the new IntelliFlex delivery system was extremely straightforward and the device was a real pleasure to use,” said Holden. “I particularly appreciated the new Y-mechanism that enables greater control and precision when deploying the graft. This enhanced design is a significant advancement for the Aorfix system and the device can now be used even more reliably in both routine and challenging anatomies.”
Hill added, “The new IntelliFlex delivery system provides greater control of the Aorfix stent graft when deploying the proximal end of the device near the renal arteries, allowing for very accurate placement. The integrated exchange sheath was really beneficial and the haemostatic valve copes extremely well with multiple catheter exchanges.”
“The procedure advantages of the IntelliFlex LP together with the outstanding long-term clinical results of Aorfix will provide physicians with an even more compelling reason to use this technology to treat an even greater range of patient anatomies,” said chief executive officer Simon Hubbert. “We believe the combination of Aorfix with IntelliFlex LP and our new Altura stent graft which delivers multiple advantages for the treatment of patients with more routine abdominal aortic aneurysm anatomy will be a significant driver of revenue growth in 2016 and beyond. We look forward to our European commercial launch of IntelliFlex in July.”













