 Mercator MedSystems has announced the first patient enrolment into the TANGO (Temsirolimus Adventitial Delivery to Improve Angiographic Outcomes Below the Knee) clinical trial. TANGO will study the effects of using Mercator’s proprietary Bullfrog micro-infusion device for the adventitial delivery of the drug temsirolimus (Torisel, Pfizer) after revascularisation of lesions below the knee in patients with critical limb ischaemia.
Mercator MedSystems has announced the first patient enrolment into the TANGO (Temsirolimus Adventitial Delivery to Improve Angiographic Outcomes Below the Knee) clinical trial. TANGO will study the effects of using Mercator’s proprietary Bullfrog micro-infusion device for the adventitial delivery of the drug temsirolimus (Torisel, Pfizer) after revascularisation of lesions below the knee in patients with critical limb ischaemia.
TANGO is the fourth clinical trial ongoing with the Bullfrog device in below-the-knee (the third in the USA). Outside of trials using the Bullfrog device for drug delivery, only one intravascular drug delivery technology (a paclitaxel-coated angioplasty balloon) is being investigated in a single US below-the-knee study.
“This is the first clinical trial of its kind. While studies have been done outside the USA with -limus-eluting stents in focal lesions in the legs, the local delivery of a -limus agent without a permanent implant and in lesions longer than 5cm (2 inches) has not yet been studied in the US,” says Ian Cawich, Principal Investigator for TANGO and an interventional cardiologist at the Arkansas Heart Hospital. “We are thrilled to be a part of the TANGO study and to be at the forefront of utilising this novel approach to address the tremendous medical need for an effective critical limb ischaemia treatment.”
TANGO is a prospective, multicentre, randomised, dose-escalation trial recruiting approximately 60 patients who have critical limb ischaemia related to arterial obstructions in below-the-knee arteries. In these patients, temsirolimus will be delivered into the tissue immediately around the arterial wall after balloon angioplasty or mechanical atherectomy is used to open the obstructions.
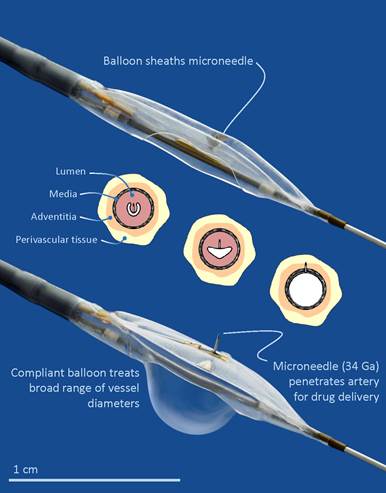
Temsirolimus belongs to a family of well-known agents (the -limus analogs) that are commonly delivered by coronary drug-eluting stents following revascularisation of lesions. The use of Mercator’s Bullfrog delivery system in the TANGO study is intended to allow the local delivery of this agent in below-the-knee lesions, but eliminate the need to leave behind an implant in the body. While the small and tortuous anatomy of the below-the-knee vessels can make navigation and drug delivery from a coated balloon difficult, the small profile of the Bullfrog device is designed to allow it to adjust well to small vessels. According to a company release, it is able to deliver drug volumes that are independent of vessel size.













