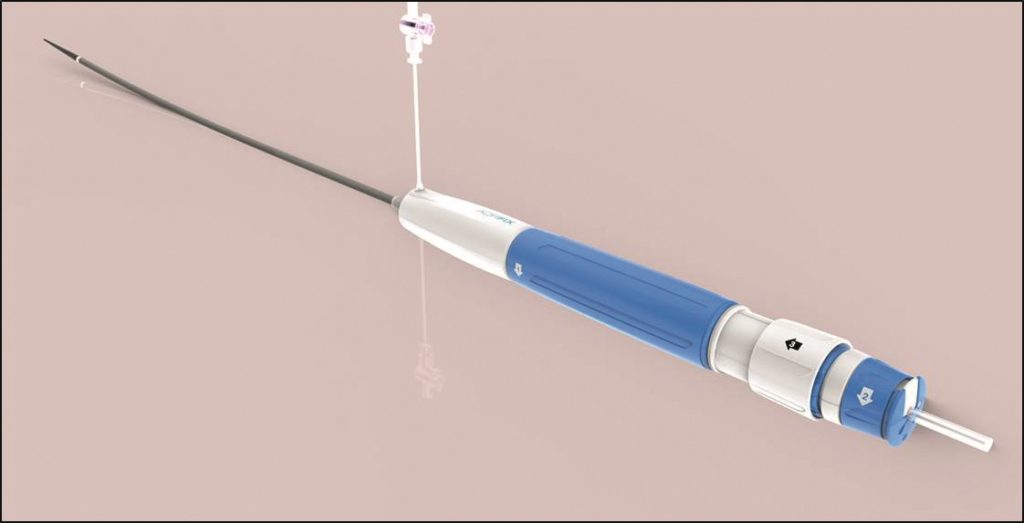
Lombard Medical has announced the full commercial launch in Japan of its new low-profile IntelliFlex LP delivery system for the Aorfix abdominal aortic aneuryism (AAA) system.
The new delivery system, used to deliver Lombard’s Aorfix AAA stent graft, is exclusively marketed in Japan by Osaka-based Medico’s Hirata, a supplier of medical devices for the Japanese healthcare industry.
Clinical cases confirming the performance and ease-of-use of the IntelliFlex LP delivery catheter in complex aortic aneurysm anatomies were presented at this year’s Japanese Endovascular Symposium (23–24 August; Tokyo, Japan). Makoto Sumi, of Saitama Cardiovascular and Respiratory Center for Vascular Surgery, Saitama, Japan, presented the data.
“After a carefully controlled release of Lombard’s new delivery catheter for Aorfix, positive first-hand customer experience is increasing demand in our domestic market. We are confident that the investment made by Lombard in the technology—and our firm in sales and marketing—will pay dividends in terms of increased sales for the balance of 2017 and beyond,” says Masataka Hirata, president of Medico’s Hirata. “In addition, early data from the Medico’s-sponsored Japanese clinical study (JANIS) recently presented at the Japanese Society for Vascular Surgery independently confirmed the excellent clinical data in the clinical study which led to Lombard’s US Food and Drug (FDA) approval (PYTHAGORAS).”
Details on the PYTHAGORAS US clinical study can be found in the July edition of the Journal of Vascular Surgery.
Kurt Lemvigh, chief executive officer of Lombard, comments, “Japan is perhaps the most demanding market in the world in terms of quality of products and clinical outcomes…by focusing on our UK home market and the fast-growing AAA markets in Japan and China, we believe we can achieve cash flow breakeven and profitability on rather modest revenue. The operational turnaround at Lombard is tangible.”













