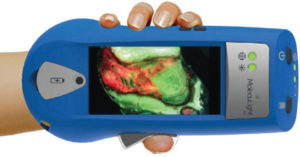
Results from the Fluorescence Imaging Assessment and Guidance (FLAAG) clinical trial, published recently in Advances in Wound Care, reveal that 82% of wounds in enrolled patients—which included diabetic foot ulcers, venous leg ulcers, pressure ulcers, and other chronic wounds—had a high bacterial burden (>104 CFU/g). Use of the MolecuLight i:X fluorescence imaging device resulted in detection of 45% more wounds with high bacterial loads that would otherwise have been missed by standard of care assessment of clinical signs and symptoms; these results were consistent across all wound types, study sites, and clinicians.
This large prospective, multicentre, controlled clinical trial included 350 patients, across 14 US outpatient wound care centres and 20 clinicians. The trial, independently led by Thomas E Serena and his team at the SerenaGroup, evaluated whether fluorescence imaging (MolecuLight i:X) improves detection of high bacterial loads when used in combination with clinical signs and symptoms of assessment. The trial also examined how point-of-care information on bacterial presence and location impacted clinical treatment planning.
The objective, diagnostic information on bacterial load provided by fluorescence imaging changed clinical treatment planning in 69% of wounds and influenced wound bed preparation in 85% of study wounds. Most importantly, clinicians reported that use of fluorescence imaging improved patient care in 90% of study wounds.
“The FLAAG trial, one of the largest prospective clinical trials in wound care, demonstrated that clinical signs and symptoms are poor indicators of bacterial burden in chronic wounds”, says Serena, founder and medical director of The SerenaGroup and lead investigator for the clinical trial. “Most of the wounds missed by standard of care in this study had alarmingly high bacterial loads, indicative of asymptomatic infection. Use of the MolecuLight i:X enabled earlier detection of bacterial burden in wounds that would have otherwise been missed by standard of care assessment; we saw this improvement in detection across all wound types. The information provided by fluorescence imaging had utility across in all aspects of wound care, influencing treatment planning and overall patient care.
I believe that the MolecuLight i:X will quickly become a standard of care in wound care and play a pivotal role in antimicrobial stewardship in the wound clinic.”
“As an investigator in this trial and a user of MolecuLight i:X in my routine clinical practice, I can attest to the clinical benefit that fluorescence imaging provides. Identifying the presence and location of significant bacteria facilitates optimal preparation and treatment of my patients’ wounds enables me to quickly course correct a patient’s treatment when needed. With this immediate diagnostic feedback on bacteria and infection available at point-of-care, I no longer need to wait for weeks to determine if treatments are effective”, says Windy Cole, director of Wound Care Research, Kent State University College of Podiatric Medicine. “The results of this robust clinical trial add to the large body of evidence from prior studies, including a study I previously published where I used fluorescence imaging of bacteria to guide debridement and select appropriate treatments that led to accelerated wound healing”.
Up to 15% of Medicare beneficiaries had at least one type of wound or wound-related infection, and the prevalence of chronic wounds continues to increase each year. Without objective methods to detect bacterial burden in wounds, wound healing stalls and inappropriate treatments are applied, further inflating these costs. “Results from this trial reveal the signification proportion of chronic wounds with high bacterial burden that are missed by standard of care assessment of clinical signs and symptoms,” says Anil Amlani, CEO of MolecuLight Inc. “The results of this study show how use of point-of-care fluorescence imaging enables more accurate detection of bacterial burden in wounds. The diagnostic information provided by the device has the potential to fundamentally change the paradigm in wound care, leading to improved healing rates, reduced costs, and improved patient outcomes”.
The early results of this FLAAG trial were shared with the AMA and CMS, who, after a critical review of the large body of supporting clinical evidence, issued two CPT codes (Category III) for physician work and facility payment for Hospital Outpatient Department (HOPD) and Ambulatory Surgical Center (ASC) settings through an Ambulatory Payment Classification (APC) assignment. These new codes were issued by AMA and CMS in recognition of the medical necessity of this MolecuLight fluorescence imaging procedure.