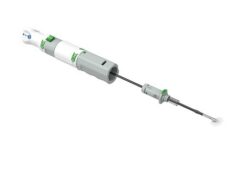
Vasorum has received approval of its premarket approval application from the US Food and Drug Administration (FDA). Celt ACD, which has received CE mark and is sold in Europe, is indicated for arterial puncture closure in both diagnostic and anticoagulated percutaneous interventional cardiology and radiology patients.
Vasorum says that the Celt ACD “offers excellent time to haemostasis in a wide variety of clinical situations”. A randomised controlled clinical trial which recruited 207 interventional cardiology procedure patients was carried out in four international cardiology centres across the USA and Europe. The trial’s principal investigator was Shing-Chiu Wong, director of cardiac catheterisation at the New York-Presbyterian/Weill Cornell Medical Center. Wong commented that he “was very pleased with the positive outcome of the clinical trial which shows that Celt ACD can help in addressing the clear need for quicker and more efficient methods of increasing patient throughput in healthcare facilities.”
There are currently over eight million catheter procedures performed annually, which support an estimated US$1bn femoral artery closure device market. The number of procedures is expected to exceed 10 million by 2020. In addition to interventional cardiology procedures, the market growth is being driven by an increasing number of peripheral vascular, neuro-vascular and other catheter procedures which demand more patient friendly devices and more efficient patient discharge from hospitals. “Given its ease of use and wide clinical applicability,” a Vasorum press release suggests, “Celt ACD is well positioned to address this broad and growing market opportunity.”
“With more than 20,000 patient implants to date in Europe, Celt ACD has proven itself to be a best-in-class arterial puncture closure device. Celt ACD allows immediate closure of multiple re-sticks in calcified vessels and is also very comfortable for patients. The FDA approval is a very significant milestone allowing US market entry by Vasorum,” stated James Coleman co-founder and chief executive officer of Vasorum.