Essential Medical has announced the publication of the MANTA CE marking study results in JACC Interventions titled: “Percutaneous plug-based arteriotomy closure device for large-bore access”.
This first, prospective, peer reviewed scientific evaluation of vascular access complications after large bore cardiovascular procedures demonstrated extremely low rates of vascular access and bleeding complications with the Manta closure device. Fifty patients were treated resulting in one VARC-2 major complication and zero VARC-2 minor complications. The mean and median time-to-haemostasis was 144 seconds and 24 seconds, respectively with 74% of patients having haemostasis in <60 seconds. The authors noted that the results compared extremely favourably to outcomes observed with currently used methods.
William W O’Neill, chairman of the Scientific Advisory Board at Essential Medical and medical director of the Center for Structural Heart Disease at Henry Ford Hospital, Detroit, USA, commented: “The device is extremely easy to use compared to current methods. The results of this study and real-world registry data emerging from commercialisation in Europe are extraordinarily encouraging.”
Closure of large bore femoral access sites has been associated with significant morbidity including higher rate of complications and higher total cost of care. A recent JAMA Cardiology publication demonstrated the need for a reliable large bore closure device to reduce the current 17.7% bleeding complication seen and unsettling costs. Compared to median total health care costs of US$29,968 for patients without bleeding complications, median total health care costs were US$48,663 for patients with bleeding complications. As demonstrated in the European study data, Manta’s fail-safe deployment provides rapid and reliable haemostasis with low complication rates, with potential to minimise these aforementioned bleeding complication implications.
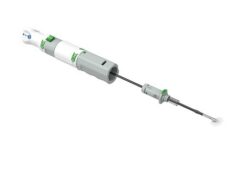
Manta is a novel CE marked vascular closure device that is commercially available in the Netherlands. It is designed to close punctures ranging from 10–25F at femoral arterial access sites after cardiac catheterisation procedures such as transcatheter aortic valve replacement, endovascular treatment of abdominal aortic aneurysms, ventricular assist, and balloon aortic valvuloplasty. An IDE study is currently underway in the USA.