Tag: below the knee
SUCCESS PTA stratified analysis finds equal benefit for sirolimus DEB above...
Positive 12-month outcomes for the Selution Sustained Limus Release (SLR; Cordis) drug-eluting balloon (DEB) were observed, with consistent haemodynamic, functional and clinical improvements irrespective...
RECOIL study: Serranator demonstrates 49% less recoil than plain balloon angioplasty...
Cagent Vascular has announced the results of its below-the-knee (BTK) RECOIL study. This core lab-adjudicated Recoil analysis— the first of its kind, according to...
Surmodics receives 510(k) clearance for Pounce LP thrombectomy system
Surmodics announced in a press release that it has received US Food and Drug Administration (FDA) 510(k) clearance for its Pounce LP (low...
Pedal artery revascularisation: Is it ready for prime time?
Reviewing the available evidence for below-the-ankle interventions in the treatment of critical limb-threatening ischaemia (CLTI), Srini Tummala proposes that pedal artery intervention “should be...
Lutonix 014 DCB gathers first randomised evidence of positive safety and...
Six-month outcomes of the Lutonix BTK IDE trial, a multicentre randomised controlled trial using the Lutonix 014 drug-coated balloon (DCB; BD), has demonstrated that...
First-ever CE mark of a bioresorbable scaffold for below-the-knee PAD
Reva Medical, a company developing bioresorbable polymer technologies for vascular applications, has announced that its Motiv bioresorbable scaffold is the first drug-eluting bioresorbable scaffold...
Angiolite BTK below-the-knee sirolimus-eluting stent receives CE mark approval
iVascular has announced the release of Angiolite BTK, a sirolimus-eluting stent which has received CE mark approval for treating chronic and acute arterial lesions in...
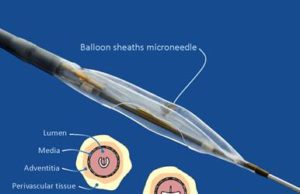
First patients enrolled in Mercator MedSystems TANGO trial
Mercator MedSystems has announced the first patient enrolment into the TANGO (Temsirolimus Adventitial Delivery to Improve Angiographic Outcomes Below the Knee) clinical trial. TANGO...
Medtronic launches European below-the-knee clinical study of drug-coated balloon
Medtronic has launched the IN.PACT BTK study to evaluate the effectiveness of using a drug-coated balloon in patients with below-the-knee (BTK) peripheral artery disease.
This...
North Dallas Research Associates begin enrolling TOBA II BTK patients
North Dallas Research Associates, Dallas, Texas, and its private practice, Cardiac Center of Texas, have announced their participation in the Tack Optimized Balloon Angioplasty II...
Tack endovascular system secures CE mark for repair of dissections following...
Intact Vascular has attained the CE mark for the Tack endovascular system for the repair of arterial dissections following percutaneous transluminal angioplasty (PTA) below...