 Fusion imaging can be used for infrarenal endovascular aneurysm repair (EVAR) following a short learning curve with the benefit of lower radiation dose and shorter procedure times, according to a presentation giving at the 2017 Vascular Annual Meeting (VAM; 31 May–3 June, San Diego, USA).
Fusion imaging can be used for infrarenal endovascular aneurysm repair (EVAR) following a short learning curve with the benefit of lower radiation dose and shorter procedure times, according to a presentation giving at the 2017 Vascular Annual Meeting (VAM; 31 May–3 June, San Diego, USA).
Debra Chong, who presented the data on behalf of Blandine Maurel and the research team at the Royal Free Hospital, London, UK, noted that the benefits of fusion imaging have already been proven in complex EVAR, reducing radiation dose for patients and operating room staff, the contrast volume needed, and the length of the procedure. Chong and colleagues’ research sought to discover if these benefits could be extended to infrarenal EVAR.
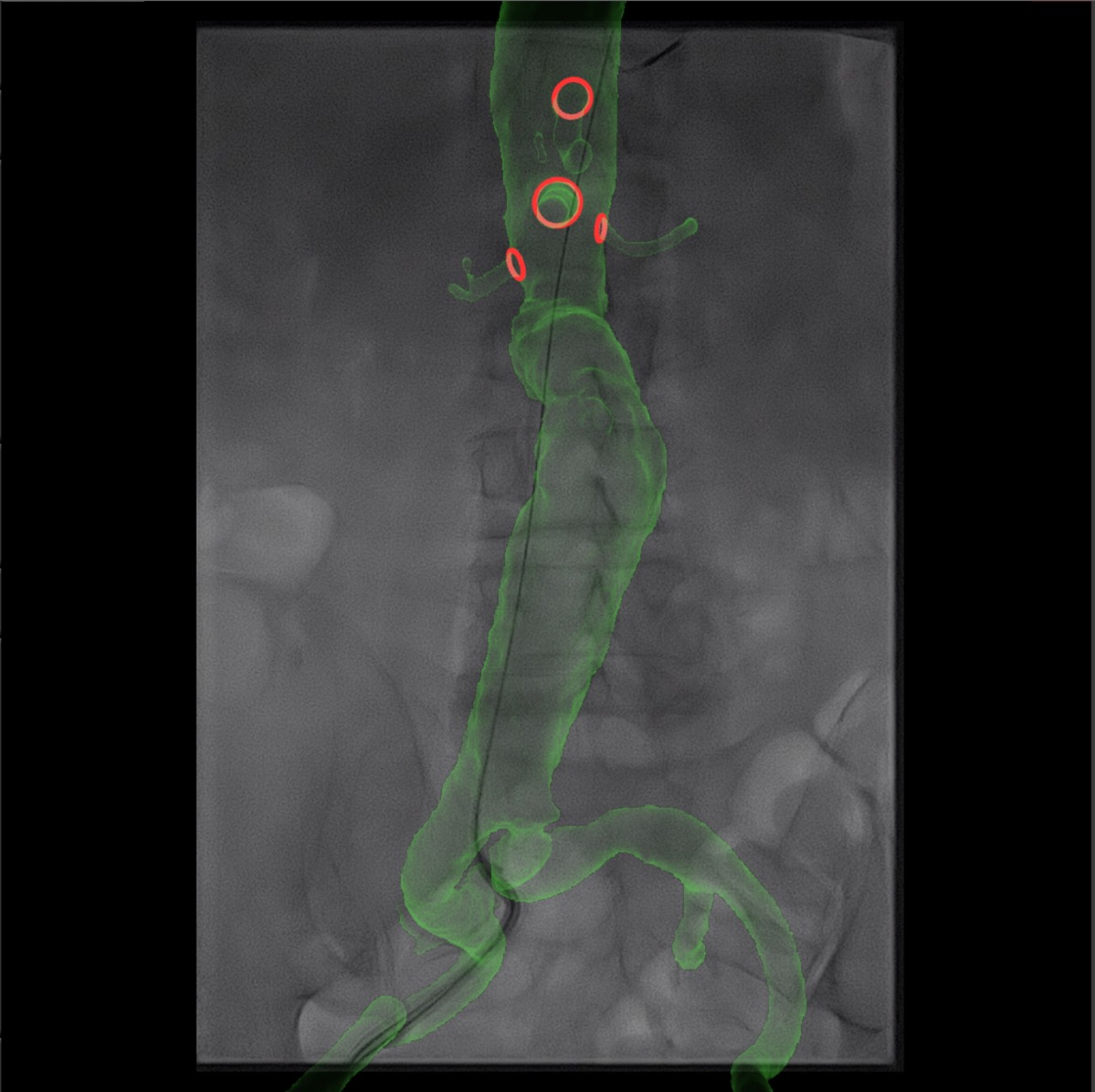
The system used in their study was the CYDAR imaging guidance system (Cydar Medical), cloud-based software which deduces patient position by automatically comparing the vertebrae visible on real-time X-ray to those on computed tomography angiography. Importantly, this software is suitable for use with a mobile C-arm or mobile flat panel detector, making it adaptable to any operating theatre. CYDAR imaging covers eight vertebrae (T10–L5) in a range of angles (60 degrees laterally and 30 degrees cranio-caudally). The system takes five seconds to detect the vertebrae and match them with an aortic overlay.
This prospective, non-randomised, single-centre trial ran from December 2014 to April 2017, enrolling 76 patients. The CYDAR group was comprised of 44 patients who underwent infrarenal EVAR in a hybrid room or mobile C-arm with CYDAR between March 2016 and April 2017. These patients were compared with those in the control group (n=32) who underwent infrarenal EVAR in a hybrid room without fusion between December 2014 and March 2016.
Before procedures were undertaken, CYDAR group operators learned how to use the system and attended teaching sessions on radiation protection, which explained the ALARA (As low as reasonably achievable) principle.
Patients were excluded if they had additional procedures, ruptured aneurysms, or were classed as emergency procedures. The groups were demographically well matched.
Chong and colleagues’ results showed evidence of a surgeon learning curve with CYDAR imaging, for both wire-to-wire operating time and number of digital subtraction angiography runs.
The median wire-to-wire operation time and fluoroscopy time were 90 minutes (range 72.5–105 minutes) and 29 minutes (range 22–35.5 minutes) respectively, with no significant differences between the groups.
Radiation dose—measured by air kerma (AK) and dose area product (DAP)—was significantly lower for the CYDAR group than the control group. The investigators reported that AK product was significantly higher in the control group (142mGy [range 63–565.5mGy]) compared with 80mGy (range 51–109mGy) in the Cydar group (p=0.028). The DAP in the control group was 2,173µGym2 (range 944.5–8,794µGym2) compared with 1,178.5µGym2 in the Cydar group (range 689–2,172µGym2; p=0.08). No significant differences between the two groups were reported for rates of adverse events, endoleaks, or additional procedures required.
To gather further feedback on the system, the CYDAR users were surveyed after the procedures. When asked if the CYDAR overlay was accurate enough at the renal level to deploy the stent graft, 48% (n=21) said that it was, and 50% (n=22) said it was not. The vast majority (95%, n=42) believed that the procedure was easier when using CYDAR, with just two responders (5%) saying that it was not.
“Fusion imaging can be used for infrarenal EVAR with a short learning curve,” Chong concluded. “The benefits of CYDAR fusion imaging are similar to those in complex EVAR cases, specifically the lower radiation dose and shorter procedure time. It should be remembered that deformation of the aorta due to tortuosity may impact the accuracy of the overlay.”













